Abstract
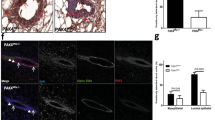
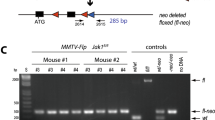
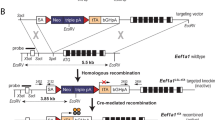
Aurora-A, a serine/threonine mitotic kinase, was reported to be overexpressed in various human cancers, and its overexpression induces aneuploidy, centrosome amplification and tumorigenic transformation in cultured human and rodent cells. However, the underlying mechanisms and pathological settings by which Aurora-A promotes tumorigenesis are largely unknown. Here, we created a transgenic mouse model to investigate the involvement of Aurora-A overexpression in the development of mammary glands and tumorigenesis using a Cre-loxP system. The conditional expression of Aurora-A resulted in significantly increased binucleated cell formation and apoptosis in the mammary epithelium. The surviving mammary epithelial cells composed hyperplastic areas after a short latency. Induction of Aurora-A overexpression in mouse embryonic fibroblasts prepared from the transgenic mice also led to aberrant mitosis and binucleated cell formation followed by apoptosis. The levels of p53 protein were remarkably increased in these Aurora-A-overexpressing cells, and the apoptosis was significantly suppressed by deletion of p53. Given that no malignant tumor formation was found in the Aurora-A-overexpressing mouse model after a long latency, additional factors, such as p53 inactivation, are required for the tumorigenesis of Aurora-A-overexpressing mammary epithelium. Our findings indicated that this mouse model is a useful system to study the physiological roles of Aurora-A and the genetic pathways of Aurora-A-induced carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- CAG:
-
chicken beta-actin
- CAT:
-
chloramphenicol acetyltransferase
- TUNEL:
-
TdT-mediated dUTP-biotin nick-end labeling
- MEFs:
-
mouse embryonic fibroblasts
- FITC:
-
fluorescein isothiocyanate
- FACS:
-
fluorescence-activated cell sorting
References
Anand S, Penrhyn-Lowe S and Venkitaraman AR . (2003). Cancer Cell, 3, 51–62.
Araki K, Araki M, Miyazaki J and Vassalli P . (1995). Proc. Natl. Acad. Sci. USA, 92, 160–164.
Baudino TA, Maclean KH, Brennan J, Parganas E, Yang C, Aslanian A, Lees JA, Sherr CJ, Roussel MF and Cleveland JL . (2003). Mol. Cell, 11, 905–914.
Bischoff JR, Anderson L, Zhu Y, Mossie K, Ng L, Souza B, Schryver B, Flanagan P, Clairvoyant F, Ginther C, Chan CS, Novotny M, Slamon DJ and Plowman GD . (1998). EMBO J., 17, 3052–3065.
Bischoff JR and Plowman GD . (1999). Trends Cell Biol., 9, 454–459.
Borel F, Lohez OD, Lacroix FB and Margolis RL . (2002). Proc. Natl. Acad. Sci. USA, 99, 9819–9824.
Cahill DP, Kinzler KW, Vogelstein B and Lengauer C . (1999). Trends Cell Biol., 9, M57–M60.
Cardiff RD and Wellings SR . (1999). J. Mammary Gland Biol. Neoplasia, 4, 105–122.
Castro A, Arlot-Bonnemains Y, Vigneron S, Labbe JC, Prigent C and Lorca T . (2002). EMBO Rep., 3, 457–462.
Chan CS and Botstein D . (1993). Genetics, 135, 677–691.
Chen SS, Chang PC, Cheng YW, Tang FM and Lin YS . (2002). EMBO J., 21, 4491–4499.
Evan GI, Wyllie AH, Gilbert CS, Littlewood TD, Land H, Brooks M, Waters CM, Penn LZ and Hancock DC . (1992). Cell, 69, 119–128.
Ewart-Toland A, Briassouli P, de Koning JP, Mao JH, Yuan J, Chan F, MacCarthy-Morrogh L, Ponder BA, Nagase H, Burn J, Ball S, Almeida M, Linardopoulos S and Balmain A . (2003). Nat. Genet., 34, 403–412.
Francisco L and Chan CS . (1994). Cell Mol. Biol. Res., 40, 207–213.
Glover DM, Leibowitz MH, McLean DA and Parry H . (1995). Cell, 81, 95–105.
Gritsko TM, Coppola D, Paciga JE, Yang L, Sun M, Shelley SA, Fiorica JV, Nicosia SV and Cheng JQ . (2003). Clin. Cancer Res., 9, 1420–1426.
Hermeking H and Eick D . (1994). Science, 265, 2091–2093.
Hirota T, Kunitoku N, Sasayama T, Marumoto T, Zhang D, Nitta M, Hatakeyama K and Saya H . (2003). Cell, 114, 585–598.
Hogan B, Beddington R, Costantini F and Lacy E . (1994). Manipulating the Mouse Embryo: A Laboratory Manual 2nd edn. Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY.
Honda K, Mihara H, Kato Y, Yamaguchi A, Tanaka H, Yasuda H, Furukawa K and Urano T . (2000). Oncogene, 19, 2812–2819.
Kallioniemi A, Kallioniemi OP, Piper J, Tanner M, Stokke T, Chen L, Smith HS, Pinkel D, Gray JW and Waldman FM . (1994). Proc. Natl. Acad. Sci. USA, 91, 2156–2160.
Kanegae Y, Lee G, Sato Y, Tanaka M, Nakai M, Sakaki T, Sugano S and Saito I . (1995). Nucleic Acids Res., 23, 3816–3821.
Katayama H, Sasai K, Kawai H, Yuan ZM, Bondaruk J, Suzuki F, Fujii S, Arlinghaus RB, Czerniak BA and Sen S . (2004). Nat. Genet., 36, 55–62.
Lengauer C, Kinzler KW and Vogelstein B . (1998). Nature, 396, 643–649.
Li D, Zhu J, Firozi PF, Abbruzzese JL, Evans DB, Cleary K, Friess H and Sen S . (2003). Clin. Cancer Res., 9, 991–997.
Lund LR, Romer J, Thomasset N, Solberg H, Pyke C, Bissell MJ, Dano K and Werb Z . (1996). Development, 122, 181–193.
Margolis RL, Lohez OD and Andreassen PR . (2003). J. Cell Biochem., 88, 673–683.
Marumoto T, Hirota T, Morisaki T, Kunitoku N, Zhang D, Ichikawa Y, Sasayama T, Kuninaka S, Mimori T, Tamaki N, Kimura M, Okano Y and Saya H . (2002). Genes Cells, 7, 1173–1182.
Marumoto T, Honda S, Hara T, Nitta M, Hirota T, Kohmura E and Saya H . (2003). J. Biol. Chem., 278, 51786–51795.
Meraldi P, Honda R and Nigg EA . (2002). EMBO J., 21, 483–492.
Miyoshi Y, Iwao K, Egawa C and Noguchi S . (2001). Int. J. Cancer, 92, 370–373.
Nigg EA . (2001). Nat. Rev. Mol. Cell. Biol., 2, 21–32.
Sakakura C, Hagiwara A, Yasuoka R, Fujita Y, Nakanishi M, Masuda K, Shimomura K, Nakamura Y, Inazawa J, Abe T and Yamagishi H . (2001). Br. J. Cancer, 84, 824–831.
Schlegel J, Stumm G, Scherthan H, Bocker T, Zirngibl H, Ruschoff J and Hofstadter F . (1995). Cancer Res., 55, 6002–6005.
Sen S, Zhou H and White RA . (1997). Oncogene, 14, 2195–2200.
Tanaka T, Kimura M, Matsunaga K, Fukada D, Mori H and Okano Y . (1999). Cancer Res., 59, 2041–2044.
Tsukada T, Tomooka Y, Takai S, Ueda Y, Nishikawa S, Yagi T, Tokunaga T, Takeda N, Suda Y, Abe S, Mastuo I, Ikawa Y and Aizawa S . (1993). Oncogene, 8, 3313–3322.
Uetake Y and Sluder G . (2004). J. Cell Biol., 165, 609–615.
Vogel C, Kienitz A, Hofmann I, Muller R and Bastians H . (2004). Oncogene, 23, 6845–6853.
Wagner AJ, Kokontis JM and Hay N . (1994). Genes Dev., 8, 2817–2830.
Wagner KU, Wall RJ, St-Onge L, Gruss P, Wynshaw-Boris A, Garrett L, Li M, Furth PA and Hennighausen L . (1997). Nucleic Acids Res., 25, 4323–4330.
Xu X, Wagner KU, Larson D, Weaver Z, Li C, Ried T, Hennighausen L, Wynshaw-Boris A and Deng CX . (1999). Nat. Genet., 22, 37–43.
Zhou H, Kuang J, Zhong L, Kuo WL, Gray JW, Sahin A, Brinkley BR and Sen S . (1998). Nat. Genet., 20, 189–193.
Zindy F, Eischen CM, Randle DH, Kamijo T, Cleveland JL, Sherr CJ and Roussel MF . (1998). Genes Dev., 12, 2424–2433.
Acknowledgements
We thank Dr Kimi Araki (Kumamoto University) for providing pCAG-CAT-lacZ plasmid; Mr Takenobu Nakagawa (Kumamoto University) for technical assistance; Drs Izumu Saito and Yumi Kanegae (University of Tokyo) for providing adenoviral luciferase and AxCANCre virus; members of the Saya lab for valuable suggestions; and members of the Gene Technology Center at Kumamoto University for their contributions to the technical assistance. This work was supported by the Research for the Future program of the Japan Society for the promotion of Science and by a grant for Cancer Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (to HS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, D., Hirota, T., Marumoto, T. et al. Cre-loxP-controlled periodic Aurora-A overexpression induces mitotic abnormalities and hyperplasia in mammary glands of mouse models. Oncogene 23, 8720–8730 (2004). https://doi.org/10.1038/sj.onc.1208153
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208153
Keywords
This article is cited by
-
Aurora kinase A inhibition induces synthetic lethality in SMAD4-deficient colorectal cancer cells via spindle assembly checkpoint activation
Oncogene (2022)
-
Aneuploidy as a promoter and suppressor of malignant growth
Nature Reviews Cancer (2021)
-
Bora phosphorylation substitutes in trans for T-loop phosphorylation in Aurora A to promote mitotic entry
Nature Communications (2021)
-
The therapeutic potential of Aurora kinases targeting in glioblastoma: from preclinical research to translational oncology
Journal of Molecular Medicine (2020)
-
Insights into the non-mitotic functions of Aurora kinase A: more than just cell division
Cellular and Molecular Life Sciences (2020)