Abstract
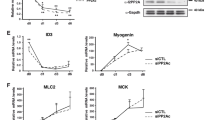
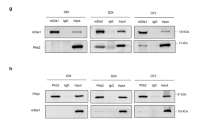
Cdk5 regulates myogenesis but the signaling cascade through which Cdk5 modulates this process remains to be characterized. Here, we investigated whether PI3K, Akt, p70S6K, p38 MAPK, p44/42 MAPK, and Egr-1 serve as upstream regulators of Cdk5 during L6 myoblast differentiation. Upon serum reduction, we found that besides elevated expression of Cdk5 and its activator, p35, and increased Cdk5/p35 activity, Egr-1, Akt, p70S6K, and p38 MAPK activity were upregulated in differentiating L6 cells. However, p44/42 MAPK was downregulated and SAPK/JNK was unaffected. LY294002, a PI3K inhibitor, blocked the activation of Akt and p70S6K, indicating that Akt and p70S6K activation is linked to PI3K activation. The lack of LY294002 effect on p38 MAPK suggests that p38 MAPK activation is not associated with PI3K activation. Rapamycin, a specific inhibitor of FRAP/mTOR (the upstream kinase of p70S6K), also blocked p70S6K activation, indicating the involvement of FRAP/mTOR activation. LY294002 and rapamycin also blocked the enhancement of Egr-1 level, Cdk5 activity, and myogenin expression, suggesting that upregulation of these factors is coupled to PI3K–p70S6K activation. Overexpression of dominant-negative-Akt also reduced Cdk5/p35 activity and myogenin expression, indicating that the PI3K–p70S6K–Egr-1–Cdk5 signaling cascade is linked to Akt activation. SB2023580, a p38 MAPK inhibitor, had no effect on p70S6K, Egr-1, or Cdk5 activity, suggesting that p38 MAPK activation lies in a pathway distinct from the PI3K–Akt–p70S6K–Egr-1 pathway that we identify as the upstream modulator of Cdk5 activity during L6 myoblast differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Agullo G, Gamet-Payrastre L, Manenti S, Viala C, Remesy C, Chap H and Payrastre B . (1997). Biochem. Pharmacol., 53, 1649–1657.
Arnold HH, Braun T, Bober E, Buchberger A, Winter B and Salminen A . (1992). Symp. Soc. Exp. Biol., 46, 37–51.
Cao XM, Koski RA, Gashler A, McKiernan M, Morris CF, Gaffney R, Hay RV and Sukhatme VP . (1990). Mol. Cell Biol., 10, 1931–1939.
Chen F and Studzinski GP . (2001). Blood, 97, 3763–3767.
Chung J, Grammer TC, Lemon KP, Kazlauskas A and Blenis J . (1994). Nature (London), 370, 71–75.
Conejo R, Alvaro C, Benito M, Cuadrado A and Lorenzo M . (2002). Oncogene, 21, 3739–3753.
Coolican SA, Samuel DS, Ewton DZ, Clade FJ and Florini JR . (1997). J. Biol. Chem., 272, 6653–6662.
Datta K, Bellacosa A, Chan TO and Tsichlis PN . (1996). J. Biol. Chem., 271, 30835–30839.
Davies SP, Reddy H, Caivano M and Cohen P . (2000). Biochem. J., 351, 95–105.
Dhavan R and Tsai LH . (2001). Nat. Rev. Mol. Cell Biol., 10, 749–759.
Gao CY, Zakeri Z, Zhu Y, He H and Zelenka PS . (1997). Dev. Genet., 20, 267–275.
Gredinger E, Gerber AN, Tamir Y, Tapscott SJ and Bengal E . (1998). J. Biol. Chem., 273, 10436–10444.
Guidato S, McLoughlin DM, Grierson AJ and Miller CC . (1998). J. Neurochem., 70, 335–340.
Han JW, Pearson RB, Dennis PB and Thomas G . (1995). J. Biol. Chem., 270, 21396–21403.
Harada S, Esch GL, Holgado-Madruga M and Wong AJ . (2001a). DNA Cell Biol., 20, 223–229.
Harada T, Morooka T, Ogawa S and Nishida E . (2001b). Nat. Cell Biol., 3, 453–459.
Jones NC, Fedorov YV, Rosenthal RS and Olwin BB . (2001). J. Cell Physiol., 186, 104–115.
Kim S, Jung Y, Kim D, Koh H and Chung J . (2000). J. Biol. Chem., 275, 25979–25984.
Kitzmann M and Fernandez A . (2001). Cell. Mol. Life Sci., 58, 571–579.
Konishi H, Matsuzaki H, Tanaka M, Ono Y, Tokunaga C, Kuroda S and Kikkawa U . (1996). Proc. Natl. Acad. Sci. USA, 93, 7639–7643.
Laprise P, Poirier EM, Vezina A, Rivard N and Vachon PH . (2002). J. Cell Physiol., 191, 69–81.
Lazaro JB, Kitzmann M, Poul MA, Vandromme M, Lamb NJ and Fernandez A . (1997). J. Cell Sci., 110, 1251–1260.
Lechner C, Zahalka MA, Giot JF, Moller NP and Ullrich A . (1996). Proc. Natl. Acad. Sci. USA, 93, 4355–4359.
Lee K-Y, Qi Z, Yu YP and Wang JH . (1997). Int. J. Biochem. Cell Biol., 29, 951–958.
Lee K-Y, Rosales JL, Tang D and Wang JH . (1996). J. Biol. Chem., 271, 1538–1543.
Lew J and Wang JH . (1995). Trends Biochem. Sci., 20, 33–37.
Liu JW, Lacy J, Sukhatme VP and Coleman DL . (1991). J. Biol. Chem., 266, 5929–5933.
Miyajima M, Nornes HO and Neuman T . (1995). NeuroReport, 6, 1130–1136.
Morgan DO . (1997). Annu. Rev. Cell Dev. Biol., 13, 261–291.
Moule SK, Welsh GI, Edgell NJ, Foulstone EJ, Proud CG and Denton RM . (1997). J. Biol. Chem., 272, 7713–7719.
Ostrovsky O and Bengal E . (2003). J. Biol. Chem., 278, 21221–21231.
Philpott A, Porro EB, Kirschner MW and Tsai LH . (1997). Genes Dev., 11, 1409–1421.
Reif K, Burgering BM and Cantrell DA . (1997). J. Biol. Chem., 272, 14426–14433.
Reusch HP, Zimmermann S, Schaefer M, Paul M and Moelling K . (2001). J. Biol. Chem., 276, 33630–33637.
Rommel C, Clarke BA, Zimmermann S, Nunez L, Rossman R, Reid K, Moelling K, Yancopoulos GD and Glass DJ . (1999). Science, 286, 1738–1741.
Rosales JL, Lee BC, Modarressi M, Sarker K, Lee KY, Jeong YG, Oko R and Lee K-Y . (2004). J. Biol. Chem., 279, 1224–1232.
Sable CL, Flippase N, Hemming B and Van Obberghen E . (1997). FEBS Lett., 409, 253–257.
Sarker KP, Biswas KK, Yamakuchi M, Lee K-Y, Hahiguchi T, Kracht M, Kitajima I and Maruyama I . (2003). J. Neurochem., 85, 50–61.
Sarker KP, Nakata M, Kitajima I, Nakajima T and Maruyama I . (2000). J. Mol. Neurosci., 15, 243–250.
Scheid MP, Marignani PA and Woodgett JR . (2002). Mol. Cell Biol., 22, 6247–6260.
Tamir Y and Bengal E . (2000). J. Biol. Chem., 275, 34424–34432.
Tureckova J, Wilson EM, Cappalonga JL and Rotwein P . (2001). J. Biol. Chem., 276, 39264–39270.
Winter B and Arnold HH . (2000). J. Cell Sci., 113, 4211–4220.
Xiong Y, Zhang H and Beach D . (1992). Cell, 71, 505–514.
Zetser A, Frank D and Bengal E . (2001). Dev. Biol., 240, 168–181.
Zhang H, Xiong Y and Beach D . (1993). Mol. Biol. Cell, 9, 897–906.
Acknowledgements
We thank Dr Jim Woodgett (University of Toronto) for providing the mutant and wild-type Akt expressing plasmids. We also thank Dr Ji Won Yoon (University of Calgary) for providing us L6 cells. KPS is an Alberta Cancer Board postdoctoral fellow. This work was supported by a grant from the Canadian Institutes of Health Research (CIHR) to KYL, an Alberta Heritage Foundation for Medical Research (AHFMR) Senior Scholar.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarker, K., Lee, KY. L6 myoblast differentiation is modulated by Cdk5 via the PI3K–AKT–p70S6K signaling pathway. Oncogene 23, 6064–6070 (2004). https://doi.org/10.1038/sj.onc.1207819
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207819
Keywords
This article is cited by
-
Cyclin-dependent kinase 5 mediates pleiotrophin-induced endothelial cell migration
Scientific Reports (2018)
-
The myogenic kinome: protein kinases critical to mammalian skeletal myogenesis
Skeletal Muscle (2011)
-
The phosphoinositide 3-kinase/Akt pathway is essential for the retinoic acid-induced differentiation of F9 cells
Oncogene (2006)