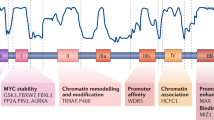
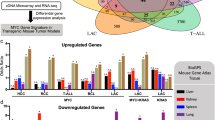
Abstract
A paradox for the cancer biology field has been the revelation that oncogenes, once thought to simply provide advantages to a cancer cell, actually put it at dire risk of cell suicide. Myc is the quintessential oncogene in this respect, as in normal cells it is required for cell cycle traverse, whereas in cancers it is overexpressed and functions as the angiogenic switch. Nonetheless, Myc overexpression kills normal cells dead in their tracks. Here we review Myc-induced pathways that contribute to the apoptotic response. Molecular analysis of Myc-induced tumors has established that some of these apoptotic pathways are essential checkpoints that guard the cell from cancer, as they are selectively bypassed during tumorigenesis. The precise mechanism(s) by which Myc targets these pathways are largely unresolved, but we propose that they involve crosstalk and feedback regulatory loops between arbiters of cell death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Abrahams VM, Kamsteeg M and Mor G . (2003). Mol. Biotechnol., 25, 19–30.
Adams JM, Harris AW, Pinkert CA, Corcoran LM, Alexander WS, Cory S, Palmiter RD and Brinster RL . (1985). Nature, 318, 533–538.
Alarcon-Vargas D, Tansey WP and Ronai Z . (2002). Oncogene, 21, 4384–4391.
Alitalo K, Bishop JM, Smith DH, Chen EY, Colby WW and Levinson AD . (1983). Proc. Natl. Acad. Sci. USA, 80, 100–104.
Alt JR, Greiner TC, Cleveland JL and Eischen CM . (2003). EMBO J., 22, 1442–1450.
Amanullah A, Liebermann DA and Hoffman B . (2002). Oncogene, 21, 1600–1610.
Amati B, Littlewood TD, Evan GI and Land H . (1993). EMBO J., 12, 5083–5087.
Antonsson B, Montessuit S, Sanchez B and Martinou JC . (2001). J. Biol. Chem., 276, 11615–11623.
Ashe PC and Berry MD . (2003). Prog. Neuropsychopharmacol. Biol. Psychiatry, 27, 199–214.
Askew DS, Ashmun RA, Simmons BC and Cleveland JL . (1991). Oncogene, 6, 1915–1922.
Ayer DE, Kretzner L and Eisenman RN . (1993). Cell, 72, 211–222.
Ayer DE, Laherty CD, Lawrence QA, Armstrong AP and Eisenman RN . (1996). Mol. Cell. Biol., 16, 5772–5781.
Ayer DE, Lawrence QA and Eisenman RN . (1995). Cell, 80, 767–776.
Bates S, Phillips AC, Clark PA, Stott F, Peters G, Ludwig RL and Vousden KH . (1998). Nature, 395, 124–125.
Baudino TA and Cleveland JL . (2001). Mol. Cell. Biol., 21, 691–702.
Baudino TA, Maclean KH, Brennan J, Parganas E, Yang C, Aslanian A, Lees JA, Sherr CJ, Roussel MF and Cleveland JL . (2003). Mol. Cell., 11, 905–914.
Baudino TA, McKay C, Pendeville-Samain H, Nilsson JA, Maclean KH, White EL, Davis AC, Ihle JN and Cleveland JL . (2002). Genes Dev., 16, 2530–2543.
Beg AA, Sha WC, Bronson RT, Ghosh S and Baltimore D . (1995). Nature, 376, 167–170.
Bellmeyer A, Krase J, Lindgren J and LaBonne C . (2003). Dev. Cell, 4, 827–839.
Bhatia K, Huppi K, Spangler G, Siwarski D, Iyer R and Magrath I . (1993). Nat. Genet., 5, 56–61.
Biswas SC and Greene LA . (2002). J. Biol. Chem., 277, 49511–49516.
Blackwell TK, Huang J, Ma A, Kretzner L, Alt FW, Eisenman RN and Weintraub H . (1993). Mol. Cell. Biol., 13, 5216–5224.
Blackwell TK, Kretzner L, Blackwood EM, Eisenman RN and Weintraub H . (1990). Science, 250, 1149–1151.
Blackwood EM and Eisenman RN . (1991). Science, 251, 1211–1217.
Blackwood EM, Luscher B and Eisenman RN . (1992). Genes Dev., 6, 71–80.
Boon K, Caron HN, van Asperen R, Valentijn L, Hermus MC, van Sluis P, Roobeek I, Weis I, Voute PA, Schwab M and Versteeg R . (2001). EMBO J., 20, 1383–1393.
Bowen H, Biggs TE, Phillips E, Baker ST, Perry VH, Mann DA and Barton CH . (2002). J. Biol. Chem., 277, 34997–35006.
Breitschopf K, Haendeler J, Malchow P, Zeiher AM and Dimmeler S . (2000). Mol. Cell. Biol., 20, 1886–1896.
Brummelkamp TR, Kortlever RM, Lingbeek M, Trettel F, MacDonald ME, van Lohuizen M and Bernards R . (2002). J. Biol. Chem., 277, 6567–6572.
Brunner T, Kasibhatla S, Pinkoski MJ, Frutschi C, Yoo NJ, Echeverri F, Mahboubi A and Green DR . (2000). J. Biol. Chem., 275, 9767–9772.
Burgering BM and Medema RH . (2003). J. Leukoc. Biol., 73, 689–701.
Burns TF and El-Deiry WS . (2003). Cancer Biol. Ther., 2, 431–443.
Bush A, Mateyak M, Dugan K, Obaya A, Adachi S, Sedivy J and Cole M . (1998). Genes Dev., 12, 3797–3802.
Cameron ER, Morton J, Johnston CJ, Irvine J, Bell M, Onions DE, Neil JC, Campbell M and Blyth K . (2000). Cell Death Differ., 7, 80–88.
Carlson H, Ota S, Song Y, Chen Y and Hurlin PJ . (2002). Oncogene, 21, 3827–3835.
Cavalieri F and Goldfarb M . (1987). Mol. Cell. Biol., 7, 3554–3560.
Cavalieri F and Goldfarb M . (1988). Oncogene, 2, 289–291.
Channavajhala P and Seldin DC . (2002). Oncogene, 21, 5280–5288.
Charron J, Malynn BA, Fisher P, Stewart V, Jeannotte L, Goff SP, Robertson EJ and Alt FW . (1992). Genes Dev., 6, 2248–2257.
Chen J, Marechal V and Levine AJ . (1993). Mol. Cell. Biol., 13, 4107–4114.
Chen MK, Strande LF, Beierle EA, Kain MS, Geldziler BD and Doolin EJ . (1999). J. Surg. Res., 84, 82–87.
Cheng EH, Kirsch DG, Clem RJ, Ravi R, Kastan MB, Bedi A, Ueno K and Hardwick JM . (1997). Science, 278, 1966–1968.
Cohen O, Inbal B, Kissil JL, Raveh T, Berissi H, Spivak-Kroizaman T, Feinstein E and Kimchi A . (1999). J. Cell. Biol., 146, 141–148.
Cole MD and McMahon SB . (1999). Oncogene, 18, 2916–2924.
Coller HA, Grandori C, Tamayo P, Colbert T, Lander ES, Eisenman RN and Golub TR . (2000). Proc. Natl. Acad. Sci. USA, 97, 3260–3265.
Condorelli F, Salomoni P, Cotteret S, Cesi V, Srinivasula SM, Alnemri ES and Calabretta B . (2001). Mol. Cell. Biol., 21, 3025–3036.
Conzen SD, Gottlob K, Kandel ES, Khanduri P, Wagner AJ, O'Leary M and Hay N . (2000). Mol. Cell. Biol., 20, 6008–6018.
Coppola JA and Cole MD . (1986). Nature, 320, 760–763.
Coultas L and Strasser A . (2003). Semin. Cancer Biol., 13, 115–123.
Creagh EM and Martin SJ . (2001). Biochem. Soc. Trans., 29, 696–702.
Cretney E, Takeda K, Yagita H, Glaccum M, Peschon JJ and Smyth MJ . (2002). J. Immunol., 168, 1356–1361.
Crews S, Barth R, Hood L, Prehn J and Calame K . (1982). Science, 218, 1319–1321.
Cross TG, Scheel-Toellner D, Henriquez NV, Deacon E, Salmon M and Lord JM . (2000). Exp. Cell. Res., 256, 34–41.
Dalla-Favera R, Bregni M, Erikson J, Patterson D, Gallo RC and Croce CM . (1982). Proc. Natl. Acad. Sci. USA, 79, 7824–7827.
Dang CV . (1999). Mol. Cell. Biol., 19, 1–11.
Datta SR, Brunet A and Greenberg ME . (1999). Genes Dev., 13, 2905–2927.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y and Greenberg ME . (1997). Cell, 91, 231–241.
Davis AC, Wims M, Spotts GD, Hann SR and Bradley A . (1993). Genes Dev., 7, 671–682.
de Alboran IM, O’Hagan RC, Gartner F, Malynn B, Davidson L, Rickert R, Rajewsky K, DePinho RA and Alt FW . (2001). Immunity, 14, 45–55.
de Stanchina E, McCurrach ME, Zindy F, Shieh SY, Ferbeyre G, Samuelson AV, Prives C, Roussel MF, Sherr CJ and Lowe SW . (1998). Genes Dev., 12, 2434–2442.
Debbas M and White E . (1993). Genes Dev., 7, 546–554.
DeGregori J, Leone G, Miron A, Jakoi L and Nevins JR . (1997). Proc. Natl. Acad. Sci. USA, 94, 7245–7250.
del Peso L, Gonzalez-Garcia M, Page C, Herrera R and Nunez G . (1997). Science, 278, 687–689.
Deverman BE, Cook BL, Manson SR, Niederhoff RA, Langer EM, Rosova I, Kulans LA, Fu X, Weinberg JS, Heinecke JW, Roth KA and Weintraub SJ . (2002). Cell, 111, 51–62.
Dimri GP, Itahana K, Acosta M and Campisi J . (2000). Mol. Cell. Biol., 20, 273–285.
Doi TS, Marino MW, Takahashi T, Yoshida T, Sakakura T, Old LJ and Obata Y . (1999). Proc. Natl. Acad. Sci. USA, 96, 2994–2999.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery Jr CA, Butel JS and Bradley A . (1992). Nature, 356, 215–221.
Dugan KA, Wood MA and Cole MD . (2002). Oncogene, 21, 5835–5843.
Duyao MP, Buckler AJ and Sonenshein GE . (1990). Proc. Natl. Acad. Sci. USA, 87, 4727–4731.
Earnshaw WC, Martins LM and Kaufmann SH . (1999). Annu. Rev. Biochem., 68, 383–424.
Eilers M, Schim S and Bishop JM . (1991). EMBO J, 10, 133–141.
Eischen CM, Packham G, Nip J, Fee BE, Hiebert SW, Zambetti GP and Cleveland JL . (2001a). Oncogene, 20, 6983–6993.
Eischen CM, Roussel MF, Korsmeyer SJ and Cleveland JL . (2001b). Mol. Cell. Biol., 21, 7653–7662.
Eischen CM, Weber JD, Roussel MF, Sherr CJ and Cleveland JL . (1999). Genes Dev., 13, 2658–2669.
Eischen CM, Woo D, Roussel MF and Cleveland JL . (2001c). Mol. Cell. Biol., 21, 5063–5070.
Ellis RE, Yuan JY and Horvitz HR . (1991). Annu. Rev. Cell. Biol., 7, 663–698.
Ellwood-Yen K, Graeber TG, Wongvipat J, Iruela-Arispe ML, Zhang J, Matusik R, Thomas GV and Sawyers CL . (2003). Cancer Cell, 4, 223–238.
Evan GI, Wyllie AH, Gilbert CS, Littlewood TD, Land H, Brooks M, Waters CM, Penn LZ and Hancock DC . (1992). Cell, 69, 119–128.
Fanidi A, Harrington EA and Evan GI . (1992). Nature, 359, 554–556.
Felsher DW and Bishop JM . (1999). Mol. Cell., 4, 199–207.
Fernandez PC, Frank SR, Wang L, Schroeder M, Liu S, Greene J, Cocito A and Amati B . (2003). Genes Dev., 17, 1115–1129.
Ferri KF and Kroemer G . (2001). Nat. Cell. Biol., 3, E255–263.
Fox CJ, Hammerman PS, Cinalli RM, Master SR, Chodosh LA and Thompson CB . (2003). Genes Dev., 17, 1841–1854.
Freedman DA and Levine AJ . (1998). Mol. Cell. Biol., 18, 7288–7293.
Freytag SO . (1988). Mol. Cell. Biol., 8, 1614–1624.
Fridman JS, Hernando E, Hemann MT, De Stanchina E, Cordon-Cardo C and Lowe SW . (2003). Cancer Res., 63, 5703–5706.
Giaccia AJ and Kastan MB . (1998). Genes Dev., 12, 2973–2983.
Godfried MB, Veenstra M, v Sluis P, Boon K, v Asperen R, Hermus MC, v Schaik BD, Voute TP, Schwab M, Versteeg R and Caron HN . (2002). Oncogene, 21, 2097–2101.
Gostissa M, Hengstermann A, Fogal V, Sandy P, Schwarz SE, Scheffner M and Del Sal G . (1999). EMBO J., 18, 6462–6471.
Grandori C, Cowley SM, James LP and Eisenman RN . (2000). Annu. Rev. Cell. Dev. Biol., 16, 653–699.
Green DR, Droin N and Pinkoski M . (2003). Immunol. Rev., 193, 70–81.
Griffith TS, Brunner T, Fletcher SM, Green DR and Ferguson TA . (1995). Science, 270, 1189–1192.
Grossman SR, Deato ME, Brignone C, Chan HM, Kung AL, Tagami H, Nakatani Y and Livingston DM . (2003). Science, 300, 342–344.
Grossman SR, Perez M, Kung AL, Joseph M, Mansur C, Xiao ZX, Kumar S, Howley PM and Livingston DM . (1998). Mol. Cell., 2, 405–415.
Groth A, Weber JD, Willumsen BM, Sherr CJ and Roussel MF . (2000). J. Biol. Chem., 275, 27473–27480.
Gu W and Roeder RG . (1997). Cell, 90, 595–606.
Haggerty TJ, Zeller KI, Osthus RC, Wonsey DR and Dang CV . (2003). Proc. Natl. Acad. Sci. USA, 100, 5313–5318.
Hahn P, Lindsten T, Ying GS, Bennett J, Milam AH, Thompson CB and Dunaief JL . (2003). Invest. Ophthalmol. Vis. Sci., 44, 3598–3605.
Han J, Flemington C, Houghton AB, Gu Z, Zambetti GP, Lutz RJ, Zhu L and Chittenden T . (2001). Proc. Natl. Acad. Sci. USA, 98, 11318–11323.
Harada H, Becknell B, Wilm M, Mann M, Huang LJ, Taylor SS, Scott JD and Korsmeyer SJ . (1999). Mol. Cell., 3, 413–422.
Harris MH and Thompson CB . (2000). Cell Death Differ., 7, 1182–1191.
He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT, Morin PJ, Vogelstein B and Kinzler KW . (1998). Science, 281, 1509–1512.
Heikkila R, Schwab G, Wickstrom E, Loke SL, Pluznik DH, Watt R and Neckers LM . (1987). Nature, 328, 445–449.
Heinzel T, Lavinsky RM, Mullen TM, Soderstrom M, Laherty CD, Torchia J, Yang WM, Brard G, Ngo SD, Davie JR, Seto E, Eisenman RN, Rose DW, Glass CK and Rosenfeld MG . (1997). Nature, 387, 43–48.
Hermeking H and Eick D . (1994). Science, 265, 2091–2093.
Herold S, Wanzel M, Beuger V, Frohme C, Beul D, Hillukkala T, Syvaoja J, Saluz HP, Haenel F and Eilers M . (2002). Mol. Cell., 10, 509–521.
Honda R, Tanaka H and Yasuda H . (1997). FEBS Lett., 420, 25–27.
Honda R and Yasuda H . (1999). EMBO J., 18, 22–27.
Huang E, Ishida S, Pittman J, Dressman H, Bild A, Kloos M, D'Amico M, Pestell RG, West M and Nevins JR . (2003). Nat. Genet., 34, 226–230.
Hueber AO, Zornig M, Lyon D, Suda T, Nagata S and Evan GI . (1997). Science, 278, 1305–1309.
Hurlin PJ, Queva C and Eisenman RN . (1997). Genes Dev., 11, 44–58.
Hurlin PJ, Queva C, Koskinen PJ, Steingrimsson E, Ayer DE, Copeland NG, Jenkins NA and Eisenman RN . (1995). EMBO J., 14, 5646–5659.
Hurlin PJ, Steingrimsson E, Copeland NG, Jenkins NA and Eisenman RN . (1999). EMBO J., 18, 7019–7028.
Hurlin PJ, Zhou ZQ, Toyo-Oka K, Ota S, Walker WL, Hirotsune S and Wynshaw-Boris A . (2003). EMBO J., 22, 4584–4596.
Iaccarino I, Hancock D, Evan G and Downward J . (2003). Cell Death Differ., 10, 599–608.
Inoue K, Roussel MF and Sherr CJ . (1999). Proc. Natl. Acad. Sci. USA, 96, 3993–3998.
Iritani BM, Delrow J, Grandori C, Gomez I, Klacking M, Carlos LS and Eisenman RN . (2002). EMBO J., 21, 4820–4830.
Jacks T, Remington L, Williams BO, Schmitt EM, Halachmi S, Bronson RT and Weinberg RA . (1994). Curr. Biol., 4, 1–7.
Jacobs JJ, Keblusek P, Robanus-Maandag E, Kristel P, Lingbeek M, Nederlof PM, van Welsem T, van de Vijver MJ, Koh EY, Daley GQ and van Lohuizen M . (2000). Nat. Genet., 26, 291–299.
Jacobs JJ, Kieboom K, Marino S, DePinho RA and van Lohuizen M . (1999a). Nature, 397, 164–168.
Jacobs JJ, Scheijen B, Voncken JW, Kieboom K, Berns A and van Lohuizen M . (1999b). Genes Dev., 13, 2678–2690.
Jacobsen KA, Prasad VS, Sidman CL and Osmond DG . (1994). Blood, 84, 2784–2794.
Jain M, Arvanitis C, Chu K, Dewey W, Leonhardt E, Trinh M, Sundberg CD, Bishop JM and Felsher DW . (2002). Science, 297, 102–104.
James MC and Peters G . (2000). Prog. Cell Cycle Res., 4, 71–81.
Jeffers JR, Parganas E, Lee Y, Yang C, Wang J, Brennan J, MacLean KH, Han J, Chittenden T, Ihle JN, McKinnon PJ, Cleveland JL and Zambetti GP . (2003). Cancer Cell, 4.
Jiang M and Milner J . (2003). Genes Dev., 17, 832–837.
Jones SN, Roe AE, Donehower LA and Bradley A . (1995). Nature, 378, 206–208.
Juin P, Hueber AO, Littlewood T and Evan G . (1999). Genes Dev., 13, 1367–1381.
Juin P, Hunt A, Littlewood T, Griffiths B, Swigart LB, Korsmeyer S and Evan G . (2002). Mol. Cell. Biol., 22, 6158–6169.
Kahyo T, Nishida T and Yasuda H . (2001). Mol. Cell, 8, 713–718.
Kamijo T, Bodner S, van de Kamp E, Randle DH and Sherr CJ . (1999). Cancer Res., 59, 2217–2222.
Kamijo T, Weber JD, Zambetti G, Zindy F, Roussel MF and Sherr CJ . (1998). Proc. Natl. Acad. Sci. USA, 95, 8292–8297.
Kamijo T, Zindy F, Roussel MF, Quelle DE, Downing JR, Ashmun RA, Grosveld G and Sherr CJ . (1997). Cell, 91, 649–659.
Kandasamy K, Srinivasula SM, Alnemri ES, Thompson CB, Korsmeyer SJ, Bryant JL and Srivastava RK . (2003). Cancer Res., 63, 1712–1721.
Kasibhatla S, Beere HM, Brunner T, Echeverri F and Green DR . (2000). Curr. Biol., 10, 1205–1208.
Kauffmann-Zeh A, Rodriguez-Viciana P, Ulrich E, Gilbert C, Coffer P, Downward J and Evan G . (1997). Nature, 385, 544–548.
Kelly K, Cochran BH, Stiles CD and Leder P . (1983). Cell, 35, 603–610.
Khosravi R, Maya R, Gottlieb T, Oren M, Shiloh Y and Shkedy D . (1999). Proc. Natl. Acad. Sci. USA, 96, 14973–14977.
Kimchi A . (1998). Biochim. Biophys. Acta, 1377, F13–33.
Kime L and Wright SC . (2003). Biochem. J., 370, 291–298.
Klefstrom J, Arighi E, Littlewood T, Jaattela M, Saksela E, Evan GI and Alitalo K . (1997). EMBO. J., 16, 7382–7392.
Klefstrom J, Vastrik I, Saksela E, Valle J, Eilers M and Alitalo K . (1994). EMBO J., 13, 5442–5450.
Klefstrom J, Verschuren EW and Evan G . (2002). J. Biol. Chem., 277, 43224–43232.
Kogel D, Reimertz C, Dussmann H, Mech P, Scheidtmann KH and Prehn JH . (2003). Eur. J. Cancer, 39, 249–256.
Kohl NE, Kanda N, Schreck RR, Bruns G, Latt SA, Gilbert F and Alt FW . (1983). Cell, 35, 359–367.
Kowalik TF, DeGregori J, Schwarz JK and Nevins JR . (1995). J. Virol., 69, 2491–2500.
Lane DP . (1992). Nature, 358, 15–16.
Lawlor MA and Alessi DR . (2001). J. Cell. Sci., 114, 2903–2910.
Leder A, Pattengale PK, Kuo A, Stewart TA and Leder P . (1986). Cell, 45, 485–495.
Leone G, DeGregori J, Sears R, Jakoi L and Nevins JR . (1997). Nature, 387, 422–426.
Leone G, Sears R, Huang E, Rempel R, Nuckolls F, Park CH, Giangrande P, Wu L, Saavedra HI, Field SJ, Thompson MA, Yang H, Fujiwara Y, Greenberg ME, Orkin S, Smith C and Nevins JR . (2001). Mol. Cell, 8, 105–113.
Levine AJ . (1997). Cell, 88, 323–331.
Li H, Zhu H, Xu CJ and Yuan J . (1998). Cell, 94, 491–501.
Li LY, Luo X and Wang X . (2001). Nature, 412, 95–99.
Li Q and Zhu GD . (2002). Curr. Top. Med. Chem., 2, 939–971.
Lin AW and Lowe SW . (2001). Proc. Natl. Acad. Sci. USA, 98, 5025–5030.
Lindsten T, Ross AJ, King A, Zong WX, Rathmell JC, Shiels HA, Ulrich E, Waymire KG, Mahar P, Frauwirth K, Chen Y, Wei M, Eng VM, Adelman DM, Simon MC, Ma A, Golden JA, Evan G, Korsmeyer SJ, MacGregor GR and Thompson CB . (2000). Mol. Cell, 6, 1389–1399.
Lindstrom MS, Klangby U and Wiman KG . (2001). Oncogene, 20, 2171–2177.
Lindstrom MS and Wiman KG . (2003). Oncogene, 22, 4993–5005.
Lingbeek ME, Jacobs JJ and van Lohuizen M . (2002). J. Biol. Chem., 277, 26120–26127.
Littlewood TD, Hancock DC, Danielian PS, Parker MG and Evan GI . (1995). Nucleic Acids Res., 23, 1686–1690.
Liu X, Kim CN, Yang J, Jemmerson R and Wang X . (1996). Cell, 86, 147–157.
Lowe SW and Ruley HE . (1993). Genes Dev., 7, 535–545.
Maclean KH, Keller UB, Rodriguez-Galindo C, Nilsson JA and Cleveland JL . (2003). Mol. Cell Biol., 23, 7256–7270.
Maestro R, Dei Tos AP, Hamamori Y, Krasnokutsky S, Sartorelli V, Kedes L, Doglioni C, Beach DH and Hannon GJ . (1999). Genes Dev., 13, 2207–2217.
Mao DY, Watson JD, Yan PS, Barsyte-Lovejoy D, Khosravi F, Wong WW, Farnham PJ, Huang TH and Penn LZ . (2003). Curr. Biol., 13, 882–886.
Marcu KB, Patel AJ and Yang Y . (1997). Curr. Top. Microbiol. Immunol., 224, 47–56.
Maruyama K, Schiavi SC, Huse W, Johnson GL and Ruley HE . (1987). Oncogene, 1, 361–367.
Mateyak MK, Obaya AJ, Adachi S and Sedivy JM . (1997). Cell Growth Differ., 8, 1039–1048.
Matsuzaki Y, Nakayama K, Tomita T, Isoda M, Loh DY and Nakauchi H . (1997). Blood, 89, 853–862.
Maya R, Balass M, Kim ST, Shkedy D, Leal JF, Shifman O, Moas M, Buschmann T, Ronai Z, Shiloh Y, Kastan MB, Katzir E and Oren M . (2001). Genes Dev., 15, 1067–1077.
McKeller RN, Fowler JL, Cunningham JJ, Warner N, Smeyne RJ, Zindy F and Skapek SX . (2002). Proc. Natl. Acad. Sci. USA, 99, 3848–3853.
McMahon SB, Van Buskirk HA, Dugan KA, Copeland TD and Cole MD . (1998). Cell, 94, 363–374.
McMahon SB, Wood MA and Cole MD . (2000). Mol. Cell. Biol., 20, 556–562.
Menssen A and Hermeking H . (2002). Proc. Natl. Acad. Sci. USA, 99, 6274–6279.
Meroni G, Reymond A, Alcalay M, Borsani G, Tanigami A, Tonlorenzi R, Nigro CL, Messali S, Zollo M, Ledbetter DH, Brent R, Ballabio A and Carrozzo R . (1997). EMBO J., 16, 2892–2906.
Mikhailov V, Mikhailova M, Degenhardt K, Venkatachalam MA, White E and Saikumar P . (2003). J. Biol. Chem., 278, 5367–5376.
Mitchell KO, Ricci MS, Miyashita T, Dicker DT, Jin Z, Reed JC and El-Deiry WS . (2000). Cancer Res., 60, 6318–6325.
Miyashita T, Harigai M, Hanada M and Reed JC . (1994a). Cancer Res., 54, 3131–3135.
Miyashita T, Krajewski S, Krajewska M, Wang HG, Lin HK, Liebermann DA, Hoffman B and Reed JC . (1994b). Oncogene, 9, 1799–1805.
Miyashita T and Reed JC . (1995). Cell, 80, 293–299.
Moens CB, Auerbach AB, Conlon RA, Joyner AL and Rossant J . (1992). Genes Dev., 6, 691–704.
Momand J and Zambetti GP . (1997). J. Cell. Biochem., 64, 343–352.
Momand J, Zambetti GP, Olson DC, George D and Levine AJ . (1992). Cell, 69, 1237–1245.
Montes de Oca Luna R, Wagner DS and Lozano G . (1995). Nature, 378, 203–206.
Motoyama N, Wang F, Roth KA, Sawa H, Nakayama K, Negishi I, Senju S, Zhang Q and Fujii S et al. (1995). Science, 267, 1506–1510.
Muller S, Berger M, Lehembre F, Seeler JS, Haupt Y and Dejean A . (2000). J. Biol. Chem., 275, 13321–13329.
Nakano K and Vousden KH . (2001). Mol. Cell., 7, 683–694.
Nakayama K, Negishi I, Kuida K, Sawa H and Loh DY . (1994). Proc. Natl. Acad. Sci. USA, 91, 3700–3704.
Nau MM, Brooks BJ, Battey J, Sausville E, Gazdar AF, Kirsch IR, McBride OW, Bertness V, Hollis GF and Minna JD . (1985). Nature, 318, 69–73.
Neiman PE, Ruddell A, Jasoni C, Loring G, Thomas SJ, Brandvold KA, Lee R, Burnside J and Delrow J . (2001). Proc. Natl. Acad. Sci. USA, 98, 6378–6383.
Nicholson DW . (1999). Cell Death Differ., 6, 1028–1042.
Nilsson JA, Maclean KH, Keller UB, Pendeville H, Baudino TA and Cleveland JL . (2003). Mol. Cell. Biol., in press.
Noguchi K, Kokubu A, Kitanaka C, Ichijo H and Kuchino Y . (2001). Biochem. Biophys. Res. Commun., 281, 1313–1320.
Nomura T, Khan MM, Kaul SC, Dong HD, Wadhwa R, Colmenares C, Kohno I and Ishii S . (1999). Genes Dev., 13, 412–423.
Nunez G, Benedict MA, Hu Y and Inohara N . (1998). Oncogene, 17, 3237–3245.
O’Connell BC, Cheung AF, Simkevich CP, Tam W, Ren X, Mateyak MK and Sedivy JM . (2003). J. Biol. Chem., 278, 12563–12573.
Oda E, Ohki R, Murasawa H, Nemoto J, Shibue T, Yamashita T, Tokino T, Taniguchi T and Tanaka N . (2000). Science, 288, 1053–1058.
Orian A, van Steensel B, Delrow J, Bussemaker HJ, Li L, Sawado T, Williams E, Loo LW, Cowley SM, Yost C, Pierce S, Edgar BA, Parkhurst SM and Eisenman RN . (2003). Genes Dev., 17, 1101–1114.
Owen-Schaub LB, Zhang W, Cusack JC, Angelo LS, Santee SM, Fujiwara T, Roth JA, Deisseroth AB, Zhang WW and Kruzel E et al. (1995). Mol. Cell. Biol., 15, 3032–3040.
Owyang AM, Tumang JR, Schram BR, Hsia CY, Behrens TW, Rothstein TL and Liou HC . (2001). J. Immunol., 167, 4948–4956.
Packham G, White EL, Eischen CM, Yang H, Parganas E, Ihle JN, Grillot DA, Zambetti GP, Nunez G and Cleveland JL . (1998). Genes Dev., 12, 2475–2487.
Park J, Kunjibettu S, McMahon SB and Cole MD . (2001). Genes Dev., 15, 1619–1624.
Park J, Wood MA and Cole MD . (2002). Mol. Cell. Biol., 22, 1307–1316.
Peduto Eberl L, Guillou L, Saraga E, Schroter M, French LE, Tschopp J and Juillerat-Jeanneret L . (1999). Int. J. Cancer, 81, 772–778.
Pelengaris S, Khan M and Evan GI . (2002). Cell, 109, 321–334.
Pelengaris S, Littlewood T, Khan M, Elia G and Evan G . (1999). Mol. Cell, 3, 565–577.
Peukert K, Staller P, Schneider A, Carmichael G, Hanel F and Eilers M . (1997). EMBO J., 16, 5672–5686.
Popescu NC and Zimonjic DB . (2002). J. Cell. Mol. Med., 6, 151–159.
Prendergast GC . (1999). Oncogene, 18, 2967–2987.
Prendergast GC and Ziff EB . (1991). Science, 251, 186–189.
Putcha GV, Le S, Frank S, Besirli CG, Clark K, Chu B, Alix S, Youle RJ, LaMarche A, Maroney AC and Johnson Jr EM . (2003). Neuron, 38, 899–914.
Qin XQ, Livingston DM, Kaelin WG, Jr and Adams PD . (1994). Proc. Natl. Acad. Sci. USA, 91, 10918–10922.
Quelle DE, Zindy F, Ashmun RA and Sherr CJ . (1995). Cell, 83, 993–1000.
Rao L, Debbas M, Sabbatini P, Hockenbery D, Korsmeyer S and White E . (1992). Proc. Natl. Acad. Sci. USA, 89, 7742–7746.
Rathmell JC, Lindsten T, Zong WX, Cinalli RM and Thompson CB . (2002). Nat. Immunol., 3, 932–939.
Raveh T, Droguett G, Horwitz MS, DePinho RA and Kimchi A . (2001). Nat. Cell. Biol., 3, 1–7.
Reisman D, Elkind NB, Roy B, Beamon J and Rotter V . (1993). Cell Growth Differ., 4, 57–65.
Renan MJ . (1989). Cancer Lett., 47, 1–9.
Rocha S, Campbell KJ and Perkins ND . (2003). Mol. Cell, 12, 15–25.
Rodriguez MS, Desterro JM, Lain S, Midgley CA, Lane DP and Hay RT . (1999). EMBO J., 18, 6455–6461.
Rosenfeld ME, Prichard L, Shiojiri N and Fausto N. . (2000). Am. J. Pathol., 156, 997–1007.
Roth J, Dobbelstein M, Freedman DA, Shenk T and Levine AJ . (1998). EMBO J., 17, 554–564.
Rounbehler RJ, Rogers PM, Conti CJ and Johnson DG . (2002). Cancer Res., 62, 3276–3281.
Rounbehler RJ, Schneider-Broussard R, Conti CJ and Johnson DG . (2001). Oncogene, 20, 5341–5349.
Ruas M and Peters G . (1998). Biochim. Biophys. Acta, 1378, F115–177.
Ruggero D and Pandolfi PP . (2003). Nat. Rev. Cancer, 3, 179–192.
Russell JL, Powers JT, Rounbehler RJ, Rogers PM, Conti CJ and Johnson DG . (2002). Mol. Cell. Biol., 22, 1360–1368.
Sakaguchi K, Herrera JE, Saito S, Miki T, Bustin M, Vassilev A, Anderson CW and Appella E . (1998). Genes Dev., 12, 2831–2841.
Sawai S, Shimono A, Hanaoka K and Kondoh H . (1991). New Biol., 3, 861–869.
Schmitt CA, Fridman JS, Yang M, Baranov E, Hoffman RM and Lowe SW . (2002). Cancer Cell, 1, 289–298.
Schmitt CA, McCurrach ME, de Stanchina E, Wallace-Brodeur RR and Lowe SW . (1999). Genes Dev., 13, 2670–2677.
Schmidt D and Muller S . (2002). Proc. Natl. Acad. Sci. USA, 99, 2872–2877.
Schuldiner O and Benvenisty N . (2001). Oncogene, 20, 4984–4994.
Schwab M, Varmus HE, Bishop JM, Grzeschik KH, Naylor SL, Sakaguchi AY, Brodeur G and Trent J . (1984). Nature, 308, 288–291.
Scorrano L and Korsmeyer SJ . (2003). Biochem. Biophys. Res. Commun., 304, 437–444.
Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T and Korsmeyer SJ . (2003). Science, 300, 135–139.
Sears R, Leone G, DeGregori J and Nevins JR . (1999). Mol. Cell, 3, 169–179.
Sears R, Ohtani K and Nevins JR . (1997). Mol. Cell. Biol., 17, 5227–5235.
Sedger LM, Glaccum MB, Schuh JC, Kanaly ST, Williamson E, Kayagaki N, Yun T, Smolak P, Le T, Goodwin R and Gliniak B . (2002). Eur. J. Immunol., 32, 2246–2254.
Seeger RC, Brodeur GM, Sather H, Dalton A, Siegel SE, Wong KY and Hammond D . (1985). N. Engl. J. Med., 313, 1111–1116.
Seoane J, Le HV and Massague J . (2002). Nature, 419, 729–734.
Seoane J, Pouponnot C, Staller P, Schader M, Eilers M and Massague J . (2001). Nat. Cell. Biol., 3, 400–408.
Serrano M, Hannon GJ and Beach D . (1993). Nature, 366, 704–707.
Shan B and Lee WH . (1994). Mol. Cell. Biol., 14, 8166–8173.
Sheiness D and Bishop JM . (1979). J. Virol., 31, 514–521.
Sheiness D, Fanshier L and Bishop JM . (1978). J. Virol., 28, 600–610.
Sheiness DK, Hughes SH, Varmus HE, Stubblefield E and Bishop JM . (1980). Virology, 105, 415–424.
Sherr CJ . (1998). Genes Dev., 12, 2984–2991.
Sherr CJ . (2001). Nat. Rev. Mol. Cell. Biol., 2, 731–737.
Sherr CJ and DePinho RA . (2000). Cell, 102, 407–410.
Shi Y, Glynn JM, Guilbert LJ, Cotter TG, Bissonnette RP and Green DR . (1992). Science, 257, 212–214.
Shiraki K, Tsuji N, Shioda T, Isselbacher KJ and Takahashi H . (1997). Proc. Natl. Acad. Sci. USA, 94, 6420–6425.
Shurin GV, Gerein V, Lotze MT and Barksdale Jr EM . (1998). Nat. Immun., 16, 263–274.
Soengas MS, Alarcon RM, Yoshida H, Giaccia AJ, Hakem R, Mak TW and Lowe SW . (1999). Science, 284, 156–159.
Soengas MS, Capodieci P, Polsky D, Mora J, Esteller M, Opitz-Araya X, McCombie R, Herman JG, Gerald WL, Lazebnik YA, Cordon-Cardo C and Lowe SW . (2001). Nature, 409, 207–211.
Soucie EL, Annis MG, Sedivy J, Filmus J, Leber B, Andrews DW and Penn LZ . (2001). Mol. Cell. Biol., 21, 4725–4736.
Staller P, Peukert K, Kiermaier A, Seoane J, Lukas J, Karsunky H, Moroy T, Bartek J, Massague J, Hanel F and Eilers M . (2001). Nat. Cell. Biol., 3, 392–399.
Stanton BR, Reid SW and Parada LF . (1990). Mol. Cell. Biol., 10, 6755–6758.
Stewart M, Cameron E, Campbell M, McFarlane R, Toth S, Lang K, Onions D and Neil JC . (1993). Int. J. Cancer, 53, 1023–1030.
Stoneley M, Chappell SA, Jopling CL, Dickens M, MacFarlane M and Willis AE . (2000). Mol. Cell. Biol., 20, 1162–1169.
Strand S and Galle PR . (1998). Mol. Med. Today, 4, 63–68.
Strasser A, Harris AW, Bath ML and Cory S . (1990). Nature, 348, 331–333.
Sugimoto M, Kuo ML, Roussel MF and Sherr CJ . (2003). Mol. Cell, 11, 415–424.
Susin SA, Zamzami N, Castedo M, Hirsch T, Marchetti P, Macho A, Daugas E, Geuskens M and Kroemer G . (1996). J. Exp. Med., 184, 1331–1341.
Suzuki M, Youle RJ and Tjandra N . (2000). Cell, 103, 645–654.
Takahashi T, Tanaka M, Brannan CI, Jenkins NA, Copeland NG, Suda T and Nagata S . (1994). Cell, 76, 969–976.
Takamizawa S, Okamoto S, Wen J, Bishop W, Kimura K and Sandler A . (2000). J. Pediatr. Surg., 35, 375–379.
Tao W and Levine AJ . (1999). Proc. Natl. Acad. Sci. USA, 96, 6937–6941.
Teitz T, Wei T, Valentine MB, Vanin EF, Grenet J, Valentine VA, Behm FG, Look AT, Lahti JM and Kidd VJ . (2000). Nat. Med., 6, 529–535.
Tonini GP and Romani M . (2003). Cancer Lett., 197, 69–73.
Trumpp A, Refaeli Y, Oskarsson T, Gasser S, Murphy M, Martin GR and Bishop JM . (2001). Nature, 414, 768–773.
van Loo G, Schotte P, van Gurp M, Demol H, Hoorelbeke B, Gevaert K, Rodriguez I, Ruiz-Carrillo A, Vandekerckhove J, Declercq W, Beyaert R and Vandenabeele P . (2001). Cell Death Differ., 8, 1136–1142.
Vaux DL and Silke J . (2003). Biochem. Biophys. Res. Commun., 304, 499–504.
Veis DJ, Sorenson CM, Shutter JR and Korsmeyer SJ . (1993). Cell, 75, 229–240.
Wagner AJ, Kokontis JM and Hay N . (1994). Genes Dev., 8, 2817–2830.
Wang HG, Rapp UR and Reed JC . (1996). Cell, 87, 629–638.
Wang X . (2001). Genes Dev., 15, 2922–2933.
Wang Z, Bhattacharya N, Weaver M, Petersen K, Meyer M, Gapter L and Magnuson NS . (2001). J. Vet. Sci., 2, 167–179.
Watanabe-Fukunaga R, Brannan CI, Copeland NG, Jenkins NA and Nagata S . (1992). Nature, 356, 314–317.
Watson JD, Oster SK, Shago M, Khosravi F and Penn LZ . (2002). J. Biol. Chem., 277, 36921–36930.
Weber JD, Jeffers JR, Rehg JE, Randle DH, Lozano G, Roussel MF, Sherr CJ and Zambetti GP . (2000). Genes Dev., 14, 2358–2365.
Weber JD, Taylor LJ, Roussel MF, Sherr CJ and Bar-Sagi D . (1999). Nat. Cell. Biol., 1, 20–26.
Wei MC, Lindsten T, Mootha VK, Weiler S, Gross A, Ashiya M, Thompson CB and Korsmeyer SJ . (2000). Genes Dev., 14, 2060–2071.
Weissinger EM, Eissner G, Grammer C, Fackler S, Haefner B, Yoon LS, Lu KS, Bazarov A, Sedivy JM, Mischak H and Kolch W . (1997). Mol. Cell. Biol., 17, 3229–3241.
Wen R, Wang D, McKay C, Bunting KD, Marine JC, Vanin EF, Zambetti GP, Korsmeyer SJ, Ihle JN and Cleveland JL . (2001). Mol. Cell. Biol., 21, 678–689.
White E . (1996). Genes Dev., 10, 1–15.
Wu GS, Burns TF, McDonald 3rd ER, Jiang W, Meng R, Krantz ID, Kao G, Gan DD, Zhou JY, Muschel R, Hamilton SR, Spinner NB, Markowitz S, Wu G and el-Deiry WS . (1997). Nat. Genet., 17, 141–143.
Wu S, Cetinkaya C, Munoz-Alonso MJ, von der Lehr N, Bahram F, Beuger V, Eilers M, Leon J and Larsson LG . (2003). Oncogene, 22, 351–360.
Wu X, Bayle JH, Olson D and Levine AJ . (1993). Genes Dev., 7, 1126–1132.
Wu X and Levine AJ . (1994). Proc. Natl. Acad. Sci. USA, 91, 3602–3606.
Xiao Q, Claassen G, Shi J, Adachi S, Sedivy J and Hann SR . (1998). Genes Dev., 12, 3803–3808.
Xiong Y, Zhang H and Beach D . (1993). Genes Dev., 7, 1572–1583.
Yeh WC, Pompa JL, McCurrach ME, Shu HB, Elia AJ, Shahinian A, Ng M, Wakeham A, Khoo W, Mitchell K, El-Deiry WS, Lowe SW, Goeddel DV and Mak TW . (1998). Science, 279, 1954–1958.
You Z, Madrid LV, Saims D, Sedivy J and Wang CY . (2002). J. Biol. Chem., 277, 36671–366777.
Yu J, Zhang L, Hwang PM, Kinzler KW and Vogelstein B . (2001). Mol. Cell, 7, 673–682.
Yu Q, He M, Lee NH and Liu ET . (2002). J. Biol. Chem., 277, 13059–13066.
Zambetti GP, Bargonetti J, Walker K, Prives C and Levine AJ . (1992). Genes Dev., 6, 1143–1152.
Zambetti GP and Levine AJ . (1993). FASEB J., 7, 855–865.
Zeller KI, Haggerty TJ, Barrett JF, Guo Q, Wonsey DR and Dang CV . (2001). J. Biol. Chem., 276, 48285–48291.
Zeller KI, Jegga AG, Aronow BJ, O'Donnell KA and Dang CV . (2003). Genome Biol., 4, R69.
Zha J, Harada H, Yang E, Jockel J and Korsmeyer SJ . (1996). Cell, 87, 619–628.
Zha J, Weiler S, Oh KJ, Wei MC and Korsmeyer SJ . (2000). Science, 290, 1761–1765.
Zindy F, Eischen CM, Randle DH, Kamijo T, Cleveland JL, Sherr CJ and Roussel MF . (1998). Genes Dev., 12, 2424–2433.
Zornig M, Grzeschiczek A, Kowalski MB, Hartmann KU and Moroy T . (1995). Oncogene, 10, 2397–2401.
Zou X, Lin Y, Rudchenko S and Calame K . (1997). Curr. Top. Microbiol. Immunol., 224, 57–66.
Acknowledgements
We thank current and past members of the laboratory for their efforts, dedication and insights into Myc, cell suicide and cancer. We are also deeply indebted to the Roussel-Sherr, Zambetti and Ihle laboratories for our exciting and productive collaborations over the past decade. We apologize for any omissions of work from our colleagues relevant to the problem, as tackling Myc and apoptosis certainly ‘takes a village’. It was simply our intent to focus the review on those pathways shown to be most relevant to cancer. Our research is supported by grants from the NIH and by the American Lebanese Syrian Associated Charities (ALSAC). JA Nilsson is a George J. Mitchell Endowed Postdoctoral Fellow of St Jude Children's Research Hospital.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is cited by
-
Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions
Signal Transduction and Targeted Therapy (2023)
-
MicroRNAs as the critical regulators of Cisplatin resistance in ovarian cancer cells
Journal of Ovarian Research (2021)
-
MYC: a multipurpose oncogene with prognostic and therapeutic implications in blood malignancies
Journal of Hematology & Oncology (2021)
-
A bacterial protease depletes c-MYC and increases survival in mouse models of bladder and colon cancer
Nature Biotechnology (2021)
-
Cadmium sulfate induces apoptosis in planarians
Environmental Science and Pollution Research (2020)