Abstract
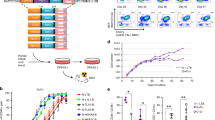
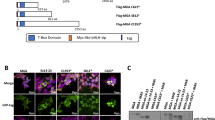
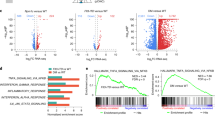
Molecular evidence suggests a multistep process in the development of acute leukemia. Since inappropriate activation of cytokine signaling cascades is a recurring theme in human leukemia, we performed expression screens to identify genes that transform cytokine-dependent cells. Using retroviral cDNA libraries derived from peripheral blood mononuclear cells of patients with myeloproliferative disorders, we isolated numerous genes that genetically complement cytokine requirements for proliferation of BaF/3 and TF-1 cells. The majority of recovered genes represent members of the kinase family, including several previously linked to leukemogenesis. Our unbiased screen highlights the central role of kinase activation in hematopoietic cell proliferation and identifies a number of potential leukemic oncoproteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Abbreviations
- MPDs:
-
myeloproliferative disorders
- FACS:
-
fluorescence-activated cell sorter
- IL-3:
-
interleukin-3
References
Allen JD and Adams JM . (1993). Blood, 81, 3242–3251.
Allen JD, Harris AW, Bath ML, Strasser A, Scollay R and Adams JM . (1995). J. Immunol., 154, 1531–1542.
Allen JD, Lints T, Jenkins NA, Copeland NG, Strasser A, Harvey RP and Adams JM . (1991). Genes Dev., 5, 509–520.
Aoki M, Hamada F, Sugimoto T, Sumida S, Akiyama T and Toyoshima K . (1993). J. Biol. Chem., 268, 22723–22732.
Baker BW, Boettiger D, Spooncer E and Norton JD . (1992). Nucleic Acids Res., 20, 5234.
Baker DA, Glover HR and Dibb NJ . (1994). Leukemia, 8, 141–150.
Beck TW, Huleihel M, Gunnell M, Bonner TI and Rapp UR . (1987). Nucleic Acids Res., 15, 595–609.
Ben-Neriah Y, Daley GQ, Mes-Masson AM, Witte ON and Baltimore D . (1986). Science, 233, 212–214.
Boudard D, Vasselon C, Bertheas MF, Jaubert J, Mounier C, Reynaud J, Viallet A, Chautard S, Guyotat D and Campos L . (2002). Am. J. Hematol., 70, 115–125.
Bourette RP, Von Ruden T, Ballmer-Hofer K, Morle F, Blanchet JP and Mouchiroud G . (1992). Growth Factors, 7, 315–325.
Carroll M, Tomasson MH, Barker GF, Golub TR and Gilliland DG . (1996). Proc. Natl. Acad. Sci. USA, 93, 14845–14850.
Chan AM, Chedid M, McGovern ES, Popescu NC, Miki T and Aaronson SA . (1993). Oncogene, 8, 1329–1333.
Cillo C, Faiella A, Cantile M and Boncinelli E . (1999). Exp. Cell. Res., 248, 1–9.
Crompton MR, Bartlett TJ, MacGregor AD, Manfioletti G, Buratti E, Giancotti V and Goodwin GH . (1992). Nucleic Acids Res., 20, 5661–5667.
Daley GQ and Baltimore D . (1988). Proc. Natl. Acad. Sci. USA, 85, 9312–9316.
Daley GQ, Van Etten RA and Baltimore D . (1990). Science, 247, 824–830.
Dash AB, Williams IR, Kutok JL, Tomasson MH, Anastasiadou E, Lindahl K, Li S, Van Etten RA, Borrow J, Housman D, Druker B and Gilliland DG . (2002). Proc. Natl. Acad. Sci. USA, 99, 7622–7627.
Deguchi K and Gilliland DG . (2002). Leukemia, 16, 740–744.
Deguchi Y, Thevenin C and Kehrl JH . (1992). J. Biol. Chem., 267, 8222–8229.
DuBridge RB, Tang P, Hsia HC, Leong PM, Miller JH and Calos MP . (1987). Mol. Cell. Biol., 7, 379–387.
Golub TR, Barker GF, Lovett M and Gilliland DG . (1994). Cell, 77, 307–316.
Golub TR, Goga A, Barker GF, Afar DE, McLaughlin J, Bohlander SK, Rowley JD, Witte ON and Gilliland DG . (1996). Mol. Cell. Biol., 16, 4107–4116.
Greenberger JS, Sakakeeny MA, Humphries RK, Eaves CJ and Eckner RJ . (1983). Proc. Natl. Acad. Sci. USA, 80, 2931–2935.
Groffen J, Stephenson JR, Heisterkamp N, de Klein A, Bartram CR and Grosveld G . (1984). Cell, 36, 93–99.
Hampe A, Laprevotte I, Galibert F, Fedele LA and Sherr CJ . (1982). Cell, 30, 775–785.
Han J, Wang X, Jiang Y, Ulevitch RJ and Lin S . (1997). FEBS Lett., 403, 19–22.
Hanada M, Kobayashi T, Ohnishi M, Ikeda S, Wang H, Katsura K, Yanagawa Y, Hiraga A, Kanamaru R and Tamura S . (1998). FEBS Lett., 437, 172–176.
Hansen GM and Justice MJ . (1999). Oncogene, 18, 6531–6539.
Hariharan IK, Adams JM and Cory S . (1988). Oncogene Res., 3, 387–399.
Ho JM, Beattie BK, Squire JA, Frank DA and Barber DL . (1999). Blood, 93, 4354–4364.
Kato JY, Roussel MF, Ashmun RA and Sherr CJ . (1989). Mol. Cell. Biol., 9, 4069–4073.
Kitamura T, Tange T, Terasawa T, Chiba S, Kuwaki T, Miyagawa K, Piao YF, Miyazono K, Urabe A and Takaku F . (1989). J. Cell Physiol., 140, 323–334.
Klucher KM, Lopez DV and Daley GQ . (1998). Blood, 91, 3927–3934.
Koh EY, Chen T and Daley GQ . (2002). Nucleic Acids Res., 30, e142.
Koh EY and Daley GQ . (2001). Chronic Myeloid Leukaemia: Biology and Treament, Carella AM, Daley GQ, Eaves CJ, Goldman JM and Hehlmann R (eds). Martin Dunitz: London, pp. 56–72.
Kulkarni S, Heath C, Parker S, Chase A, Iqbal S, Pocock CF, Kaeda J, Cwynarski K, Goldman JM and Cross NC . (2000). Cancer Res., 60, 3592–3598.
Kyba M, Perlingeiro RC and Daley GQ . (2002). Cell, 109, 29–37.
Lewis TS, Shapiro PS and Ahn NG . (1998). Adv. Cancer Res., 74, 49–139.
Li S, Ilaria RL, Jr, Million RP, Daley GQ and Van Etten RA . (1999). J. Exp. Med., 189, 1399–1412.
Magli MC, Largman C and Lawrence HJ . (1997). J. Cell. Physiol., 173, 168–177.
Manfioletti G, Gattei V, Buratti E, Rustighi A, De Iuliis A, Aldinucci D, Goodwin GH and Pinto A . (1995). Blood, 85, 1237–1245.
Maniatis T, Fritsch EF and Sambrook J . (1989). Molecular Cloning: A Laboratory Manual, 2nd edn. Cold Spring Harbor Laboratory: Cold Spring Harbor, NY.
McArthur GA, Rohrschneider LR and Johnson GR . (1994). Blood, 83, 972–981.
McCubrey JA, Steelman LS, Hoyle PE, Blalock WL, Weinstein-Oppenheimer C, Franklin RA, Cherwinski H, Bosch E and McMahon M . (1998). Leukemia, 12, 1903–1929.
Miyoshi J, Higashi T, Mukai H, Ohuchi T and Kakunaga T . (1991). Mol. Cell. Biol., 11, 4088–4096.
Naviaux RK, Costanzi E, Haas M and Verma IM . (1996). J. Virol., 70, 5701–5705.
Nelson KL, Rogers JA, Bowman TL, Jove R and Smithgall TE . (1998). J. Biol. Chem., 273, 7072–7077.
Onishi M, Nosaka T, Misawa K, Mui AL, Gorman D, McMahon M, Miyajima A and Kitamura T . (1998). Mol. Cell. Biol., 18, 3871–3879.
Palacios R and Steinmetz M . (1985). Cell, 41, 727–734.
Papadopoulos P, Ridge SA, Boucher CA, Stocking C and Wiedemann LM . (1995). Cancer Res., 55, 34–38.
Peters DG, Klucher KM, Perlingeiro RC, Dessain SK, Koh EY and Daley GQ . (2001). Oncogene, 20, 2636–2646.
Reiter A, Sohal J, Kulkarni S, Chase A, Macdonald DH, Aguiar RC, Goncalves C, Hernandez JM, Jennings BA, Goldman JM and Cross NC . (1998). Blood, 92, 1735–1742.
Sauvageau G, Lansdorp PM, Eaves CJ, Hogge DE, Dragowska WH, Reid DS, Largman C, Lawrence HJ and Humphries RK . (1994). Proc. Natl. Acad. Sci. USA, 91, 12223–12227.
Shimoda K, Kato K, Aoki K, Matsuda T, Miyamoto A, Shibamori M, Yamashita M, Numata A, Takase K, Kobayashi S, Shibata S, Asano Y, Gondo H, Sekiguchi K, Nakayama K, Nakayama T, Okamura T, Okamura S and Niho Y . (2000). Immunity, 13, 561–571.
Spiekermann K, Biethahn S, Wilde S, Hiddemann W and Alves F . (2001). Eur. J. Haematol., 67, 63–71.
Starr R, Willson TA, Viney EM, Murray LJ, Rayner JR, Jenkins BJ, Gonda TJ, Alexander WS, Metcalf D, Nicola NA and Hilton DJ . (1997). Nature, 387, 917–921.
Tabrizi M, Yang W, Jiao H, DeVries EM, Platanias LC, Arico M and Yi T . (1998). Leukemia, 12, 200–206.
Tobal K, Pagliuca A, Bhatt B, Bailey N, Layton DM and Mufti GJ . (1990). Leukemia, 4, 486–489.
Trus MD, Sodroski JG and Haseltine WA . (1982). J. Biol. Chem., 257, 2730–2733.
Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M, Nakamura Y, White R, Smits AM and Bos JL . (1988). N. Engl. J. Med., 319, 525–532.
Yates KE and Gasson JC . (1996). Stem Cells, 14, 117–123.
Acknowledgements
We acknowledge the efforts of Phil Waite and the members of the Blood Donor Center at the Massachusetts General Hospital in providing patient samples. This work was supported by grants from the NIH (CA76418 and CA86991). GQD is the Birnbaum Scholar of the Leukemia and Lymphoma Society of America. EYK was supported by NIH training Grants (GM07753-22 and CA09541).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koh, E., Chen, T. & Daley, G. Genetic complementation of cytokine signaling identifies central role of kinases in hematopoietic cell proliferation. Oncogene 23, 1214–1220 (2004). https://doi.org/10.1038/sj.onc.1207209
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207209
Keywords
This article is cited by
-
Activation of tyrosine kinases by mutation of the gatekeeper threonine
Nature Structural & Molecular Biology (2008)