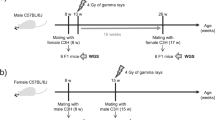
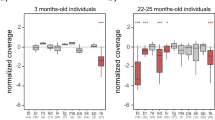
Abstract
Many studies confirmed that radiation induces genomic instability in whole-body systems. However, the results of the studies are not always consistent with each other. Attempts are made in the present review to resolve the discrepancies. Many of the studies in human and experimental animals utilize the length change mutation of minisatellite sequences as a marker of genomic instability. Minisatellite sequences frequently change their length, and the data obtained by conventional Southern blotting give rather qualitative information, which is sometimes difficult to scrutinize quantitatively. This is the problem inevitably associated with the study of minisatellite mutations and the source of some conflicts among studies in humans and mice. Radiation induction of genomic instability has also been assessed in whole-body experimental systems, using other markers such as the mouse pink-eyed unstable allele and the specific pigmentation loci of medaka fish (Oryzias latipes). Even though there are some contradictions, all these studies have demonstrated that genomic instability is induced in the germ cells of irradiated parents, especially of males, and in offspring born to them. Among these, transmission of genomic instability to the second generation of irradiated parents is limited to the mouse minisatellite system, and awaits further clarification in other experimental systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Adler I . (1996). Mutat. Res., 352, 169–172.
Auerbach C . (1976). Mutation Research. Chapman & Hall: London, pp. 270–278.
Barber R, Plumb M, Smith AG, Cesar CE, Boulton E, Jeffreys AJ and Dubrova YE . (2000). Mutat. Res., 457, 79–91.
Barber R, Plumb MA, Boulton E, Roux I and Dubrova YE . (2002). Proc. Natl. Acad. Sci. USA, 99, 6877–6882.
Biological Effects of Ionizing Radiation Report V (BEIRR V). (1990). National Academy of Science. National Research Council, Washington DC, USA.
Bishop AJ, Kosaras B, Sidman RL and Schiestl RH . (2000). Mutat. Res., 457, 31–40.
Boudsocq F, Ling H, Yang W and Woodgate R . (2002). DNA Repair (Amst.), 1, 343–358.
Bridges B . (2001). Radiat. Res., 156, 631–641.
Brilliant MH, Williams RW, Conti CJ, Angel JM, Oakey RJ and Holdner BC . (1994). Mamm. Genome, 5, S104–S123.
Buard J, Bourdet A, Yardley J, Dubrova Y and Jeffreys AJ . (1998). EMBO J., 17, 3495–3502.
Buard J, Collick A, Brown J and Jeffreys AJ . (2000). Genomics, 65, 95–103.
Cho C, Jung-Ha H, Willis WD, Goulding EH, Stein P, Xu Z, Schultz RM, Hecht NB and Eddy EM . (2003). Biol. Reprod. (e-publication ahead of print).
Cox M . (2000). Proc. Natl. Acad. Sci. USA, 98l, 8173–8180.
Crompton NEA, Barth B and Kiefer J . (1990). Radiat. Res., 124, 300–308.
Dubrova YE, Bersimbaev RI, Djansugurova LB, Tankimanova MK, Mamyrbaeva ZZ, Mustonen R, Lindholm C, Hulten M and Salomaa S . (2002b). Science, 295, 103.
Dubrova YE, Grant G, Chumak AA, Stezhka VA and Karakasian AN . (2002b). Am J Hum Genet., 71, 801–809.
Dubrova YE, Jeffreys AJ and Malashenko AM . (1993). Nature Genet., 5, 92–94.
Dubrova YE, Plumb M, Brown J, Boulton E, Goodhead D and Jeffreys AJ . (2000). Mutat. Res., 453, 17–24.
Dubrova YE, Plumb M, Brown J, Fennelly J, Bois P, Goodhead D and Jeffreys AJ . (1998a). Proc. Natl. Acad. Sci. USA, 95, 6251–6255.
Dubrova YE, Plumb M, Brown J and Jeffreys AJ . (1998b). Int. J. Radiat. Biol., 74, 689–696.
Dubrova YE, Nesterov VN, Krouchinsky NG, Ostapenko VA, Neumann R, Neil DL and Jeffreys AJ . (1996). Nature, 380, 683–686.
Dubrova YE, Nesterov VN, Krouchinsky NG, Ostapenko VA, Vergnaud G, Giraudeau F, Buard J and Jeffreys AJ . (1997). Mutat. Res., 381, 267–278.
Fan YJ, Wang Z, Sadamoto S, Ninomiya Y, Kotomura N, Kamiya K, Dohi K, Kominami R and Niwa O . (1995). Int. J. Radiat. Biol., 68, 177–183.
Friedberg EC, Walker GC and Siede W . (1995). DNA Repair and Mugatenesis. ASM Press: Washington, DC, pp. 59–61.
Gibbs M, Collick A, Kelly RG and Jeffreys A . (1993). Genomics, 17, 121–128.
Gondo Y, Gardner JM, Nakatsu Y, Durham-Pierre D, Deveau SA, Kuper C and Brilliant MH . (1993). Proc. Natl. Acad. Sci. USA, 90, 297–301.
Goodhead DT . (1999). J. Radiat. Res., 40 (Suppl.), 1–13.
Haber JE . (2000). Trends Genet., 16, 259–264.
Hill CK, Han A and Elkind MM . (1984). Int. J. Radiat. Biol., 46, 11–15.
Jeffereys AJ . (1997). Clin. Sci., 93, 383–390.
Jeffereys AJ and Neumann R . (1997). Hum. Mol. Genet., 136–145.
Jeffereys AJ, Tamaki K, MacLeod A, Monkton DG, Neil DL and Armour JA . (1994). Nature Genet., 6, 136–145.
Jeffereys AJ, Wilson V and Thein SL . (1985). Nature, 314, 67–74.
Kelly R, Bulfield G, Collick A, Gibbs M and Jeffreys A . (1989). Genomics, 5, 844–856.
Kitajima TS, Yokobayashi S, Yamamoto M and Watanabe Y . (2003). Science, 300, 1152–1155.
Kiuru A, Auvinen A, Luokkamaki M, Kakkonen K, Veidebaum T, tekkel M, Rahu M, Kakulinen T, Servomaa K, Rytomaa T and Mustonen R . (2003). Radiat. Res., 159, 651–655.
Kodaira M, Satoh C, Hiyama K and Toyama K . (1995). Am. J. Hum. Genet., 57, 1275–1283.
Leroy C, Lee SE, Vaze MB, Ochsenbien F, Guerois R, Haber JE and Marsolier-Kergoat MC . (2003). Mol Cell, 11, 827–835.
Livshits LA, Malyarchuk SG, Kravchenko SA, Matsuka GH, Lukyanova EM, Antipkin YG, Arabskaya LP, Petit E, Giraudeau F, Gourmelon P, Vergnaud G and Le Guen B . (2001). Radiat. Res., 155, 74–80.
Mannazzu I, Simonetti E, Marinangeli P, Guerra E, Budroni M, Thangavelu M and Clementi F . (2002). Appl. Environ. Microbiol., 68, 5437–5444.
Martini E and Keeney S . (2002). Mol. Cell, 9, 700–702.
May CA, Tamaki K, Neumann R, Wilson G, Zagars G, Pollack A, Dubrovva YE, Jeffreys AJ and Meistrich ML . (2000). Mutat. Res., 453, 67–75.
Mitani K, Takahashi Y and Kominami R . (1990). J. Biol. Chem., 265, 15203–15210.
Morgan WF . (2003a). Radiat Res., 159, 567–580.
Morgan WF . (2003b). Radiat Res., 159, 581–596.
Neel JV, Schull WJ, Awa AA, Satoh C, Kato H, Otake M and Yoshimoto Y . (1990). Am. J. Hum. Genet., 46, 1053–1072.
Nikjoo H, Munson RJ and Bridges BA . (1999). J. Radiat. Res., 40 (Suppl.), 85–105.
Niwa O, Fan Y, Numoto M, Kamiya K and Kominami R . (1996). J. Radiat. Res., 37, 217–224.
Niwa O and Kominami R . (2001). Proc.Natl. Acad. Sci. USA, 98, 1705–1710.
Radman M . (2001). Nature, 413, 115.
Rassoulzadegan M, Magliano M and Cuzin F . (2002). EMBO J., 21, 440–450.
Richard G-F and Paques F . (2000). EMBO Rep., 1, 122–126.
Russell WL, Bangham JW and Russell LB . (1998). Genetics, 148, 1567–1578.
Russell LB, Hunsicker PR, Nestor LA, Cacheiro LA and Generoso WM . (1991). Mutat. Res., 262, 101–107.
Russell WL and Kelly EM . (1982). Proc. Natl. Acad. Sci. USA, 79, 542–544.
Russell LB, Russell WL, Rinchik EM and Hunsicker PR . (1990). Bumbury Report 34, Cold Spring Harbor Laboratory. Cold Spring Harbor, NY, pp 271–289.
Sadamoto S, Suzuki S, Kamiya K, Kominami R, Dohi K and Niwa O . (1994). Int. J. Radiat. Biol., 65, 549–557.
Sankaranarayanan K and CHakraborty R . (2000). Mutat. Res., 453, 107–127.
Satoh C and Kodaira M . (1996). Nature, 383, 226.
Schiestl RH, Aubrecht J, Khogali F and Carls N . (1997). Proc. Natl. Acad. Sci. USA, 94, 4576–4581.
Schiestl RH, Khogali F and Carls N . (1994). Science, 266, 1573–1576.
Schwacha A and Kleickner N . (1997). Cell, 90, 1123–1135.
Shimada A and Shima A . (1991). Proc. Natl. Acad. Sci. USA, 88, 2545–2549.
Shimada A and Shima A . (1998). Mutat. Res., 399, 149–165.
Shimada A and Shima A . (2001). Mutat. Res., 495, 33–42.
Shimura T, Inoue M, Taga M, Shiraishi K, Uematsu N, Takei N, Yuan Z, Shinohara T and Niwa O . (2002). Mol. Cell. Biol., 22, 2220–2228.
Shiraishi K, Shimura T, Taga M, Uematsu N, Gondo Y, Ohtaki M, Kominami R and Niwa O . (2002). Radiat. Res., 157, 661–667.
United Nations Scientific Committee on the Effect of Atomic Radiation. (1993). United Nations, New York.
United Nations Scientific Committee on the Effect of Atomic Radiation. (2000). United Nations, New York.
Wyman AR and White R . (1980). Proc. Natl. Acad. Sci. USA, 77, 6754–6758.
Yauk CL, Dubrova YE, Grant GR and Jeffreys AJ . (2002). Mutat. Res., 500, 147–156.
Yokomori K . (2003). Curr. Top. Microbiol. Immunol., 274, 79–112.
Zheng N, Moncton DG, Wilson G, Hagemeister F, Chakraborty R, Conor TH, Siciliano MJ and Meistrich ML . (2000). Environ. Mol. Mutagen., 36, 134–145.
Acknowledgements
This work is supported by a Grant-in-aid from the Ministry of Education, Culture, Sports, Science and Technology of Japan, and by a grant from the Nuclear Safety Research Association.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niwa, O. Induced genomic instability in irradiated germ cells and in the offspring; reconciling discrepancies among the human and animal studies. Oncogene 22, 7078–7086 (2003). https://doi.org/10.1038/sj.onc.1207037
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207037
Keywords
This article is cited by
-
Nuclear DNA fragmentation negatively affects zona binding competence of Y bearing mouse spermatozoa
Journal of Assisted Reproduction and Genetics (2013)
-
New germline mutations in the hypervariable minisatellite CEB1 in the parents of children with leukaemia
British Journal of Cancer (2007)