Abstract
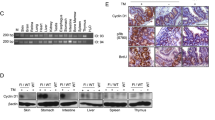
Cyclin D1 is frequently overexpressed in human neoplasias by gene rearrangement and amplification, but no mutations in the CCND1 gene have so far been reported. However, in vitro mutagenesis of CCND1 has shown that substitutions affecting threonine 286 residue produced cyclin D1 nuclear accumulation, by interfering with protein degradation and induced neoplastic transformation in murine fibroblasts. To test whether similar genetic changes may occur in vivo, we analysed a series of 60 endometrioid endometrial carcinomas (EECs) for cyclin D1 expression and gene amplification by immunohistochemistry and FISH, respectively. Two of 17 carcinomas showing cyclin D1 expression in more than 5% of neoplastic cells, but without gene amplification, were found to harbor single-base substitutions in CCND1 that changed proline 287 into threonine and serine, respectively. Both cases expressed cyclin D1 in more than 50% of neoplastic cells. Additionally, seven tumors with cyclin D1 overexpression of an independent series of 59 EECs were also analysed, and a 12-bp in-frame deletion that eliminated amino acids 289–292 was detected in one case with cylin D1 expression in more than 50% of neoplastic cells. In contrast, no mutations of the CCND1 gene were detected in a set of breast carcinomas with cyclin D1 overexpression without gene amplification. In summary, our data indicate that mutations of CCND1, which probably render the protein insensitive to degradation, represent a previously unreported mechanism of cyclin D1 overexpression in human tumors in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Alt JR, Cleveland JL, Hannink M and Diehl JA . (2000). Genes Dev., 24, 3102–3114.
Campo E, Raffeld M and Jaffe ES . (1999). Semin. Hemathol., 36, 115–127.
Cao QJ, Einstein MH, Anderson PS, Runowicz CD, Balan R and Jones JG . (2002). Int. J. Gynecol. Pathol., 21, 147–154.
Diehl JA, Cheng M, Roussel MF and Sherr CJ . (1998). Genes Dev., 12, 3499–3511.
Diehl JA, Zindy F and Sherr CJ . (1997). Genes Dev., 11, 957–972.
Germain D, Russell A, Thompsonm A and Hendley J . (2000). J. Biol. Chem., 275, 12074–12079.
Hoechtlen-Vollmar W, Menzelm G, Bartl R, Lamerz R, Wick M and Seidel D . (2000). Br. J. Haematol., 109, 30–38.
Kaltz-Wittmer C, Klenk U, Glaessgen A, Aust DE, Diebold J, Lohrs U and Baretton GB . (2000). Lab. Invest., 80, 1455–1464.
Machin P, Catasus L, Pons C, Munozn J, Matias-Guiu X and Prat J . (2002). Hum. Pathol., 33, 206–212.
Moreno-Bueno G, Hardisson D, Sánchez C, Sarrió D, Cassia R, García-Rostán G and Palacios J . (2002). Oncogene, 21, 7981–7991.
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B and Kinzler KW . (1997). Science, 275, 1787–1790.
Nikaido T, Li SF, Shiozawa T and Fujii S . (1996). Cancer, 78, 1248–1253.
Quddus M, Latkovich P, Castellani WJ, James Sung C Steinhoff MM, Briggs RC and Miranda RN . (2002). Arch. Pathol. Lab. Med., 126, 459–463.
Russell A, Thompson MA, Hendley J, Trute L, Armes J and Germain D . (1999). Oncogene, 18, 1983–1991.
Schmitz MJ, Hendricks DT, Farley J, Taylor RR, Geradts J, Rose GS and Birrer MJ . (2000). Gynecol. Oncol., 77, 439–445.
Schraml P, Kononenm J, Bubendorf L, Moch H, Bissig H, Nocito A, Mihatsch MJ, Kallioniemi OP and Sauter G . (1999). Clin. Cancer Res., 5, 1966–1975.
Sherr CJ . (1996). Science, 274, 1672–1677 (review).
Sherr CJ and Roberts JM . (1999). Genes Dev., 13, 501–512 (review).
Simpson JF, Quan DE, O’Malley F, Odom-Maryon T and Clarke PE . (1997). Am. J. Pathol., 151, 161–168.
Soslow RA, Shen PU, Chung MH, Isacson C and Baergen RN . (2000). Int. J. Gynecol. Pathol., 19, 329–334.
Steeg PS and Zhou Q . (1998). Breast Cancer Res. Treat., 52, 17–28 (review).
Tsuda H, Yamamoto K, Inoue T, Uchiyama I and Umesaki N . (2000). Br. J. Cancer, 82, 675–682.
Weinstat-Saslow D, Merino MJ, Manrow RE, Lawrence JA, Bluth RF, Wittenbel KD, Simpson JF, Page DL and Steeg PS . (1995). Nat. Med., 1, 1257–1260.
Acknowledgements
We are grateful to Raúl Cassia and Alicia Barroso for excellent technical assistance. The study is supported in part by Grants FIS PI020355, FIS 01/1656 and SAF2001-0065. GM-B is the recipient of a postdoctoral research grant for de CNIO Spain. SR-P is a recipient of a research grant from the CNIO, Spain. DS is a recipient of a research grant from FIS (BEFI, 1/9132).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moreno-Bueno, G., Rodríguez-Perales, S., Sánchez-Estévez, C. et al. Cyclin D1 gene (CCND1) mutations in endometrial cancer. Oncogene 22, 6115–6118 (2003). https://doi.org/10.1038/sj.onc.1206868
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206868
Keywords
This article is cited by
-
Aberrant Cyclin D1 splicing in cancer: from molecular mechanism to therapeutic modulation
Cell Death & Disease (2023)
-
Targeting CDK4 and CDK6 in cancer
Nature Reviews Cancer (2022)
-
Single-cell analysis of gastric pre-cancerous and cancer lesions reveals cell lineage diversity and intratumoral heterogeneity
npj Precision Oncology (2022)
-
Calcineurin regulates cyclin D1 stability through dephosphorylation at T286
Scientific Reports (2019)
-
Gankyrin facilitates follicle-stimulating hormone-driven ovarian cancer cell proliferation through the PI3K/AKT/HIF-1α/cyclin D1 pathway
Oncogene (2016)