Abstract
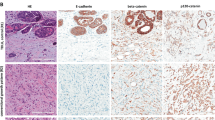
Claudins are transmembrane proteins that seal tight junctions, and are critical for maintaining cell-to-cell adhesion in epithelial cell sheets. However, their role in cancer progression remains largely unexplored. Here, we report that Claudin-7 (CLDN-7) expression is lower in invasive ductal carcinomas (IDC) of the breast than in normal breast epithelium, as determined by both RT–PCR (9/10) and Western analysis (6/8). Immunohistochemical (IHC) analysis of ductal carcinoma in situ (DCIS) and IDC showed that the loss of CLDN-7 expression correlated with histological grade in both DCIS (P<0.001, n=38) and IDC (P=0.014, n=31), occurring predominantly in high-grade (Nuclear and Elston grade 3) lesions. Tissue array analysis of 355 IDC cases further confirmed the inverse correlation between CLDN-7 expression and histological grade (P=0.03). This pattern of expression is consistent with the biological function of CLDN-7, as greater discohesion is typically observed in high-grade lesions. In line with this observation, by IHC analysis, CLDN-7 expression was lost in the vast majority (13/17) of cases of lobular carcinoma in situ, which is defined by cellular discohesion. In fact, inducing disassociation of MCF-7 and T47D cells in culture by treating with HGF/scatter factor resulted in a loss of CLDN-7 expression within 24 h. Silencing of CLDN-7 expression correlated with promoter hypermethylation as determined by methylation-specific PCR (MSP) and nucleotide sequencing in breast cancer cell lines (3/3), but not in IDCs (0/5). In summary, these studies provide insight into the potential role of CLDN-7 in the progression and ability of breast cancer cells to disseminate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Abbreviations
- CLDN:
-
Claudin
- IDC:
-
invasive ductal carcinoma
- RT–PCR:
-
reverse transcription–PCR
- IHC:
-
immunohistochemical analysis
- DCIS:
-
ductal carcinoma in situ
- LCIS:
-
lobular carcinoma in situ
- HGF/scatter factor:
-
hepatocyte growth factor/scatter factor
- KLH:
-
keyhole limpet hemocyanin
- MSP:
-
methylation-specific PCR
- DAB:
-
3 3′-diaminobenzamidine
- GAPDH:
-
glyceraldehyde phosphate dehydrogenase
- 5-aza-dC:
-
5′-aza-2′-deoxycytidine
- HMEC:
-
human mammary epithelial cells
References
Balda MS and Matter K . (2000). EMBO J., 19, 2024–2033.
Bergstraessar LM and Weitzman SA . (1993). Cancer Res., 53, 2644–2654.
Evron E, Dooley WC, Umbricht CB, Rosenthal D, Sacchi N, Gabrielson E, Soito AB, Hung DT, Ljung B, Davidson NE and Sukumar S . (2001a). Lancet, 357, 1335–1336.
Evron E, Umbricht CB, Korz D, Raman V, Loeb DM, Niranjan B, Buluwela L, Weitzman SA, Marks J and Sukumar S . (2001b). Cancer Res., 61, 2782–2787.
Ferguson AT, Evron E, Umbricht CB, Pandita TK, Chan TA, Hermeking H, Marks JR, Lambers AR, Futreal PA, Stampfer MR and Sukumar S . (2000). Proc. Natl. Acad. Sci. USA, 97, 6049–6054.
Fujii T, Dracheva T, Player A, Chacko S, Clifford R, Strausberg RL, Buetow K, Azumi N, Travis WD and Jen J . (2002). Cancer Res., 62, 3340–3346.
Furuse M, Fujita K, Hiiragi T, Fujimoto K and Tsukita S . (1998). J. Cell Biol., 141, 1539–1550.
Furuse M, Hata M, Furuse K, Yoshida Y, Haratake A, Sugitani Y, Noda T, Kubo A and Tsukita S . (2002). J. Cell Biol., 156, 1099–1111.
Gomm JJ, Browne PJ, Coope RC, Liu QY, Buluwela L and Coombes RC . (1995). Anal. Biochem., 226, 91–99.
Iacobuzio-Donahue CA, Maitra A, Shen-Ong GL, van Heek T, Ashfaq R, Meyer R, Walter K, Berg K, Hollingsworth MA, Cameron JL, Yeo CJ, Kern SE, Goggins M and Hruban RH . (2002). Am. J. Pathol., 160, 1239–1249.
Itoh M, Furuse M, Morita K, Kubota K, Saitou M and Tsukita S . (1999). J. Cell Biol., 147, 1351–1363.
Jiang W, Hiscox S, Matsumoto K and Nakamura T . (1999). Crit. Rev. Oncology Hematology, 29, 209–248.
Jones PA and Baylin SB . (2002). Nat Rev Genet., 3, 415–428.
Kramer F, White K, Kubbies M, Swisshelm K and Weber BHF . (2000). Hum. Genet., 107, 249–256.
Loeb DM, Evron E, Patel CB, Sharma PM, Niranjan B, Buluwela L, Weitzman SA, Korz D and Sukumar S . (2001). Cancer Res., 61, 921–925.
Mitic LL and Anderson JM . (1998). Annu. Rev. Physiol., 60, 121–142.
Mitic LL, Van Itallie CM and Anderson JM . (2000). Am. J. Physiol. Gastrointest. Liver Physiol., 279, G250–G254.
Morita K, Furuse M, Fujimoto K and Tsukita S . (1999). Proc. Natl. Acad. Sci. USA, 96, 511–516.
Nacht M, Ferguson AT, Zhang W, Petroziello JM, Cook BP, Gao YH, Maguire S, Riley D, Coppola G, Landes GM, Madden SL and Sukumar S . (1999). Cancer Res., 59, 5464–5470.
Simon DB, Lu Y, Choate KA, Velazques H, Al-Sabban E, Praga M, Casari G, Bettinelli A, Colussi G, Rodriguez-Soriano J, McCredie D, Milford D, Sanjad S and Lifton RP . (1999). Science, 285, 103–106.
Sirotkin H, Morrow B, Saint-Jore B, Puech A, Das Gupta R, Patanjali SR, Skoultchi A, Weissman SM and Kucherlapati R . (1997). Genomics, 42, 245–251.
Soler AP, Knudsen KA, Jaurand M-C, Johnson KR, Wheelock MJ, Klein-Szanto AJP and Salazar H . (1995). Hum. Pathol., 26, 1363–1369.
Tsukita S and Furuse M . (1999). Trends Cell Biol., 9, 268–273.
Wheelock MJ, Soler AP and Knudsen KA . (2001). J. Mammary Gland Biol. Neoplasia, 6, 275–285.
Wilcox ER, Burton QL, Naz S, Riazuddin S, Smith TN, Ploplis B, Belyantseva I, Ben-Yosef T, Liburd NA, Morell RJ, Kachar B, Wu DK, Griffith AJ, Riazuddin S and Friedman TB . (2001). Cell, 104, 165–172.
Acknowledgements
We gratefully acknowledge Leslie Meszler at the Cell Imaging Core Facility, Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, for expert assistance with confocal microscopy. We thank Dr Vimla Band, Dr Sigmund A Weitzman, Dr Martha Stampfer, Dr Steven Ethier, Dr Birunthi Niranjan, and Dr Lakjaya Buluwela for their generous gift of breast cells of normal and cancer origin. We are also thankful to the members of the Sukumar laboratory for useful advice and discussions. This work was supported by PHHS Grants SPORE P50 CA88843 (to S Sukumar), and DAMD17-01-1-0285 (to S Sukumar) and BC010495 (to SL Kominsky) from the US Army Medical Research and Materiel Command.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kominsky, S., Argani, P., Korz, D. et al. Loss of the tight junction protein claudin-7 correlates with histological grade in both ductal carcinoma in situ and invasive ductal carcinoma of the breast. Oncogene 22, 2021–2033 (2003). https://doi.org/10.1038/sj.onc.1206199
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206199
Keywords
This article is cited by
-
Truncated O-glycosylation in metastatic triple-negative breast cancer reveals a gene expression signature associated with extracellular matrix and proteolysis
Scientific Reports (2024)
-
Ubiquitin-specific protease 28: the decipherment of its dual roles in cancer development
Experimental Hematology & Oncology (2023)
-
Claudin 1, 4 and 7 Expression in Malignant Melanoma
Indian Journal of Surgery (2023)
-
Cooperative interaction between ERα and the EMT-inducer ZEB1 reprograms breast cancer cells for bone metastasis
Nature Communications (2022)
-
The F11 Receptor (F11R)/Junctional Adhesion Molecule-A (JAM-A) (F11R/JAM-A) in cancer progression
Molecular and Cellular Biochemistry (2022)