Abstract
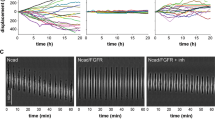
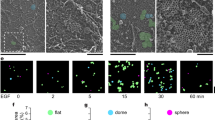
Substrate-bound FGF2 promotes endothelial cell adhesion by interacting with αvβ3 integrin. Here, endothelial GM7373 cells spread and organize focal adhesion plaques on immobilized FGF2, fibronectin (FN), and vitronectin (VN). αvβ3 integrin, paxillin, focal adhesion kinase, vinculin and pp60src localize in cell-substratum contact sites on FGF2, FN or VN. However, only immobilized FGF2 induces a long-lasting activation of extracellular signal-regulated kinases1/2 (ERK1/2) and cell proliferation that was inhibited by the ERK1/2 inhibitor PD 098059 and the tyrosine kinase (TK) inhibitor tyrphostin 23, pointing to the engagement of FGF receptor (FGFR) at the basal side of the cell. To assess this hypothesis, GM7373 cells were transfected with a dominant negative TK−-ΔFGFR1 mutant (GM7373-ΔFGFR1 cells) or with the full-length receptor (GM7373-FGFR1 cells). Both transfectants adhere and spread on FGF2 but GM7373-ΔFGFR1 cells do not proliferate. Also, parental and GM7373-FGFR1 cells, but not GM7373-ΔFGFR1 cells, undergo morphological changes and increased motility on FGF2-coated plastic. Finally, FGFR1, but not TK−-ΔFGFR1, localizes in cell adhesion contacts on immobilized FGF2. In conclusion, substrate-bound FGF2 induces endothelial cell proliferation, motility, and the recruitment of FGFR1 in cell-substratum contacts. This may contribute to the cross talk among intracellular signaling pathways activated by FGFR1 and αvβ3 integrin in endothelial cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Albelda SM, Buck CA . 1990 FASEB J. 4: 2868–2880
Alessi DR, Cuenda A, Cohen P, Dudley DT, Saltiel AR . 1995 J. Biol. Chem. 270: 27489–27494
Baird A, Schubert D, Ling N, Guillemin R . 1988 Proc. Natl. Acad. Sci. USA 85: 2324–2328
Basilico C, Moscatelli D . 1992 Adv. Cancer Res. 59: 115–165
Besser D, Presta M, Nagamine Y . 1995 Cell Growth Differ. 6: 1009–1017
Boyer B, Thiery JP . 1993 J. Cell Biol. 120: 767–776
Broadley KN, Aquino AM, Woodward SC, Buckley-Sturrock A, Sato Y, Rifkin DB, Davidson JM . 1989 Lab. Invest. 61: 571–575
Brooks PC, Richard AF, Cheresh DA . 1994 Science 264: 569–571
Carmeliet P . 2000 Nature Med. 6: 389–395
Carmeliet P, Jain RK . 2000 Nature 407: 249–257
Cheresh DA . 1987 Proc. Natl. Acad. Sci. USA 84: 6471–6475
Cuenda A, Rouse J, Doza YN, Meier R, Cohen P, Gallagher TF, Young PR, Lee JC . 1995 FEBS Lett. 364: 229–233
Culp LA . 1976 Biochemistry 15: 4094–4104
Davis CM, Danehower SC, Laurenza A, Molony JL . 1993 J. Cell. Biochem. 51: 206–218
Del Rosso M, Pedersen N, Fibbi G, Pucci M, Dini G, Anichini E, Blasi F . 1992 Exp. Cell Res. 203: 427–434
Eliceiri BP, Klemke R, Stromblad S, Cheresh DA . 1998 J. Cell Biol. 140: 1255–1263
Enenstein J, Waleh NS, Kramer RH . 1992 Exp. Cell Res. 203: 499–503
Folkman J, Klagsbrun M, Sasse J, Wadzinsky M, Ingber D, Vlodavsky I . 1988 Am. J. Pathol. 130: 393–400
Friedlander M, Brooks PC, Shaffer RW, Kincaid CM, Varner JA, Cheresh DA . 1995 Science 270: 1500–1502
Gazit A, Yaish P, Gilon C, Levitzki A . 1989 J. Biol. Chem. 264: 14503–14509
Giancotti FG, Rouslathi E . 1999 Science 285: 1028–1032
Giuliani R, Bastaki M, Coltrini D, Presta M . 1999 J. Cell Sci. 112: 2597–2606
Grinspan JB, Stephen NM, Levine EM . 1983 J. Cell Physiol. 114: 328–338
Hynes RO . 1992 Cell 69: 11–25
Ingber DE, Folkman J . 1989a Cell 58: 803–805
Ingber DE, Folkman J . 1989b J. Cell Biol. 109: 317–330
Isacchi A, Statuto M, Chiesa R, Bergonzoni L, Rusnati M, Sarmientos P, Ragnotti G, Presta M . 1991 Proc. Natl. Acad. Sci. USA 88: 2628–2632
Johnson DE, Williams LT . 1993 Adv. Cancer Res. 60: 1–41
Klein S, Giancotti FG, Presta M, Albelda SM, Clayton AB, Rifkin DB . 1993 Mol. Biol. Cell 4: 973–982
Kumar CC . 1998 Oncogene 17: 1365–1373
Li Y, Basilico C, Mansukhani A . 1994 Mol. Cell Biol. 14: 7600–7669
Miyamoto S, Teramoto H, Coso OA, Gutkind JS, Burbelo PD, Akiyama SK, Yamada KM . 1995 J. Cell Biol. 131: 791–805
Miyamoto S, Teramoto H, Gutkind JS, Yamada KM . 1996 J. Cell Biol. 135: 1633–1642
Moscatelli D, Presta M, Rifkin DB . 1986 Proc. Natl. Acad. Sci. USA 83: 2091–2095
Plopper GE, McNamee HP, Dike LE, Bojanowski K, Ingber DE . 1995 Mol. Biol. Cell 6: 1349–1365
Ribatti D, Urbinati C, Nico B, Rusnati M, Roncali L, Presta M . 1995 Dev. Biol. 170: 39–49
Ribatti D, Leali D, Vacca A, Giuliani R, Gualandris A, Roncali L, Nolli ML, Presta M . 1999 J. Cell Sci. 112: 4213–4221
Richard C, Liuzzo JP, Moscatelli D . 1995 J. Biol. Chem. 270: 24188–24196
Rusnati M, Dell'Era P, Urbinati C, Tanghetti E, Massardi ML, Nagamine Y, Monti E, Presta M . 1996 Mol. Biol. Cell 7: 369–381
Rusnati M, Tanghetti E, Dell'Era P, Gualandris A, Presta M . 1997 Mol. Biol. Cell 8: 2449–2461
Schubert D, Ling N, Baird A . 1987 J. Cell Biol. 104: 635–643
Smith JC, Singh JP, Lillquist JS, Goon DS, Stiles CD . 1982 Nature 269: 154–156
Soldi R, Mitola S, Strasly M, Defilippi P, Tarone G, Bussolino F . 1999 EMBO J. 18: 882–892
Stromblad C, Cheresh DA . 1996 Trends Cell Biol. 6: 462–468
Tsuboi R, Sato Y, Rifkin DB . 1990 J. Cell Biol. 110: 511–517
Vlodavsky I, Folkman J, Sullivan R, Fridman R, Ishai-Michaeli R, Sasse J, Klagsbrun M . 1987 Proc. Natl. Acad. Sci. USA 84: 2292–2296
Zhan X, Plourde S, Hu X, Friesel R, Maciag T . 1994 J. Biol. Chem. 269: 1–4
Acknowledgements
This work was supported by MURST (60% and Cofin 2000 to M Rusnati and to M Presta.; Centro di Eccellenza ‘IDET’ to M Presta), CNR (Progetto Finalizzato Biotecnologie), Associazione Italiana per la Ricerca sul Cancro, Istituto Superiore di Sanità (AIDS Project), and Centro per lo Studio del Trattamento dello Scompenso Cardiaco (University of Brescia) to M Presta
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tanghetti, E., Ria, R., Dell'Era, P. et al. Biological activity of substrate-bound basic fibroblast growth factor (FGF2): recruitment of FGF receptor-1 in endothelial cell adhesion contacts. Oncogene 21, 3889–3897 (2002). https://doi.org/10.1038/sj.onc.1205407
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205407
Keywords
This article is cited by
-
Hypoxia and the phenomenon of immune exclusion
Journal of Translational Medicine (2021)
-
N-tert-butyloxycarbonyl-Phe-Leu-Phe-Leu-Phe (BOC2) inhibits the angiogenic activity of heparin-binding growth factors
Angiogenesis (2018)
-
Initial Contact of Glioblastoma Cells with Existing Normal Brain Endothelial Cells Strengthen the Barrier Function via Fibroblast Growth Factor 2 Secretion: A New In Vitro Blood–Brain Barrier Model
Cellular and Molecular Neurobiology (2013)