Abstract
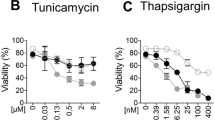
Thapsigargin (TG), by inducing perturbations in cellular Ca2+ homeostasis, has been shown to induce apoptosis. The molecular mechanisms of Ca2+ perturbation-induced apoptosis are not fully understood. In this study, we demonstrate for the first time that TG-mediated perturbations in Ca2+ homeostasis are coupled with activation of the death receptor 5 (DR5)-dependent apoptotic pathway in human cancer cells. TG selectively upregulated DR5 but had no effect on the expression of the other TRAIL receptor, DR4. TG also upregulated the expression of the DR5 ligand TRAIL (tumor necrosis factor-related apoptosis inducing ligand), albeit in a cell-type specific manner. TG-induced apoptosis has been shown to be associated with activation of the mitochondrial pathway. We found that TG upregulation of DR5 and TRAIL was coupled with caspase 8 activation and Bid cleavage, suggesting that the TG-regulated DR5 pathway could be linked to the mitochondrial pathway. TG enhanced not only DR5 mRNA stability but also increased induction of the DR5 genomic promoter-reporter gene. The TG-induced increase in DR5 expression appeared to occur as a consequence of TG-induced endoplasmic reticulum (ER) Ca2+ pool depletion. Thus, we report our novel findings that ER Ca2+ pool depletion-induced apoptotic signals are mediated, at least in part, via a DR5-dependent apoptotic pathway and there appears to be a cross-talk between the death receptor and mitochondrial pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout












Similar content being viewed by others
References
Ashkenazi A, Dixit VM . 1999 Curr. Opin. Cell Biol. 11: 255–260
Chen CY, Shyu AB . 1995 Trends Biochem. Sci. 20: 465–470
Furuya Y, Lundmo P, Short AD, Gill DL, Isaacs JT . 1994 Cancer Res. 54: 6167–6175
Griffith TS, Rauch CT, Smolak PJ, Waugh JY, Boiani N, Lynch D, Smith CA, Goodwin RG, Kubin MZ . 1999 J. Immunol. 176: 2597–2605
Gross A, McDonnell JM, Korsmeyer SJ . 2000 Genes Dev. 13: 1899–1911
Huang Y, He Q, Hillman MJ, Rong R, Sheikh MS . 2001 Cancer Res. 61: 6918–6924
Jackman J, Alamo Jr I, Fornace Jr AJ . 1994 Cancer Res. 54: 5656–5662
Kaufman RJ . 1999 Genes Dev. 13: 1211–1233
Krebs J . 1998 BioMetals. 11: 375–382
Li H, Zhu H, Xu CJ, Yuan J . 1998 Cell 94: 491–501
Li WW, Alexandre S, Cao X, Lee AS . 1993 J. Biol. Chem. 268: 12003–12009
Luo X, Budihardjo I, Zou H, Slaughter C, Wang X . 1998 Cell 94: 481–490
Orth K, O'Rourke K, Salvesen GS, Dixit VM . 1996 J. Biol. Chem. 271: 20977–20980
Ron D, Habener JF . 1992 Genes Dev. 6: 439–453
Ruth JH, Esnault S, Jarzembowski JA, Malter JS . 1999 Am. J. Respir. Cell Mol. Biol. 21: 621–628
Sagara Y, Inesi G . 1991 J. Biol. Chem. 266: 13503–13506
Schulze-Osthoff K, Ferrari D, Los M, Wesselborg S, Peter ME . 1998 Eur. J. Biochem. 254: 439–459
Sheikh MS, Li XS, Chen JC, Shao ZM, Ordonez JV, Fontana JA . 1994 Oncogene 9: 3407–3415
Sheikh MS, Burns TF, Huang Y, Wu GS, Amundson S, Brooks KS, Fornace Jr AJ, El-Deiry WS . 1998a Cancer Res. 58: 1593–1598
Sheikh MS, Antinore MJ, Huang Y, Fornace Jr AJ . 1998b Oncogene 17: 2555–2563
Sheikh MS, Fornace Jr AJ . 2000 Leukemia 14: 1509–1513
Takimoto R, El-Deiry WS . 2000 Oncogene 19: 1735–1743
Thornberry NA, Lazebnik Y . 1998 Science 281: 1312–1316
Waser M, Mesaeli N, Spencer C, Michalak M . 1997 J. Cell Biol. 138: 547–557
Wertz IE, Dixit VM . 2000 J. Biol. Chem. 275: 11470–11477
Wei MC, Lindsten T, Mootha VK, Weiler S, Gross A, Ashiya M, Thompson CB, Korsmeyer SJ . 2000 Genes Dev. 14: 2060–2071
Wu GS, Burns TF, McDonald III ER, Jiang W, Meng R, Krantz ID, Kao G, Gan DD, Zhou J-Y, Muschel R, Hamilton SR, Spinner NB, Markowitz S, Wu G, El-Deiry WS . 1997 Nat. Genet. 171: 141–143
Zhan Q, Fan S, Smith ML, Bae I, Yu K, Alamo Jr I, O'Connor PM, Fornace Jr AJ . 1996 DNA Cell Biol. 15: 805–815
Acknowledgements
Part of this work was initiated at the NCI/NIH and MS Sheikh thanks Ms Kia Brooks for help with the Northern blot analyses. This work was supported in part by United States Army Prostate Cancer Research Program grant DAMD 170010722 and NIH grant CA89043 to MS Sheikh and a Merit Review Award from the Medical Research Services of the Department of Veterans Affairs to A Hussain and National Institutes of Health Grant PO1HL27867.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, Q., Lee, D., Rong, R. et al. Endoplasmic reticulum calcium pool depletion-induced apoptosis is coupled with activation of the death receptor 5 pathway. Oncogene 21, 2623–2633 (2002). https://doi.org/10.1038/sj.onc.1205345
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205345
Keywords
This article is cited by
-
ER stress-induced cell death proceeds independently of the TRAIL-R2 signaling axis in pancreatic β cells
Cell Death Discovery (2022)
-
Synergistic antitumor effect of a penicillin derivative combined with thapsigargin in melanoma cells
Journal of Cancer Research and Clinical Oncology (2022)
-
Targeting the ESCRT-III component CHMP2A for noncanonical Caspase-8 activation on autophagosomal membranes
Cell Death & Differentiation (2021)
-
Cell death induced by the ER stressor thapsigargin involves death receptor 5, a non-autophagic function of MAP1LC3B, and distinct contributions from unfolded protein response components
Cell Communication and Signaling (2020)
-
Quinovic acid purified from medicinal plant Fagonia indica mediates anticancer effects via death receptor 5
Molecular and Cellular Biochemistry (2020)