Abstract
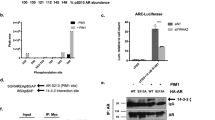
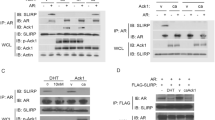
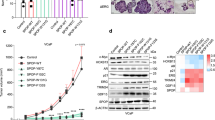
Androgen signaling influences the development and growth of prostate carcinoma. The transcriptional activity of androgen receptor (AR) is regulated by positive or negative transcriptional cofactors. We report here that PIAS1, PIAS3, and PIASy of the protein inhibitor of activated STAT (PIAS) family, which are expressed in human prostate, display distinct effects on AR-mediated gene activation in prostate cancer cells. While PIAS1 and PIAS3 enhance the transcriptional activity of AR, PIASy acts as a potent inhibitor of AR in prostate cancer cells. The effects of PIAS proteins on AR are competitive. PIASy binds to AR but does not affect the DNA binding activity of AR. An NH2-terminal LXXLL signature motif of PIASy, although not required for PIASy–AR interaction, is essential for the transrepression activity of PIASy. Our results identify PIASy as a transcriptional corepressor of AR and suggest that different PIAS proteins have distinct effects on AR signaling in prostate cancer cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Abreu-Martin MT, Chari A, Palladino AA, Craft NA, Sawyers CL . 1999 Mol. Cell. Biol. 19: 5143–5154
Bentel JM, Tilley WD . 1996 J. Endocrinol. 151: 1–11
Chung CD, Liao J, Liu B, Rao X, Jay P, Berta P, Shuai K . 1997 Science 278: 1803–1805
Darimont BD, Wagner RL, Apriletti JW, Stallcup MR, Kushner PJ, Baxter JD, Fletterick RJ, Yamamoto KR . 1998 Genes Dev. 12: 3343–3356
Darnell Jr JE . 1997 Science 277: 1630–1635
Freedman LP . 1999 Cell 97: 5–8
Glass CK, Rosenfeld MG . 2000 Genes Dev. 14: 121–141
Heery DM, Kalkhoven E, Hoare S, Parker MG . 1997 Nature 387: 733–736
Horwitz KB, Jackson TA, Bain DL, Richer JK, Takimoto GS, Tung L . 1996 Mol. Endocrinol. 10: 1167–1177
Hu X, Lazar MA . 1999 Nature 402: 93–96
Huang W, Shostak Y, Tarr P, Sawyers C, Carey M . 1999 J. Biol. Chem. 274: 25756–25768
Khan KD, Shuai K, Lindwall G, Maher SE, Darnell Jr JE, Bothwell AL . 1993 Proc. Natl. Acad. Sci. USA 90: 6806–6810
Kokontis JM, Liao S . 1999 Vitam. Horm. 55: 219–307
Liao J, Fu Y, Shuai K . 2000 Proc. Natl. Acad. Sci. USA 97: 5267–5272
Liu B, Gross M, ten Hoeve J, Shuai K . 2001 Proc. Natl. Acad. Sci. USA 98: 3203–3207
Liu B, Liao J, Rao X, Kushner SA, Chung CD, Chang DD, Shuai K . 1998 Proc. Natl. Acad. Sci. USA 95: 10626–10631
McDonald S, Brive L, Agus DB, Scher HI, Ely KR . 2000 Cancer Res. 60: 2317–2322
McInerney EM, Rose DW, Flynn SE, Westin S, Mullen TM, Krones A, Inostroza J, Torchia J, Nolte RT, Assa-Munt N, Milburn MV, Glass CK, Rosenfeld MG . 1998 Genes Dev. 12: 3357–3368
Moilanen AM, Karvonen U, Poukka H, Yan W, Toppari J, Jeanne OA, Palvimo JJ . 1999 J. Biol. Chem. 274: 3700–3704
Nagy L, Kao HY, Love JD, Li C, Banayo E, Gooch JT, Krishna V, Chatterjee K, Evans RM, Schwabe JW . 1999 Genes Dev. 13: 3209–3216
Neaear AM, Beaurang PA, Zhou S, Abraham S, Solomon W, Tjian R . 1999 Nature 398: 828–832
Parker SL, Tong T, Bolden S, Wingo PA . 1997 CA Cancer J. Clin. 47: 5–27
Perissi V, Staszewski LM, McInerney EM, Kurokawa R, Krones A, Rose DW, Lambert MH, Milburn MV, Glass CK, Rosenfeld MG . 1999 Genes Dev. 13: 3198–3208
Rachez C, Lemon DB, Suldan Z, Bromleigh V, Gamble M, Neaear AM, Erdjument-Bromage H, Tempst P, Freedman LP . 1999 Nature 398: 824–828
Shuai K . 1999 Prog. Biophys. Mol. Biol. 71: 405–422
Shuai K . 2000 Oncogene 19: 2638–2644
Shuai K, Schindler C, Prezioso VR, Darnell Jr JE . 1992 Science 258: 1808–1812
Tan J, Hall SH, Hamil KG, Grossman G, Petrusz P, Liao J, Shuai K, French FS . 2000 Mol. Endocrinol. 14: 14–24
Torchia J, Glass C, Rosenfeld MG . 1998 Curr. Opin. Cell. Biol. 10: 373–383
Torchia J, Rose DW, Inostroza J, Kamei Y, Westin S, Glass CK, Rosenfeld MG . 1997 Nature 387: 677–684
Trapman J., Cleutjens KB . 1997 Semin. Cancer Biol. 8: 29–36
Acknowledgements
We thank C Sawyers for discussion and reading the manuscript; X Rao and M Sohn for technical assistance. B Liu is a Cancer Research Institute fellow. Supported by a grant from the NIH (AI43438).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gross, M., Liu, B., Tan, Ja. et al. Distinct effects of PIAS proteins on androgen-mediated gene activation in prostate cancer cells. Oncogene 20, 3880–3887 (2001). https://doi.org/10.1038/sj.onc.1204489
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204489
Keywords
This article is cited by
-
ROR2 suppresses metastasis of prostate cancer via regulation of miR-199a-5p–PIAS3–AKT2 signaling axis
Cell Death & Disease (2020)
-
Quantitative SUMO proteomics identifies PIAS1 substrates involved in cell migration and motility
Nature Communications (2020)
-
The conserved ancient role of chordate PIAS as a multilevel repressor of the NF-κB pathway
Scientific Reports (2017)
-
PIAS1 is a determinant of poor survival and acts as a positive feedback regulator of AR signaling through enhanced AR stabilization in prostate cancer
Oncogene (2016)
-
Androgen receptor signaling in prostate cancer
Cancer and Metastasis Reviews (2014)