Abstract
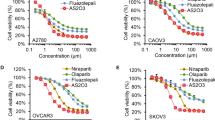
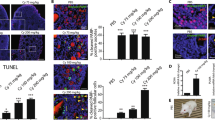
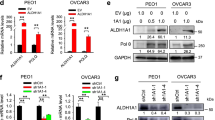
We investigated the intracellular mechanisms of retinoic acid (9-cis-RA, 13-cis-RA or all-trans-RA) and a cyclic AMP analog 8-Cl-cAMP on growth-inhibition and apoptosis in human ovarian cancer NIH: OVCAR-3 and OVCAR-8 cells. The cyclic AMP analog, 8-Cl-cAMP, acted synergistically with RA in inducing and activating retinoic acid receptor β (RARβ) which correlated with the growth inhibition, cell cycle arrest, and apoptosis in both cell types. In addition, combined treatment of cells with RA plus 8-Cl-cAMP resulted in the release of cytochrome c, loss in mitochondrial membrane potential and activation of caspase-3 followed by cleavage of anti-poly(ADP-ribose)polymerase and DNA-dependent protein kinase (catalytic subunit). Interestingly, inhibition of caspase-3 activation blocked RA plus 8-Cl-cAMP induced apoptosis. Furthermore, mutations in a CRE-related motif within the RARβ promoter resulted in loss of both transcriptional activation of RARβ and synergy between RA and 8-Cl-cAMP. Thus, RARβ can mediate RA and/or cyclic AMP action in ovarian cancer cells by promoting apoptosis. Loss of RARβ expression, therefore, may contribute to the tumorigenicity of human ovarian cancer cells. These findings suggest that RA and 8-Cl-cAMP act in a synergistic fashion in inducing apoptosis via caspase-3 activation, and may have potential for combination biotherapy for the treatment of malignant disease such as ovarian cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- RARβ:
-
retinoic acid receptor β
- RA:
-
retinoic acid
- PKA:
-
cAMP-dependent protein kinase
- C:
-
catalytic subunit
- PARP:
-
Poly(ADP-ribose)polymerase PARP
- DNA-PK:
-
DNA-dependent protein kinase
References
Angel P, Imagawa M, Chiu R, Stein B, Imbra RJ, Rahmsdorf HJ, Jonat C, Herlich P and Karin M. . 1987 Cell 49: 729–739.
Berard J, Laboune F, Mukuna M, Masse S, Kothary R and Bradley WE. . 1996 FASEB J. 10: 1091–1097.
Bernardi P, Broekemeier KM and Pfeiffer DR. . 1994 J. Bioenerg. Biomembr. 26: 509–517.
Chambon P. . 1996 FASEB J. 10: 940–954.
Cho-Chung YS. . 1990 Cancer Res. 50: 7093–7100.
de The H, Vivanco-Ruiz MDM, Tiollais P, Stunnenberg H and Dejean A. . 1990 Nature 343: 177–180.
Foulkes NS, Borrelli E and Sassone-Corsi P. . 1991 Cell 64: 739–749.
Gottlieb TM and Jackson SP. . 1993 Cell 72: 131–142.
Hartley KO, Gell D, Smith GC, Zhang H, Divecha N, Connelly MA, Admon A, Lees-Miller SP, Anderson CW and Jackson SP. . 1995 Cell 82: 849–856.
Heiden MGV, Chandel NS, Williamson EK, Schumacker PT and Thompson CB. . 1997 Cell 91: 627–637.
Henkart PA. . 1996 Immunity 4: 195–201.
Hoffman B, Lehmann JM, Zhang XK, Hermann T, Husmann M, Graupner G and Pfahl M. . 1990 Mol. Endocrinol. 4: 1727–1736.
Hong WK and Itri LM. . 1994 The Retinoids: Biology, Chemistry and Medicine. Sporn MB, Roberts AB and Goodman DS (ed).. Raven Press: New York pp. 597–630.
Hong WK, Lippman SM, Hittelman WN and Lotan R. . 1995 Clin. Cancer Res. 1: 677–686.
Houle B, Ledue F and Bradley WEC. . 1993 Exp. Cell Res. 195: 163–170.
Kaufmann SH, Desnoyers S, Ottaviano Y, Davidson NE and Poirier GG. . 1993 Cancer Res. 53: 3976–3985.
Koga M and Sutherland RL. . 1991 J. Steroid Biochem. Mol. Biol. 39: 455–460.
Krebs EG and Beavo JA. . 1979 Annu. Rev. Biochem. 88: 923–939.
Kruyt FA, Folkers G, van den Brink CE and van der Saag PT. . 1992 Nucleic Acid Res. 20: 6393–6399.
Lanotte M, Riviere JB, Hermouet S, Houge G, Wintermyr OK, Gjertsen BT and Doskeland SO. . 1991 J. Cell Physiol. 146: 73–80.
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG and Earnshaw WC. . 1994 Nature (Lond.) 371: 346–347.
Liu Y, Lee M, Wang H-G, Li Y, Hashimoto Y, Klaus M, Reed JC and Zhang X-K. . 1996a Mol. Cell. Biol. 16: 1138–1149.
Liu X, Kim CN, Yang J, Jemmerson R and Wang X. . 1996b Cell 86: 147–157.
Lees-Miller SP, Godbout R, Chan DW, Weinfeld M, Day RS III, Barron GM and Turner JA. . 1995 Science 267: 1183–1185.
Lotan R, Xu X-C, Lippman SM, Ro JY, Lee JS, Lee JJ and WK Hong WK. . 1995 N. Engl. J. Med. 332: 1405–1410.
Nieminen AL, Saylor AK, Tesfai SA, Herman B and Lemasters JJ. . 1995 Biochem. J. 307: 99–106.
Pastorini JG, Chen S-T, Tafani M, Snyder JW and Farber JF. . 1998 J. Biol. Chem. 273: 7770–7775.
Reed JC. . 1997 Cell 91: 559–562.
Sabichi AL, Hendricks DT, Bober MA and Birrer MJ. . 1998 J. Natl. Can. Inst. 90: 597–605.
Seewaldt VL, Johnson BS, Parker MB, Collins SJ and Swisshelm K. . 1995 Cell Growth Differ. 6: 1077–1088.
Song Q, Lees-Miller SP, Kumar S, Zhang N, Chan DW, Smith GCM, Jackson SP, Alnemri ES, Litwack G, Khanna KK and Lavin MF. . 1996 EMBO J. 15: 3238–3426.
Sporn MB, Roberts AB and Goodman DS. . 1994 The Retinoids: Biology, Chemistry and Medicine, Ed. 2. New York: Raven Press.
Srivastava RK, Srivastava AR, Seth P, Agrawal S and Cho-Chung YS. . 1998a Mol. Cell. Biochem. (in press).
Srivastava RK, Srivastava AR and Cho-Chung YS. . 1998b Breast Can. Res. Treat. 49: 97–107.
Srivastava RK, Srivastava AR and Cho-Chung YS. . 1998c Clin. Cancer Res. 4: 755–761.
Swisshelm K, Ryan K, Lee X, Tsou HC, Beacocke M and Sager R. . 1994 Cell Growth Differ. 5: 133–141.
Tortora G, Ciardiello F, Pepe S, Tagliaferri P, Ruggiero A, Bianco C, Guarrasi R, Miki K and Bianco AR. . 1995 Clin. Can. Res. 1: 377–384.
Vavssiäre J-L, Petit PX, Risler Y and Mignotte B. . 1994 Proc. Natl. Acad. Sci. USA 91: 11752–11756.
Vintermyr OK, Gjersten BT, Lanotte M and Doskeland SO. . 1993 Exp. Cell. Res. 206: 157–161.
White E. . 1996 Genes Dev. 10: 1–15.
Widschwendter M, Daxenbichler G, Dapunt O and Marth C. . 1995 Cancer Res. 55: 2135–2139.
Wyllie AH. . 1995 Curr. Opin. Genet. Dev. 5: 97–104.
Xu X-C, Sozzi G, Lee JS, Lee JJ, Pastorino U, Pilotti S, Kurie JM, Hong WK and Lotan R. . 1997a J. Natl. Cancer Inst. 89: 624–629.
Xu X-C, Sneige N, Liu X, Nandagiri R, Lee JJ, Lukmanji F, Hortobagyi G, Lippman SM and Lotan R. . 1997b Cancer Res. 57: 4992–4996.
Zhang X-K, Liu Y, Lee M-O and Pfahl M. . 1994 Cancer Res. 54: 5663–5669.
Acknowledgements
We are grateful to Drs P Chambon (Strasbourg, France) and X-K Zhang (California, USA) for providing retinoic acid receptor expression vectors. We also thank Dr JA Fontana (Baltimore, USA) for providing pRAREβ-tk-CAT expression vector.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Srivastava, R., Srivastava, A., Cho-Chung, Y. et al. Synergistic effects of retinoic acid and 8-Cl-cAMP on apoptosis require caspase-3 activation in human ovarian cancer cells. Oncogene 18, 1755–1763 (1999). https://doi.org/10.1038/sj.onc.1202464
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202464
Keywords
This article is cited by
-
Promoter Polymorphisms in the β-2 Adrenergic Receptor Are Associated With Drug-Induced Gene Expression Changes and Response in Acute Lymphoblastic Leukemia
Clinical Pharmacology & Therapeutics (2010)
-
Gene network analysis of oxidative stress-mediated drug sensitivity in resistant ovarian carcinoma cells
The Pharmacogenomics Journal (2010)
-
Detection of Deregulated Pathways to Lymphatic Metastasis in Oral Squamous Cell Carcinoma
Pathology & Oncology Research (2009)
-
The T393C polymorphism in the gene GNAS1 of G protein is associated with survival of patients with invasive breast carcinoma
Breast Cancer Research and Treatment (2007)
-
Adaptor proteins in protein kinase C-mediated signal transduction
Oncogene (2001)