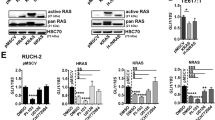
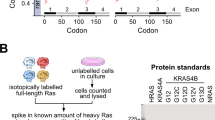
Abstract
The products (p21) of the three mammalian H-, N- and K-ras genes play important roles in intracellular signal transduction, linking membrane receptor kinases to the nuclear pathway through raf and mitogen activated protein kinase. They are involved in the regulation of proliferation and differentiation, and activating mutations of these genes are commonly associated with human cancers. Two p21 proteins are encoded by the K-ras gene (p21K-rasA and p21K-rasB) due to alternative splicing of the last exon. While the four p21ras proteins are highly homologous, their sequences diverge significantly at the C-termini, to which distinct biochemical and perhaps even functional differences may be ascribed. However, H-, N- and K-rasB appear to be ubiquitously expressed, with little evidence of tissue-specific or developmental regulation. In contrast, we now demonstrate that the expression of K-rasA is strikingly different. K-rasA is induced during differentiation of pluripotent embryonal stem cells in vitro. Its expression during early embryogenesis is limited temporally and spatially in a tissue-specific distribution which is largely maintained as an adult. This suggests a distinct biological role for p21K-rasA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pells, S., Divjak, M., Romanowski, P. et al. Developmentally-regulated expression of murine K-ras isoforms. Oncogene 15, 1781–1786 (1997). https://doi.org/10.1038/sj.onc.1201354
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1201354
Keywords
This article is cited by
-
Targeting KRAS mutant cancers: from druggable therapy to drug resistance
Molecular Cancer (2022)
-
Role of oncogenic KRAS in the prognosis, diagnosis and treatment of colorectal cancer
Molecular Cancer (2021)
-
Targeting KRAS4A splicing through the RBM39/DCAF15 pathway inhibits cancer stem cells
Nature Communications (2021)
-
RAS-targeted therapies: is the undruggable drugged?
Nature Reviews Drug Discovery (2020)
-
Splice variants of RAS—translational significance
Cancer and Metastasis Reviews (2020)