Abstract
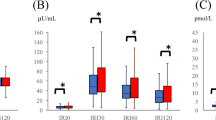
Essential hypertension is associated with changes in central catecholaminergic pathways which might also be reflected in the pituitary response to stress stimuli. The aim of this study was to determine whether the response of pituitary hormones, cortisol, plasma renin activity, aldosterone and catecholamines to insulin-induced hypoglycaemia is changed in hypertension. We studied 22 young lean male patients with newly diagnosed untreated essential hypertension and 19 healthy normotensive, age- and body mass index (BMI)-matched controls. All subjects underwent an insulin tolerance test (0.1 IU insulin/kg body weight intravenously) with blood sampling before and 15, 30, 45, 60 and 90 min after insulin administration. Increased baseline levels of norepinephrine (P<0.05), increased response of norepinephrine (P<0.001) and decreased response of growth hormone (P<0.001), prolactin (P<0.001), adrenocorticotropic hormone (P<0.05) and cortisol (P<0.001) were found in hypertensive patients when compared to normotensive controls. Increased norepinephrine levels and a decreased pituitary response to metabolic stress stimuli may represent another manifestation of chronically increased sympathetic tone in early hypertension.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Julius S . Borderline hypertension. Clin Exp Hypertens 1999; 21: 741–747.
Svetkey LP . Management of prehypertension. Hypertension 2005; 45: 1056–1061.
Amerena J, Julius S . The role of the autonomic nervous system in hypertension. Hypertens Res 1995; 18: 99–110.
Rose KM, North K, Arnett DK, Ellison RC, Hunt SC, Lewis CE et al. Blood pressure and pulse responses to three stressors: associations with sociodemographic characteristics and cardiovascular risk factors. J Hum Hypertens 2004; 18: 333–341.
Touyz RM, Campbell N, Logan A, Gledhill N, Petrella R, Padwal R . Canadian Hypertension Education Program: the 2004 Canadian recommendations for the management of hypertension: Part III – Lifestyle modifications to prevent and control of hypertension. Can J Cardiol 2004; 20: 55–59.
Elenkov IJ, Wilder RL, Chrousos GP, Vizi S . The sympathetic nerve – an integrative interface between two supersystems: the brain and the immune system. Pharmacol Rev 2000; 52: 595–638.
Tsigos C, Chrousos GP . Hypothalamic–pituitary–adrenal axis, neuroendocrine factors and stress. J Psychosom Res 2002; 53: 865–871.
Goldstein DS . Plasma catecholamines and essential hypertension. An analytical review. Hypertension 1983; 5: 86–99.
DeQuattro V, Feng M . The sympathetic nervous system: the muse of primary hypertension. J Hum Hypertens 2002; 16 (Suppl 1): S64–S69.
Al-Damluji S, Francis D . Activation of central alpha 1-adrenoceptors in humans stimulates secretion of prolactin and TSH, as well as ACTH. Am J Physiol 1993; 264 (2 Part 1): E208–E214.
Kiem DT, Barna I, Koenig JI, Makara GB . Adrenocorticotropin, prolactin and beta-endorphin stimulatory action of alpha-2-adrenoceptor antagonists. Neuroendocrinology 1995; 61: 152–158.
Vigas M, Kvetnansky R, Jurcovicova J, Jezova D, Tatar P . Comparison of catecholamine and adenopituitary hormone responses to various stress stimuli in man. In: Usdin E, Kvetnansky R and Axelrod J (eds). Stress: The Role of Catecholamines and Other Neurotransmitters. Gordon and Breach Science Publishers: New York, 1984, 865–882.
Pacak K, Palkovits M . Stressor specificity of central neuroendocrine responses: implications for stress-related disorders. Endocr Rev 2001; 22: 502–548.
European Society of Hypertension – European Society of Cardiology Guidelines Committee. European Society of Hypertension – European Society of Cardiology guidelines for the management of arterial hypertension. J Hypertens 2003; 21: 1011–1053.
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2004; 27: S5–S10.
Peuler JD, Johnson GA . Simultaneous single isotope radioenzymatic assay of plasma norepinephrine, epinephrine and dopamine. Life Sci 1977; 21: 625–636.
Carrasco GA, Van De Kar LD . Neuroendocrine pharmacology of stress. Eur J Pharmacol 2003; 463: 235–272.
De Kloet RE . Hormones, brain and stress. Endocr Regul 2003; 37: 51–68.
Jezova D, Makatsori A, Duncko R, Moncek F, Jakubek M . High trait anxiety in healthy subjects is associated with low neuroendocrine activity during psychosocial stress. Prog Neuropsychopharmacol Biol Psychiatry 2004; 28: 1331–1336.
Muller EE, Locatelli V, Cocchi D . Neuroendocrine control of growth hormone secretion. Physiol Rev 1999; 79: 511–607.
Al-Damluji S . Adrenergic control of the secretion of anterior pituitary hormones. Baillieres Clin Endocrinol Metab 1993; 7: 355–392.
Tatar P, Vigas M . Role of alpha 1- and alpha 2-adrenergic receptors in the growth hormone and prolactin response to insulin-induced hypoglycemia in man. Neuroendocrinology 1984; 39: 275–280.
Bolli GB, Fanelli CG . Physiology of glucose counterregulation to hypoglycemia. Endocrinol Metab Clin North Am 1999; 28: 467–493.
Schwartz NS, Clutter WE, Shah SD, Cryer PE . Glycemic thresholds for activation of glucose counterregulatory systems are higher than the thresholds for symptoms. J Clin Invest 1987; 79: 777–781.
Mitrakou A, Ryan C, Veneman T, Mokan M, Jenssen T, Kiss I et al. Hierarchy of glycemic thresholds for counterregulatory hormone secretion, symptoms and cerebral dysfunction. Am J Physiol Endocrinol Metab 1991; 260: E67–E74.
Kinsley BT, Levy CJ, Simonson DC . Prolactin and beta endorphin responses to hypoglycemia are reduced in well-controlled insulin-dependent diabetes mellitus. Metabolism 1996; 45: 1434–1440.
Giustina A, Doga M, Bossoni S, Bodini C, Legati F, Pizzocolo G et al. Central alpha-2 adrenergic function in patients with essential hypertension. Horm Metab Res 1990; 22: 451–452.
Julius S, Nesbitt S . Clinical consequences of the autonomic imbalance in hypertension and congestive heart failure. Scand Cardiovasc J Suppl 1998; 47: 23–30.
Schlaich MP, Lambert E, Kaye DM, Krozovski Z, Campbell DJ, Lambert G et al. Sympathetic augmentation in hypertension: role of nerve firing, norepinephrine reuptake, and angiotensin neuromodulation. Hypertension 2004; 43: 169–175.
Esler M, Lux A, Jennings G, Hastings J, Socratous F, Lambert G . Rilmenidine sympatholytic activity preserves mental stress, orthostatic sympathetic responses and adrenaline secretion. J Hypertens 2004; 22: 1529–1534.
Frey MJ, Lanoce V, Molinoff PB, Wilson JR . Skeletal muscle beta-receptors and isoproterenol-stimulated vasodialtation in canine heart failure. J Appl Physiol 1989; 67: 2026–2031.
Hayes MJ, Qing F, Rhodes CG, Rahman SU, Ind PW, Sriskandan S et al. In vivo quantification of human pulmonary beta-adrenoreceptors: effect of beta-agonist therapy. Am J Resp Crit Care Med 1996; 154: 1277–1283.
Beau SL, Tolley TK, Saffitz JE . Heterogeneous transmural distribution of beta-adrenergic receptor subtypes in failing human hearts. Circulation 1993; 88: 2501–2509.
DeRosa MA, Cryer PE . Hypoglycemia and the sympathoadrenal system: neurogenic symptoms are largely the result of sympathetic neural, rather than adrenomedullary, activation. Am J Physiol Endocrinol Metab 2004; 287: E32–E41.
Eide I, Kolloch R, DeQuattro V, Miano L, Dugger R, Van der Meulen J . Raised cerebrospinal fluid norepinephrine in some patients with primary hypertension. Hypertension 1979; 1: 255–260.
Kawano Y, Fukiyama K, Takeya Y, Abe I, Omae T . Catecholamines, angiotensin II and odium concentrations in cerebrospinal fluid in young men with borderline hypertension. Clin Exp Hypertens A 1984; 6: 1131–1145.
Cubeddu LX, Hoffman IS . Cerebrospinal fluid norepinephrine levels in essential hypertension: effects of drug treatment and withdrawal. J Cardiovasc Pharmacol 1987; 10 (Suppl 12): S205–S210.
Lambert GW, Ferrier C, Kaye DM, Kalff V, Kelly MJ, Cox HS et al. Monoaminergic neuronal activity in subcortical brain regions in essential hypertension. Blood Pressure 1994; 3: 55–66.
Lambert GW, Ferrier C, Kaye DM, Jennings GL, Kalff V, Kelly MJ et al. Central nervous system norepinephrine turnover in essential hypertension. Ann NY Acad Sci 1995; 763: 679–694.
Tan Y, Gan Q, Knuepfer MM . Central alpha-adrenergic receptors and corticotrophin releasing factor mediate hemodynamic responses to acute cold stress. Brain Res 2003; 968: 122–129.
Tsuda K, Tsuda S, Nishio I . Role of alpha-2-adrenergic receptors and cyclic adenosine monophosphate-dependent protein kinase in the regulation of norepinephrine release in the central nervous system of spontaneously hypertensive rats. J Cardiovasc Pharmacol 2003; 42 (Suppl 1): S81–S85.
Barbieri C, Ferrari C, Caldara R, Curtarelli G . Growth hormone secretion in hypertensive patients: evidence for a derangement in central adrenergic function. Clin Sci (London) 1980; 58: 135–138.
Alba-Roth J, Losa M, Spiess Y, Schopohl J, Muller OA, von Werder K . Interaction of clonidine and GHRH on GH secretion in vivo and in vitro. Clin Endocrinol (Oxford) 1989; 30: 485–491.
Cella SG, Morgese M, Mantegazza P, Muller EE . Inhibitory action of the alpha 1-adrenergic receptor on growth hormone secretion in the dog. Endocrinology 1984; 114: 2406–2408.
Jezova-Repcekova D, Klimes I, Jurcovicova J, Vigas M . Effect of adrenergic receptor blockade on cortisol and GH response to insulin-induced hypoglycemia in man. Int J Clin Pharmacol Biopharm 1979; 17: 64–67.
Karhuvaara S, Kallio A, Koulu M, Scheinin H, Scheinin M . No involvement of alpha 2- adrenoceptors in the regulation of basal prolactin secretion in healthy men. Psychoneuroendocrinology 1990; 15: 125–129.
Krulich L, Jurcovicova J, Le T . Prolactin (PRL) release-inhibiting properties of the alpha 2 adrenergic receptor antagonist idazoxan: comparison with yohimbine. Life Sci 1989; 44: 809–818.
Kapoor R, Chapman IM, Willoughby JO . Alpha 2 and beta adrenoceptors in the mediobasal hypothalamus and alpha 2 adrenoceptors in the preoptic-anterior hypothalamus stimulate prolactin secretion in the conscious male rat. J Neuroendocrinol 1993; 5: 189–193.
Jurcovicova J, Le T, Krulich L . The paradox of alpha 2 adrenergic regulation of prolactin (PRL) secretion. I. The PRL-releasing action of the alpha 2 receptor agonist. Brain Res Bull 1989; 23: 417–424.
Jurcovicova J, Le T, Krulich L . The paradox of alpha 2 adrenergic regulation of prolactin (PRL) secretion. II. The PRL-releasing action of the alpha 2 receptor antagonists. Brain Res Bull 1989; 23: 425–432.
Cuneo RC, Livesey JH, Nicholls MG, Espiner EA, Donald RA . Effects of alpha-2 adrenoreceptor blockade by yohimbine on the hormonal response to hypoglycaemic stress in normal man. Horm Metab Res 1989; 21: 33–36.
Acknowledgements
This study was supported by the grants of Slovak State Program SP 51/02280800/0280/802 and of Slovak Scientific Grants Agency VEGA 2/3150/24.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radikova, Z., Penesova, A., Cizmarova, E. et al. Decreased pituitary response to insulin-induced hypoglycaemia in young lean male patients with essential hypertension. J Hum Hypertens 20, 510–516 (2006). https://doi.org/10.1038/sj.jhh.1002026
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jhh.1002026
Keywords
This article is cited by
-
Low prolactin levels are associated with visceral adipocyte hypertrophy and insulin resistance in humans
Endocrine (2020)
-
Insulin resistance in young, lean male subjects with essential hypertension
Journal of Human Hypertension (2011)