Abstract
Objective:
To assess whether circadian desynchronization leads to metabolic alterations capable of promoting dietary obesity and/or impairing glucose tolerance.
Design:
Rats fed either with chow pellets (i.e., low-fat diet with 4% mass of fat) or high-fat diet (34% mass of fat). Half of each diet group was exposed to a fixed light–dark cycle or to a 10-h weekly shift in the light–dark cycle from Thursday to Sunday (20 shifts). To enforce the shifted animals to be active at unusual times of the day, food was available only during the daily dark period for all groups.
Results:
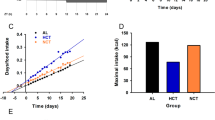
Shifting the light–dark cycle on a weekly basis was efficient to induce circadian desynchronization, as evidenced by strong disturbances in the daily expression of locomotor activity. Shifted rats fed with a nocturnal low-fat diet had lower plasma insulin and similar blood glucose compared to rats fed with the same diet under a fixed light–dark cycle. Nocturnal high-fat feeding led to an abdominal fat overload associated with increased plasma leptin and basal glucose. These metabolic changes were not significantly modified by circadian desynchronization.
Conclusion:
Chronic desynchronization with low-fat diet impaired insulin regulation. Metabolic changes induced by the high-fat diet were not aggravated by chronic desynchronization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Fukagawa K, Sakata T, Yoshimatsu H, Fujimoto K, Uchimura K, Asano C . Advance shift of feeding circadian rhythm induced by obesity progression in Zucker rats. Am J Physiol Regul Integr Comp Physiol 1992; 263: R1169–R1175.
Van Cauter E, Polonsky KS, Scheen AJ . Roles of circadian rhythmicity and sleep in human glucose regulation. Endocr Rev 1997; 18: 716–738.
Mistlberger RE, Lukman H, Nadeau BG . Circadian rhythms in the Zucker obese rat: assessment and intervention. Appetite 1998; 30: 255–267.
Van Reeth O . Sleep and circadian disturbances in shift work: strategies for their management. Horm Res 1998; 49: 158–162.
Hampton SM, Morgan LM, Lawrence N, Anastasiadou T, Norris F, Deacon S et al. Postprandial hormone and metabolic responses in simulated shift work. J Endocrinol 1996; 151: 259–267.
Ribeiro DC, Hampton SM, Morgan L, Deacon S, Arendt J . Altered postprandial hormone and metabolic responses in a simulated shift work environment. J Endocrinol 1998; 158: 305–310.
Simon C, Weibel L, Brandenberger G . Twenty-four-hour rhythms of plasma glucose and insulin secretion rate in regular night workers. Am J Physiol Endocrinol Metab 2000; 278: E413–E420.
Rosa RR, Bonnet MH, Bootzin RR, Eastman CI, Penn PE, Tepas DI et al. Intervention factors for promoting adjustement to nightwork and shiftwork. Occup Med 1990; 5: 391–414.
Rosmond R, Lapidus L, Bjorntorp P . The influence of occupational and social factors on obesity and body fat distribution in middle-aged men. Int J Obes Relat Metab Disord 1996; 20: 599–607.
Morgan L, Hampton S, Gibbs M, Arendt J . Circadian aspects of postprandial metabolism. Chronobiol Int 2003; 20: 795–808.
Karlsson B, Knutsson A, Lindahl B . Is there an association between shift work and having a metabolic syndrome? Results from a population based study of 27 485 people. Occup Environ Med 2001; 58: 747–752.
Turek FW, Pinto LH, Vitaterna MH, Penev PD, Zee PC, Takahashi JS . Pharmacological and genetic approaches for the study of circadian rhythms in mammals. Front Neuroendocrinol 1995; 16: 191–223.
Challet E, Solberg LC, Turek FW . Entrainment in calorie-restricted mice: conflicting zeitgebers and free-running conditions. Am J Physiol Regul Integr Comp Physiol 1998; 274: R1751–1761.
Challet E, Losee-Olson S, Turek FW . Reduced glucose availability attenuates circadian responses to light in mice. Am J Physiol Regul Integr Comp Physiol 1999; 276: R1063–1070.
Rutter J, Reick M, McKnight SL . Metabolism and the control of circadian rhythms. Annu Rev Biochem 2002; 71: 307–331.
Van Someren EJ, Swaab DF, Colenda CC, Cohen W, McCall WV, Rosenquist PB . Bright light therapy: improved sensitivity to its effects on rest-activity rhythms in Alzheimer patients by application of nonparametric methods. Chronobiol Int 1999; 16: 505–518.
Hill JO, Lin D, Yakubu F, Peters JC . Development of dietary obesity in rats: influence of amount and composition of dietary fat. Int J Obes Relat Metab Disord 1992; 16: 321–333.
Himms-Hagen J, Cui J . Diet-induced obesity and brown adipose tissue. In: Romsos DR, Himms-Hagen J, Suzuki M (eds) Obesity: Dietary Factors and Control. Karger: Basel, 1991. pp 81–95.
Woods SC, Seeley RJ, Rushing PA, D’Alessio D, Tso P . A controlled high-fat diet induces an obese syndrome in rats. J Nutr 2003; 133: 1081–1087.
Chalkley SM, Hettiarachchi M, Chisholm DJ, Kraegen EW . Long-term high-fat feeding leads to severe insulin resistance but not diabetes in Wistar rats. Am J Physiol Endocrinol Metab 2002; 282: E1231–1238.
Stark AH, Timar B, Madar Z . Adaptation of Sprague–Dawley rats to long-term feeding of high fat or high fructose diets. Eur J Nutr 2000; 39: 229–234.
Wang Y, Wang PY, Qin LQ, Davaasambuu G, Kaneko T, Xu J et al. The development of diabetes mellitus in Wistar rats kept on a high-fat/low-carbohydrate diet for long periods. Endocrine 2003; 22: 85–92.
Fehmann HC, Peiser C, Bode HP, Stamm M, Staats P, Hedetoft C et al. Leptin: A potent inhibitor of insulin secretion. Peptides 1997; 18: 1267–1273.
Tsiotra PC, Tsigos C, Raptis SA . TNFalpha and leptin inhibit basal and glucose-stimulated insulin secretion and gene transcription in the HIT-T15 pancreatic cells. Int J Obes Relat Metab Disord 2001; 25: 1018–1026.
Boozer CN, Schoenbach G, Atkinson RL . Dietary fat and adiposity: a dose-response relationship in adult male rats fed isocalorically. Am J Physiol Endocrinol Metab 1995; 268: E546–550.
Kalsbeek A, Barassin S, van Heerikhuize JJ, van der Vliet J, Buijs RM . Restricted daytime feeding attenuates reentrainment of the circadian melatonin rhythm after an 8-h phase advance of the light–dark cycle. J Biol Rhythms 2000; 15: 57–66.
Murakami DM, Horwitz BA, Fuller CA . Circadian rhythms of temperature and activity in obese and lean Zucker rats. Am J Physiol Regul Integr Comp Physiol 1995; 38: R1038–1043.
Cusin I, Rohner-Jeanrenaud F, Stricker-Krongrad A, Jeanrenaud B . The weight-reducing effect of an intracerebroventricular bolus injection of leptin in genetically obese fa/fa rats. Reduced sensitivity compared with lean animals. Diabetes 1996; 45: 1446–1450.
Qin LQ, Li J, Wang Y, Wang J, Xu JY, Kaneko T . The effects of nocturnal life on endocrine circadian patterns in healthy adults. Life Sci 2003; 73: 2467–2475.
Tsai LL, Tsai YC, Hwang K, Huang YW, Tzeng JE . Repeated light-dark shifts speed up body weight gain in male F344 rats. Am J Physiol Endocrinol Metab 2005. (March 1); [Epub ahead of print] doi:10.1152/ajpendo.00603.2004.
la Fleur SE, Kalsbeek A, Wortel J, Fekkes ML, Buijs RM . A daily rhythm in glucose tolerance: a role for the suprachiasmatic nucleus. Diabetes 2001; 50: 1237–1243.
Acknowledgements
We thank Pr Bernard Goichot (Service de Médecine Interne et Nutrition, Hôpital de Hautepierre, Université Louis Pasteur, Strasbourg, France), Dr Sylvie Massemin-Challet, and Dr Hugues Oudart (Centre d’Ecologie et Physiologie Energétiques, CNRS, Strasbourg, France) for help with analysis and comments on the manuscript. We are also indebted to Pr Eus JW Van Someren (Netherlands Institute for Brain Research, Amsterdam, The Netherlands) for the use of his analysis procedures to assess desynchronization of circadian rhythms. The study has been performed with the support of ‘crédits exceptionnels’ from Université Louis Pasteur.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bartol-Munier, I., Gourmelen, S., Pevet, P. et al. Combined effects of high-fat feeding and circadian desynchronization. Int J Obes 30, 60–67 (2006). https://doi.org/10.1038/sj.ijo.0803048
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803048
Keywords
This article is cited by
-
Interactions entre rythmicité circadienne et métabolisme
Obésité (2015)
-
Circadian clock control of endocrine factors
Nature Reviews Endocrinology (2014)
-
Circadian Rhythms, Metabolism, and Insulin Sensitivity: Transcriptional Networks in Animal Models
Current Diabetes Reports (2013)
-
Body weight, metabolism and clock genes
Diabetology & Metabolic Syndrome (2010)
-
Wheel running in female C57BL/6J mice: impact of oestrus and dietary fat and effects on sleep and body mass
International Journal of Obesity (2009)