Abstract
OBJECTIVE:
To examine the relation of obesity and overweight with organ-specific cancer mortality.
METHODS:
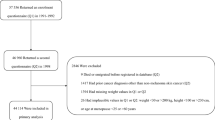
In the Whitehall prospective cohort study of London-based government employees, 18 403 middle-age men participated in a medical examination between 1967 and 1970. Subjects were followed up for cause-specific mortality for up to 35 y (median: interquartile range (25th–75th centile); 28.1 y: 18.6–33.8).
RESULTS:
There were over 3000 cancer deaths in this cohort. There was a raised risk of mortality from carcinoma of the rectum, bladder, colon, and liver, and for lymphoma in obese or overweight men following adjustment for range of covariates, which included socioeconomic position and physical activity. These relationships held after exclusion of deaths occurring in the first 20 y of follow-up.
CONCLUSION:
Avoidance of obesity and overweight in adult life may reduce the risk of developing some cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Actuarial Society of America and Association of Life Insurance Directors. Medico-actuarial mortality investigation. Spector Company: New York; 1913.
Whitlock G, Lewington S, Ni Mhurchu C . Coronary heart disease and body mass index: a systematic review of the evidence from larger prospective cohort studies. Seminars in vascular medicine (classical and emerging risk factors for cardiovascular disease, Part 2 2002. Vol 4, pp 369–381.
World Cancer Research Fund/American Institute for Cancer Research. Food, nutrition and the prevention of cancer: a global perspective. American Institute for Cancer Research: Washington; 1997.
IARC handbooks of cancer prevention. Weight control and physical activity Vol 6. International Agency for Research on Cancer: Lyons, France; 2002.
Bianchini F, Kaaks R, Vainio H . Overweight, obesity, and cancer risk. Lancet Oncol 2002; 3: 565–574.
Batty GD . Confounding effect of socioeconomic position in the study of height in relation to prostate cancer risk (letter). Br J Cancer 2004; 90: 1875.
Batty GD, Thune I . Does physical activity prevent cancer? Evidence suggests protection against colon cancer and probably breast cancer (editorial). BMJ 2000; 321: 1424–1425.
Reid DD, Hamilton PJS, McCartney P, Rose G, Jarrett RJ, Keen H, Rose G . Cardiorespiratory disease and diabetes among middle-aged male civil servants. Lancet 1974; i: 469–473.
Jarrett RJ, Shipley MJ, Rose G . Weight and mortality in the Whitehall Study. Br Med J (Clin Res Edn) 1982; 285: 535–537.
Jarrett RJ . Is there an ideal body weight? Br Med J (Clin Res Edn) 1986; 293: 493–495.
Fitzgerald AP, Jarrett RJ . Body weight and coronary heart disease mortality: an analysis in relation to age and smoking habit. 15 years follow-up data from the Whitehall Study. Int J Obes Relat Metab Disord 1992; 16: 119–123.
Davey Smith G, Leon D, Shipley MJ, Rose G . Socioeconomic differentials in cancer among men. Int J Epidemiol 1991; 20: 339–345.
Reid DD, Hamilton PJ, McCartney P, Rose G, Jarrett RJ, Keen H . Smoking and other risk factors for coronary heart-disease in British civil servants. Lancet 1976; 2: 979–984.
Davey Smith G, Shipley MJ, Rose G . Intermittent claudication, heart disease risk factors, and mortality. The Whitehall Study. Circulation 1990; 82: 1925–1931.
Rose G, McCartney P, Reid DD . Self-administration of a questionnaire on chest pain and intermittent claudication. Br J Prev Soc Med 1977; 31: 42–48.
Rose GA . The diagnosis of ischaemic heart pain and intermittent claudication in field studies. Bull World Health Organ 1962; 27: 645–658.
Committee on the aetiology of chronic bronchitis. Definition and classification of chronic bronchitis. Lancet 1965; i: 775–779.
Ben Shlomo Y, Davey Smith G, Shipley M, Marmot MG . Magnitude and causes of mortality differences between married and unmarried men. J Epidemiol Commun Health 1993; 47: 200–205.
Batty GD, Shipley M, Marmot M, Davey Smith G . Physical activity and cause-specific mortality in men: further evidence from the Whitehall study. Eur J Epidemiol 2002; 17: 863–869.
Batty GD, Shipley MJ, Marmot M, Davey Smith G . Physical activity and cause-specific mortality in men with type 2 diabetes/impaired glucose tolerance: evidence from the Whitehall study. Diabetes Med 2002; 19: 580–588.
Rose G, Hamilton PS, Keen H, Reid DD, McCartney P, Jarrett RJ . Myocardial ischaemia, risk factors and death from coronary heart-disease. Lancet 1977; 1: 105–109.
Davey Smith G, Shipley MJ, Marmot MG, Rose G . Plasma cholesterol concentration and mortality. The Whitehall Study. JAMA 1992; 267: 70–76.
Batty GD, Shipley M, Marmot M, Davey Smith G . Diabetes status and post-load plasma glucose concentration in relation to site-specific cancer mortality: findings from the original Whitehall study. Cancer Causes Control 2004; 15: 873–881.
Batty GD, Shipley MJ, Marmot MG, Davey Smith G . Blood pressure and site-specific cancer mortality: evidence from the original Whitehall study. Br J Cancer 2003; 89: 1243–1247.
Keen H, Thomas BJ, Jarrett RJ, Fuller JH . Nutrient intake, adiposity, and diabetes. Br Med J 1979; 1: 655–658.
Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ . Overweight, obesity, and mortality from cancer in a prospectively studied cohort of US adults. N Engl J Med 2003; 348: 1625–1638.
Henley SJ, Flanders WD, Manatunga A, Thun MJ . Leanness and lung cancer risk: fact or artifact? Epidemiology 2002; 13: 268–276.
Berrington dG, Sweetland S, Spencer E . A meta-analysis of obesity and the risk of pancreatic cancer. Br J Cancer 2003; 89: 519–523.
World Health Organisation. Physical status: the use and interpretation of anthropometry: report of a WHO expert committee WHO Technical Report Series. WHO: Geneva; 1995.
Anon. Manual of the international statistical classification of diseases, injuries, and causes of death (8th revision). World Health Organisation: Geneva; 1967.
Anon. Manual of the international statistical classification of diseases, injuries, and causes of death (9th revision). WHO: Geneva; 1977.
Anon. International statistical classification of diseases and related health problems (10th revision). WHO: Geneva; 1992.
Cox DR . Regression models and life-tables. J Roy Stat Soc Ser B 1972; 34: 187–220.
Davey Smith G, Shipley MJ, Batty GD, Morris JN, Marmot M . Physical activity and cause-specific mortality in the Whitehall study. Public Health 2000; 114: 308–315.
Coleman MP, Rachet B, Woods LM, Mitry E, Riga M, Cooper N, Quinn MJ, Brenner H, Esteve J . Trends and socioeconomic inequalities in cancer survival in England and Wales up to 2001. Br J Cancer 2004; 90: 1367–1373.
SAS Institute Inc. SAS/STAT(R) user's guide Version 6, volumes 1 & 2. SAS Institute Inc.: Cary, NC; 1989.
Haenszel W, Correa P . Cancer of the large intestine: epidemiologic findings. Dis Colon Rectum 1973; 16: 371–377.
Tamakoshi K, Wakai K, Kojima M, Watanabe Y, Hayakawa N, Toyoshima H, Yatsuya H, Kondo T, Tokudome S, Hashimoto S, Suzuki K, Ito Y, Tamakoshi A, JACC Study Group. A prospective study of body size and colon cancer mortality in Japan: The JACC Study. Int J Obes Relat Metab Disord 2004; 28: 551–558.
Nomura A, Heilbrun LK, Stemmermann GN . Body mass index as a predictor of cancer in men. J Natl Cancer Inst 1985; 74: 319–323.
Chyou PH, Nomura AM, Stemmermann GN . A prospective study of weight, body mass index and other anthropometric measurements in relation to site-specific cancers. Int J Cancer 1994; 57: 313–317.
Wolk A, Gridley G, Svensson M, Nyren O, McLaughlin JK, Fraumeni JF, Adam HO . A prospective study of obesity and cancer risk (Sweden). Cancer Causes Control 2001; 12: 13–21.
Moller H, Mellemgaard A, Lindvig K, Olsen JH . Obesity and cancer risk: a Danish record-linkage study. Eur J Cancer 1994; 30A: 344–350.
Hill MJ . Nutrition and human cancer. Ann NY Acad Sci 1997; 833: 68–78.
Giovannucci E . Insulin and colon cancer. Cancer Causes Control 1995; 6: 164–179.
Jones JI, Clemmons DR . Insulin-like growth factors and their binding proteins: biological actions. Endocr Rev 1995; 16: 3–34.
Renehan AG, Zwahlen M, Minder C, O'Dwyer ST, Shalet SM, Egger M . Insulin-like growth factor (IGF)-I, IGF binding protein-3, and cancer risk: systematic review and meta-regression analysis. Lancet 2004; 363: 1346–1353.
Thissen JP, Ketelslegers JM, Underwood LE . Nutritional regulation of the insulin-like growth factors. Endocr Rev 1994; 15: 80–101.
Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ . Overweight, obesity, and mortality from cancer in a prospectively studied cohort of US adults. N Engl J Med 2003; 348: 1625–1638.
Weiss NS . Can the ‘specificity’ of an association be rehabilitated as a basis for supporting a causal hypothesis? Epidemiology 2002; 13: 6–8.
Bradford Hill A . The environment and disease: association or causation? Proc Roy Soc Med 1965; 58: 295–300.
Batty GD, Lee IM . Physical activity and coronary heart disease. BMJ 2004; 328: 1089–1090.
Davey Smith G, Gunnell D, Ben-Shlomo Y . Life-course approaches to socio-economic differentials in cause-specific adult mortality. In: Leon D, Walt G (eds) Poverty, inequality and health. An international perspective 2001. OUP: Oxford.
Acknowledgements
We are grateful to the civil servants who gave of their time to take part in the baseline study. The original screening of participants in the Whitehall study was funded by the Department of Health and Social Security and the Tobacco Research Council. Martin Shipley and Elizabeth Breeze are supported by the British Heart Foundation; Michael Marmot by the UK Medical Research Council (MRC). When work on this report began, David Batty was financed by the UK MRC at the London School of Hygiene and Tropical Medicine and subsequently by a University Senior Research Fellowship at the University of Copenhagen. He is now the recipient of a Wellcome Advanced Training Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Batty, G., Shipley, M., Jarrett, R. et al. Obesity and overweight in relation to organ-specific cancer mortality in London (UK): findings from the original Whitehall study. Int J Obes 29, 1267–1274 (2005). https://doi.org/10.1038/sj.ijo.0803020
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803020
Keywords
This article is cited by
-
An increased body mass index is associated with a worse prognosis in patients administered BCG immunotherapy for T1 bladder cancer
World Journal of Urology (2019)
-
Obesity, Physical Activity and Bladder Cancer
Current Urology Reports (2015)
-
Relationship between body mass index and mortality among Europeans
European Journal of Clinical Nutrition (2012)
-
Recreational physical activity, body mass index, and survival in women with colorectal cancer
Cancer Causes & Control (2012)
-
Modifiable risk factors for prostate cancer mortality in London: forty years of follow-up in the Whitehall study
Cancer Causes & Control (2011)