Abstract
A significant number of pregnancies are lost in the first trimester and 1–2% are ectopic pregnancies (EPs). Early pregnancy loss in general can cause significant morbidity with bleeding or infection, while EPs are the leading cause of maternal mortality in the first trimester. Symptoms of pregnancy loss and EP are very similar (including pain and bleeding); however, these symptoms are also common in live normally sited pregnancies (LNSP). To date, no biomarkers have been identified to differentiate LNSP from pregnancies that will not progress beyond early gestation (non-viable or EPs), defined together as combined adverse outcomes (CAO). In this study, we present a novel machine learning pipeline to create prediction models that identify a composite biomarker to differentiate LNSP from CAO in symptomatic women. This prospective cohort study included 370 participants. A single blood sample was prospectively collected from participants on first emergency presentation prior to final clinical diagnosis of pregnancy outcome: LNSP, miscarriage, pregnancy of unknown location (PUL) or tubal EP (tEP). Miscarriage, PUL and tEP were grouped together into a CAO group. Human chorionic gonadotrophin β (β-hCG) and progesterone concentrations were measured in plasma. Serum samples were subjected to untargeted metabolomic profiling. The cohort was randomly split into train and validation data sets, with the train data set subjected to variable selection. Nine metabolite signals were identified as key discriminators of LNSP versus CAO. Random forest models were constructed using stable metabolite signals alone, or in combination with plasma hormone concentrations and demographic data. When comparing LNSP with CAO, a model with stable metabolite signals only demonstrated a modest predictive accuracy (0.68), which was comparable to a model of β-hCG and progesterone (0.71). The best model for LNSP prediction comprised stable metabolite signals and hormone concentrations (accuracy = 0.79). In conclusion, serum metabolite levels and biochemical markers from a single blood sample possess modest predictive utility in differentiating LNSP from CAO pregnancies upon first presentation, which is improved by variable selection and combination using machine learning. A diagnostic test to confirm LNSP and thus exclude pregnancies affecting maternal morbidity and potentially life-threatening outcomes would be invaluable in emergency situations.
Similar content being viewed by others
Introduction
Pelvic pain and vaginal bleeding are common in early pregnancy. Up to 30% of women will experience these symptoms during the first trimester1,2 and require a medical assessment and elimination of differential diagnosis that includes miscarriage, ectopic pregnancy (EP) or pregnancy of unknown location (PUL). Approximately 15% of known correctly sited pregnancies end in miscarriage and can cause significant morbidity if heavy bleeding or infection occurs3. A further 1–2% of pregnancies are located outside the endometrial cavity, with the majority (~ 98%) implanting in the fallopian tubes; these EPs are the leading cause of maternal mortality in the first trimester4,5. PUL describes a pregnancy that cannot be located on transvaginal ultrasonography (TVS); 5–42% of early pregnancy scans fall into this category6. Those pregnancies that are assigned as PULs at first emergency presentation have clinically heterogenous outcomes; their final diagnosis includes (i) a live normally-sited pregnancy (LNSP), (ii) a non-viable normally-sited pregnancy (NVNSP), (iii) an EP, (iv) a failed PUL or (v) a persistent PUL7. Considering the possible consequences of each pregnancy outcome, early and accurate identification upon first presentation of symptomatic women with a LNSP from any other pregnancy outcome will reduce the diagnostic burden, as well as maternal morbidity and mortality.
Diagnosis of an adverse early pregnancy outcome in clinical practice depends on a combination of ultrasonographic, biochemical, clinical, and sometimes surgical assessment. TVS at first presentation is inconclusive in approximately 4–40% of cases, likely due to (i) a LNSP before the detection limit of TVS, (ii) failure before gestation sac formation or (iii) being an EP8. The detection limit of a LNSP by TVS is ~ 6 weeks of gestation9, however, a significant proportion of pregnancies are unplanned10. Therefore, dating of the pregnancy from last menstrual period is frequently unreliable, which further complicates diagnosis at first emergency presentation. Human chorionic gonadotrophin β (β-hCG) and progesterone are used in routine clinical practice to monitor pregnancies not located on TVS; doubling of β-hCG over 48 h is indicative of a LNSP11, and TVS should be able to locate a LNSP with a serum β-hCG level ≥ 1500 IU/L12. An initial progesterone level cutoff of ≤ 2 nmol/L has been shown to accurately categorise low risk PULs13. However, serial β-hCG concentrations are required to accurately differentiate LNSPs from adverse outcomes8. Such management runs the risk of tubal rupture in women with EP, and unwarranted anxiety and clinical intervention in others with LNSP14. Since most symptomatic patients will have a LNSP, their rapid identification at first presentation and reassurance will allow focussing of the available diagnostic resources on women with a high risk of conditions that can be associated with maternal morbidity and mortality. A plethora of novel blood and urine biomarkers have been identified in the pursuit of improved diagnostics for miscarriage, EP and PUL7,8,15,16,17,18. However, none have yet been translated to routine clinical care. Few studies have assessed biomarkers for the definitive diagnosis of a LNSP in symptomatic cohorts containing multiple adverse outcomes. Therefore, there is an urgent need for a rapid, non-invasive, and single measurement test to accurately identify a LNSP, and exclude NVNSPs and EPs.
Metabolomics is the comprehensive analysis of small molecules (< 1500 Da) in biological samples such as cells, tissues and biofluids. In humans, metabolomic profiling can be used to identify disease biomarkers and elucidate metabolic pathways involved in pathological processes19. Nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry (MS) are the two most employed techniques in metabolomic research20,21. NMR metabolomic analysis provides highly reproducible, global quantitation of measurable analytes in biological samples in an unbiased and non-destructive fashion22. Routinely collected biofluids in early pregnancy assessment, such as blood and urine, are well-established sample types for NMR metabolomic profiling and are ideal for biomarker screening with simple and minimally invasive sample collection.
The analysis of omics data and its exploitation for in-silico biomarker discovery using machine learning is not trivial, with an ongoing crisis of reproducibility in the field23. We have applied a state-of-the-art pipeline to detect consensus, robust signals across patients. In this study, we propose a composite biomarker of early LNSP when compared with non-viable correctly sited pregnancies and EPs (these were grouped together as combined adverse outcomes, henceforth CAO). We apply a novel machine learning pipeline for robust biomarker identification and create models that integrate untargeted 1H NMR small molecule profiling of serum combined with biochemical markers (progesterone, β-hCG) and patient demographics.
Results
Study cohort
A total of 370 participants were recruited to this study. Of these participants, 31 were excluded before sample analysis (Supplementary Table 1); two were found to be non-pregnant, and two had twin pregnancies, which were excluded on the basis that they may confound metabolomic profiles of singleton pregnancies. Two participants had vanishing twin syndrome (dual outcome). It was decided to include only confirmed tEP within the ectopic group, therefore, a left ovarian EP and a caesarean section EP were excluded. Nine participants were excluded based on an unknown pregnancy outcome; these patients typically had a PUL or an early LNSP (a pregnancy within the uterine cavity which has a potential to develop normally)24; however, they had a termination of pregnancy before 12 weeks’ gestation or were lost to follow up. Finally, 14 participants were excluded due to missing clinicodemographic data including BMI, gestational age, and hormone levels (Supplementary Table 1). The remaining 339 participants were categorised into four pregnancy outcomes: LNSP, miscarriage, PUL and tEP (Fig. 1). Following serum spectra acquisition and quality control, 18 spectra that did not meet the minimum reporting standards were removed from further analysis. Furthermore, three spectra were found to contain EDTA, suggesting that plasma had been erroneously collected before serum during serial blood specimen acquisition and thus were excluded. Finally, one sample contained a high level of gluconic acid and was removed from further analysis (Supplementary Table 1). In total, 146 LNSPs, 77 miscarriages, 42 PULs and 51 tEPs were included in this study. Participant demographic information is summarised in Table 1. There were no significant differences in known confounding variables in metabolomics research between groups, including BMI and smoking status.
Plasma β-hCG and progesterone concentrations across early pregnancy outcomes
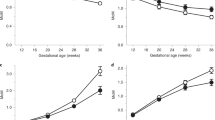
Plasma concentrations of β-hCG and progesterone were significantly higher in the LNSP group compared with the adverse outcomes of miscarriage, PUL and tEP (Table 1). Gestational age demonstrated a highly significant positive correlation (p < 0.001) with plasma β-hCG concentrations in the LNSP group, and weak positive correlations in the miscarriage and PUL groups. Progesterone concentration did not significantly correlate with either gestational age or β-hCG in any group (Supplementary Table 2).
Metabolite identification
Proton spectra of serum were divided into 162 bins, of which 92 (56.8%) were assigned to a metabolite. A total of 32 unique metabolites were annotated, which included amino acids (e.g., phenylalanine, valine, leucine, isoleucine), organic acids (e.g., lactate, formate) and saccharides (e.g., glucose) (Fig. 2, Table 2). Metabolites identified using two independent, orthogonal datasets were allocated MSI level 1 assignment in accordance with best practice25,26 (Table 2). To remove redundant peaks arising from the same molecule, correlations were calculated between all signals originating from individual metabolites. The highest correlating peaks were then selected as most representative of a given metabolite and taken forward for statistical analysis (Table 2). The refined data set contained 102 bins in total, comprising annotated metabolites (32 bins) and unannotated signals (70 bins).
Metabolite abundances across early pregnancy outcomes
The cohort was randomly split into discovery (80%, n = 253) and validation (20%, n = 63) sample sets (Fig. 3). Univariate analysis of the discovery set revealed 21 metabolite signals as significantly different in the LNSP group when compared with miscarriage, PUL or tEP groups (Supplementary Table 3). When miscarriage, PUL and tEP outcomes were grouped together (CAO) and compared with LNSP (Supplementary Table 4), there were 15 metabolite signals with significantly different abundances (Supplementary Table 5). Four group and two group comparisons showed 11 and five unique metabolite signals, respectively, with an overlap of 12 signals. Unsupervised PCA using plasma hormones and significant metabolite signals from univariate analysis did not demonstrate group separation (Supplementary Fig. 2).
Selection of stable metabolite signals for biomarker identification
The pipeline for variable selection described in the methods and Fig. 3 identified nine metabolite signals as key to discriminate LNSP from CAO using a composite marker approach: acetate, alanine, arginine, glutamate, glutamine, phenylalanine, and unlabelled signals 30, 54 and 130 (Fig. 4A). The median abundances of glutamate, phenylalanine, and unlabelled signals 30, 54 and 130 were higher in LNSP compared with CAO. Acetate, alanine, arginine, and glutamine levels were higher in the CAO group. When visualised by PCA, a discrete structure and subtle separation of groups by stable metabolite signals was apparent (Fig. 4B). Following the assessment of variance undertaken to choose the univariate analyses covariates, we also tested a model that would incorporate additional clinicodemographic variables including β-hCG, progesterone, age, BMI, and gestational age. When comparing LNSP to CAO, more clear group structure was observed in the PCA score plot (Fig. 4C). Further investigations tested signals able to discriminate between all pregnancy outcomes, but model performances were just slightly better than random (Supplementary Fig. 3A and 3B). Addition of covariates to hormone concentrations and selected metabolite signals did not significantly improved discrimination of LNSP, miscarriage, PUL and tEP groups by PCA (Supplementary Fig. 3C).
Differential abundances of biochemical markers and serum metabolites in live normally sited pregnancy (LNSP) and combined adverse outcomes (COA). (a) Boxplots of β-hCG, progesterone, acetate, alanine, arginine, glutamate, glutamine, phenylalanine, and unlabelled 30, 54 and 130 in LNSP and COA groups. (b) Principal component analysis of pregnancy outcomes using selected metabolites alone or (c) selected metabolites, β-hCG, progesterone, participant age, gestational age, and BMI.
Performance of machine learning models to predict pregnancy outcome
Random forest models were constructed for the prediction of pregnancy outcome using stable metabolite signals only or in combination with plasma hormones and clinicodemographic variables. Models were generated using the discovery cohort and performance was assessed using the independent validation cohort, which was not used for stable metabolite identification, thus preventing data leakage. Model performance was poor when all four pregnancy outcomes were included, regardless of variable inclusion (accuracy 0.48–0.58, Supplementary Table 6). When differentiating LNSP from CAO, a model with stable metabolite signals only demonstrated a modest predictive accuracy (0.68), which was comparable to a model of β-hCG and progesterone (0.71) (Table 3). The predictive accuracy of β-hCG and progesterone was marginally improved with the addition of clinicodemographic variables (accuracy = 0.73). Combining stable metabolites with hormones produced a superior model (accuracy = 0.79), while the addition of clinicodemographic variables did not improve the model further (Table 3). Clinicodemographic variables alone were unable to differentiate LNSP from CAO (accuracy = 0.44).
Discussion
We report the development of predictive models that can discriminate LNSP from multiple adverse outcomes (CAO group) at first presentation using combinations of hormone levels, metabolites and clinicodemographic features. These findings are important, since they allow for early identification of a LNSP in symptomatic women upon initial presentation to an EPAU and grants their accurate stratification for further investigations. Our study design was pragmatic to demonstrate the utility of our method in real world situations. Importantly, this study has generated a composite biomarker of early LNSP when compared with adverse pregnancy outcomes (miscarriage, PUL and tEP) using a novel machine learning pipeline. This approach has overcome the deficiencies in previous studies regarding reproducibility, while integrating untargeted 1H NMR small molecule profiling of serum with biochemical markers (β-hCG and progesterone) and patient demographics.
Previously examined cohorts are unlikely to be translatable to emergency clinical situations, yet a diagnostic biomarker in these scenarios would require withstanding demographic and behavioural heterogeneity (e.g., diet, exercise habits) amongst patients. Our study collected samples from symptomatic patients prior to medical assessment, including TVS, thus our data is applicable for all settings, including where access to TVS is not immediately available. In this regard, our patient population and the analytic approach are realistic and opportunistic, thus, make this data to be generalisable. Our methodology is unique in that it incorporates testing within the cohort to ensure reproducibility, whilst avoiding overfitting in the machine learning pipeline. The result is a selection of consensus variables with robust biomarker potential that should be further validated in a larger study.
Previous studies have shown that a single measurement of β-hCG cannot discriminate a LNSP from EP27,28. The ‘M4’ logistic regression model, which uses both the initial β-hCG concentration and β-hCG ratio to categorise PULs into low or high-risk categories29, performed well in the prediction of viable pregnancies as the final outcome from PULs7. However, its utility in a cohort of symptomatic women including miscarriage has not yet been tested. Inclusion of initial serum progesterone level in the M4 model (M6p model) has been shown to improve the discrimination of EPs from failed PULs and LNSPs in a PUL cohort13. In agreement, meta-analysis has concluded that a low progesterone concentration (11–21 nmol/L) can exclude a LNSP with high accuracy but cannot separate EP and miscarriage cases30. Furthermore, previous studies have included participants beyond 10 weeks’ gestation, at which point progesterone levels are rising in viable pregnancies and likely exhibit a larger difference compared with non-viable gestations. In this study, a considerable number of participants in the LNSP group exhibited a low progesterone level, which may be due to the transient, physiological decline in progesterone between gestational weeks six to eight, corresponding to the luteal-placental shift31. Addition of β-hCG and progesterone to selected metabolites improved the accuracy of the prediction model. Addition of the demographic features that are known to be associated with adverse pregnancy outcomes did not improve model performance for identifying LNSPs.
We have identified perturbations in the serum metabolome pertinent to pregnancy outcome using 1H NMR profiling. Maternal circulatory concentrations of most essential and non-essential amino acids decrease during normal gestation, hypothetically due to placental transfer to the foetus and pregnancy adaptation for protein conservation32. Alanine, arginine, and glutamine levels were significantly lower in the LNSP group compared with CAO, which may reflect perturbed foetal utilisation of amino acids and thus the non-viable status of miscarriage and tEP. Furthermore, increased levels of glutamine metabolites in the endometrium have been associated with recurrent miscarriage33. Some amino acids, including valine, leucine, phenylalanine and glutamate have been shown to increase in early pregnancy relative to non-pregnant women34. Glutamate and phenylalanine levels were significantly higher in LNSP compared with CAO, thus reflecting the abnormal progression of failed LNSPs and ectopically located pregnancies. Therefore, maternal amino acid levels may represent promising biomarkers to differentiate a normal pregnancy from adverse outcomes. Importantly, serum concentrations of these metabolites can be easily measured in a clinical setting, allowing for the development of predictive algorithms that could combine biochemical markers and demographic variables at the bedside.
A study by Turkoglu et al.35 identified eight plasma metabolites as significantly perturbed in tEP compared with LNSP controls including acetate, lactate, and glucose. Acetate levels were increased in tEP, which agrees with our study findings, however, the remaining seven metabolites were not significantly different in our cohort. Three unlabelled metabolite signals were found to be stable markers differentiating LNSP from CAO. Unlabelled signals were those that could not be annotated from a metabolite library. It may be possible to identify these metabolites using multidimensional NMR spectroscopy or MS methods36,37.
Blood samples included in this study were collected from a single sex cohort with a relatively small age range, thus were expected to demonstrate any striking differences in the serum metabolome relevant to pregnancy outcome. However, these samples were subject to several metabolomic confounders including age, fasting status, medication intake, symptomology, obesity, and smoking status. BMI and smoking have been shown to induce confounding effects on the serum metabolome38. Age and BMI were found to contribute substantially to the observed variance in metabolite abundance between pregnancy outcomes. Several risk factors have been identified for EP, such as smoking, tubal surgery, previous EP, and sexually transmitted infections39,40. Risk factors for miscarriage include advanced maternal age, previous miscarriage and low or high BMI41,42,43. We did not observe differences in participant age, BMI, or smoking status between groups. Accordingly, the addition of maternal age and BMI as covariates had minimal impact on predictive accuracy in machine learning models. We excluded twin pregnancies from our LNSP group, yet any one of the CAOs may have been an undiagnosed twin pregnancy. The influence of undiagnosed twin pregnancies on the metabolomic profile would be extremely challenging to unpick, however, we would assume the impact to be very small due to the low twinning rate in our population.
In summary, this study identifies a metabolite profile associated with LNSP and incorporates this in to routinely collected hormone levels and clinicodemographic data providing predictive models that can discriminate LNSP from multiple adverse early pregnancy outcomes with reasonable accuracy. These findings may be useful in the development of a future diagnostic test to confirm a LNSP in symptomatic women and thus, exclude pregnancy loss and potentially life-threatening early pregnancy outcomes. Larger studies with independent cohorts are required to validate the accuracy, translatability and clinical utility of the predictive models described herein.
Methods
Study group
A total of 370 participants were recruited to the current study. These women presented as emergency clinical attendance to the Early Pregnancy Assessment Unit (EPAU) at Liverpool Women’s Hospital with symptoms of EP (abdominal pain and/or vaginal bleeding). Therefore, at this first emergency presentation, all eligible patients were symptomatic and pregnant, were ≤ 10 weeks of gestation by their last menstruation dates, without a confirmed location of pregnancy and without known final pregnancy outcome. All eligible participants self-reported to the EPAU and were triaged upon arrival. Pregnancy was confirmed with a urinary pregnancy test. Women were provided with an information leaflet, and informed written consent was obtained. Inclusion criteria comprised pregnant women aged ≥ 18 years, presenting with abdominal pain and/or bleeding at ≤ 10 weeks of gestation (calculated from last menstrual period). Women who did not meet the inclusion criteria were excluded. Demographic data were collected at the time of consent, including age and body-mass index (BMI), as well as detailed information concerning smoking status, alcohol intake, and dietary and exercise preferences. A full medical, surgical, medication and gynaecological history was also collected.
Clinical outcomes
The final pregnancy outcomes were allocated at 12 weeks of gestation and the data were retrieved from hospital information software systems including PENS™ (Royal Liverpool University Hospitals NHS Trust, Liverpool, UK), MEDITECH™ (Westwood, MA, USA) and IDEAS™ (Mellowood Medical, Toronto, Canada) and subsequently collated. Where applicable, serial β-hCG measurements and TVS were also reviewed using Sunquest ICE™ (Sunquest Information Systems, Tucson, AZ, USA) and Picture Archiving and Communication System (PACS™) (©Carestream Health Inc, 2023, USA), respectively. Outcomes were classified in accordance with terminology guidance from the European Society of Human Reproduction and Embryology (ESHRE)24,44. Patients were divided into four groups: (i) LNSP; (ii) EP; (iii) miscarriage; and (iv) PUL. LNSP was defined as a pregnancy inside the uterine cavity with evident foetal heart pulsations. EP was defined as a pregnancy located outside of the uterine cavity, diagnosed either surgically at laparoscopy or on TVS. EPs were classified in line with ESHRE guidance24. The term miscarriage was used to describe loss of a normally sited (within the uterine cavity) pregnancy24. Miscarriage was diagnosed using guidance from the National Institute for Health and Care Excellence (NICE) on EP and miscarriage: diagnosis and initial management45. Diagnostic criteria included a mean gestational sac diameter (GSD) ≥ 25 mm with no obvious yolk sac or foetal pole on two TVS a minimum of seven days apart, or a crown-rump length (CRL) ≥ 7 mm without foetal heart pulsations on two TVS a minimum of seven days apart. Miscarriage was also diagnosed in the presence of heavy vaginal bleeding with an open cervical os, with or without products of conception. A PUL was defined as a pregnancy that had not been localised on TVS and was either treated with methotrexate or resolved spontaneously.
Sample collection
All biosamples were processed within 1 h of collection. Blood was collected into uncoated S-Monovette® Z-Gel tubes and S-Monovette® EDTA KE tubes (Sarstedt, Leicester, UK) for serum and plasma isolation, respectively. Blood in Z-Gel tubes was allowed to clot for ≥ 20 min before centrifuging at 1600×g for 10 min at 4 °C. EDTA KE tubes were processed immediately upon receipt; samples were centrifuged at 1600×g for 10 min at 4 °C. 1 mL aliquots of serum and plasma were stored in sterile cryovials at − 80 °C.
Sample preparation
Serum aliquots were thawed and 300 μL of serum was diluted to a final volume containing 50% [v/v] serum, 40% [v/v] dd 1H2O (18.2 MΩ), 10% (v/v) 1 M PO43− pH 7.4 buffer (Na2HPO4, VWR International Ltd., Radnor, Pennsylvania, USA and NaH2PO4, Sigma-Aldrich, Gillingham, UK) in deuterium oxide (2H2O, Sigma-Aldrich) and 1.2 mM sodium azide (NaN3, Sigma-Aldrich). Samples were vortexed for 1 min, centrifuged at 21,500×g at 4 °C for 5 min and 600 μL transferred into 5 mm outer diameter NMR tubes.
Spectral acquisition
Non-targeted 1D 1H NMR spectra were acquired at 37 °C using a 700 MHz Bruker Advance III spectrometer equipped with a TCI cryoprobe and chilled Sample-Jet autosampler (Bruker). 1D 1H NMR standard experiment with the cpmgpr1d filters for selective observation of low molecular weight components with optimal water suppression was acquired. Pulse sequence was vendor supplied using Carr-Purcell-Meiboom-Gill (CPMG) sequence. Serum spectra were acquired with 32 transients at 17 ppm spectral width, 72 K complex points, 3 ms echo time, 3.1 s acquisition time and a 4 s interscan delay. Full 1H spectrum parameter sets are available with the data deposited at MetaboLights public repository (MTBLS6219)46.
Spectral processing
Automated Fourier transformation and phasing were performed in Topspin version 3.2. All spectra were individually analysed to ensure conformity with the recommended minimum reporting standards set out by the Metabolites Standard Initiative (MSI)25,26. Serum spectra were aligned to glucose anomeric peak at 5.244 ppm. Overall peak shapes were appraised, and average full width half maximum (FWHM) was calculated for the alignment peak; spectra with a FWHM value > 2 standard deviations from the mean were either repeated or excluded (Supplementary Table 1). No baseline corrections were applied.
Metabolite annotation
Serum spectra were annotated using Chenomx NMR Suite 8.2 (332-mammalian metabolite library, Chenomx Inc., Edmonton, AB, Canada). Where possible, the identities of the annotated metabolites were confirmed by comparison to an in-house metabolite library in accordance with the MSI best practice. Metabolite identities were allocated to levels 1–3 of assignment confidence; level 1 required 1H information complemented with a secondary orthologous method (1H–13C heteronuclear single quantum coherence (HSQC), 1H–1H J-resolved, 1H–1H– correlation spectroscopy (COSY) and/or total correlation spectroscopy (TOCSY)); level 2 required matching 1D–1H NMR spectrum of an in-house metabolite library or external libraries available in Chenomx to the experimental spectra; level 3 comprised putatively characterised compound classes. Spectra were integrated into 162 bins with 89 annotated, corresponding to 32 unique metabolites. Annotated spectra were integrated to data matrices of peak intensities for statistical analysis. Negative intensity values, which are common in binned data47, were replaced with 1/5 of the minimum positive values of their corresponding variables. In order to select the most representative bin for metabolites giving rise to multiple signals, an in-house correlation reliability score (CRS) metric was utilised48. Correlations scores were calculated for each unique metabolite giving rise to multiple peaks, and the median score minus the standard deviation across all metabolites was set as the passing score (56.6%). When no peaks passed the CRS threshold for a given metabolite, all were included. However, such peaks are likely in overlapped regions of the spectra, therefore, metabolite annotation is of lower confidence. Spectral data is available with annotation via the MetaboLights repository (MTBLS6219)46.
Progesterone and β-hCG testing
Progesterone and β-hCG concentrations were measured at the Royal Liverpool University Hospital accredited biochemistry laboratory. Plasma progesterone and β-hCG were analysed with the Elecsys Progesterone III assay and Elecsys HCG + β assay (Roche), respectively. Assays were performed on a cobas e 801 analyser (Roche) according to the manufacturer’s instructions. Assay coefficients of variability were as follows: progesterone 3 pmol/L = 10.9%, 35 pmol/L = 7.9%, 67 pmol/L = 8.1%. β-hCG: 2.9 mIU/L = 4.9%, 25 mIU/L = 5.9%, 497 mIU/L = 6.8%.
Statistical analysis and model building
Participant age, ethnicity, BMI, smoking status, parity, gestational age, and plasma concentrations of β-hCG and progesterone were compared. Normality testing concluded that these variables did not follow a Gaussian distribution across all groups, therefore, non-parametric methods were employed. The Kruskal–Wallis test with Dunn’s multiple comparison post hoc test was used to investigate continuous variables. For categorical variables, a Chi-squared test was used. All analyses were conducted in GraphPad Prism (version 5.0). Spearman’s Rank correlation coefficients were performed in Origin Pro (version 2021b 9.85).
Metabolomics data was exploited using a univariate and multivariate statistical approach, with analyses conducted in R (version 4.1.249, using in-house scripts. Data were normalised via probabilistic quotient normalisation (PQN)50 and log2 transformed. Technical variation/batch was assessed and removed using ComBat from the package sva51. This analysis can be consulted via the GitHub repository (https://github.com/EvaCaamano/ExPeDiTe_publication/tree/v1.0.0)52. Briefly, metabolite data was split in 80/20% train/validation sets. The train set was used to undergo variable selection firstly by a univariate statistical approach. The package limma53 was used to generate linear mixed models to find metabolite signals different between the groups of study considering relevant metadata (BMI, age and gestation—these covariates were chosen as they explained a significant proportion of variance of the data—see Supplementary Fig. 1) with significance adjusted for false discovery rate (FDR) using Benjamini and Hochberg54. Significant signals at 5% FDR were taken forward for multivariate selection. The train dataset was subjected to a tenfold cross-validation in a 90/10% split, where each 90% split underwent 100 rounds of Least Absolute Shrinkage and Selection Operator (LASSO, glmnet package55) selection. Signals that were selected in at least 80% of the rounds and more than 8 folds were taken forward for modelling. Random forest models56 were constructed with selected metabolites and metadata variables and performances were assessed in both train and validation datasets. Further tests were done by building generalised linear models and calculating area under the receiver operator characteristic curves (AUROCs).
Data visualisation was performed with Origin Pro and R package ggplot257. Significant p values are highlighted with asterisks (p < 0.05*, p < 0.01**, p < 0.001***).
Ethical approval
This research study has been approved by Liverpool Women’s Hospital Research & Development Team and the North West Liverpool Central Research Ethics Committee (REC 17/NW/0646).
Data availability
NMR data is available via the EMBL-EBI MetaboLights repository (ID: MTBLS6219). Code and analyses for all main results are available via GitHub repository link https://github.com/EvaCaamano/ExPeDiTe_publication/tree/v1.0.052.
Abbreviations
- BMI:
-
Body mass index
- CI:
-
Confidence interval
- CAO:
-
Combined adverse outcomes
- CRS:
-
Correlation reliability score
- CRL:
-
Crown rump length
- EP:
-
Ectopic pregnancy
- FDR:
-
False discovery rate
- GSD:
-
Gestational sack diameter
- β-hCG:
-
Human chorionic gonadotrophin β
- IQR:
-
Interquartile range
- LNSP:
-
Live normally sited pregnancy
- NVNSP:
-
Non-viable normally sited pregnancy
- NMR:
-
Nuclear magnetic resonance
- PUL:
-
Pregnancy of unknown location
- PPV:
-
Positive predictive value
- POC:
-
Products of conception
- PCA:
-
Principal component analysis
- P4:
-
Progesterone
- TVS:
-
Transvaginal ultrasound
- tEP:
-
Tubal ectopic pregnancy
References
Everett, C. Incidence and outcome of bleeding before the 20th week of pregnancy: Prospective study from general practice. BMJ 315, 32–34. https://doi.org/10.1136/bmj.315.7099.32 (1997).
Hasan, R. et al. Patterns and predictors of vaginal bleeding in the first trimester of pregnancy. Ann. Epidemiol. 20, 524–531. https://doi.org/10.1016/j.annepidem.2010.02.006 (2010).
Quenby, S. et al. Miscarriage matters: The epidemiological, physical, psychological, and economic costs of early pregnancy loss. Lancet 397, 1658–1667. https://doi.org/10.1016/s0140-6736(21)00682-6 (2021).
Aljassim, F. et al. Exploring the presence of markers of decidualisation in the fallopian tubes: A systematic review. Biol. Reprod. https://doi.org/10.1093/biolre/ioad062 (2023).
Marion, L. L. & Meeks, G. R. Ectopic pregnancy: History, incidence, epidemiology, and risk factors. Clin. Obstet. Gynecol. 55, 376–386. https://doi.org/10.1097/GRF.0b013e3182516d7b (2012).
Kirk, E., Bottomley, C. & Bourne, T. Diagnosing ectopic pregnancy and current concepts in the management of pregnancy of unknown location. Hum. Reprod. Update 20, 250–261. https://doi.org/10.1093/humupd/dmt047 (2014).
Bobdiwala, S. et al. Diagnostic protocols for the management of pregnancy of unknown location: A systematic review and meta-analysis. BJOG 126, 190–198. https://doi.org/10.1111/1471-0528.15442 (2019).
Senapati, S. & Barnhart, K. T. Biomarkers for ectopic pregnancy and pregnancy of unknown location. Fertil. Steril. 99, 1107–1116. https://doi.org/10.1016/j.fertnstert.2012.11.038 (2013).
Murugan, V. A., Murphy, B. O., Dupuis, C., Goldstein, A. & Kim, Y. H. Role of ultrasound in the evaluation of first-trimester pregnancies in the acute setting. Ultrasonography 39(2), 178–189. https://doi.org/10.14366/usg.19043 (2020).
Enthoven, C. A. et al. Clustering of characteristics associated with unplanned pregnancies: The generation R study. BMC Public Health 22, 1957. https://doi.org/10.1186/s12889-022-14342-y (2022).
Lenton, E. A., Neal, L. M. & Sulaiman, R. Plasma concentrations of human chorionic gonadotropin from the time of implantation until the second week of pregnancy. Fertil. Steril. 37, 773–778. https://doi.org/10.1016/s0015-0282(16)46337-5 (1982).
Visconti, K. & Zite, N. hCG in ectopic pregnancy. Clin. Obstet. Gynecol. 55, 410–417. https://doi.org/10.1097/GRF.0b013e31825109dc (2012).
Van Calster, B. et al. Managing pregnancy of unknown location based on initial serum progesterone and serial serum hCG levels: Development and validation of a two-step triage protocol. Ultrasound Obstet. Gynecol. 48, 642–649. https://doi.org/10.1002/uog.15864 (2016).
National Guideline Alliance (UK). Progestogens for preventing miscarriage: Ectopic pregnancy and miscarriage: diagnosis and initial management: Evidence review C (National Institute for Health and Care Excellence (NICE), London, 2021)
Tsoutsouki, J., Patel, B., Comninos, A. N., Dhillo, W. S. & Abbara, A. Kisspeptin in the prediction of pregnancy complications. Front. Endocrinol. 13, 942664. https://doi.org/10.3389/fendo.2022.942664 (2022).
Tong, S., Skubisz, M. M. & Horne, A. W. Molecular diagnostics and therapeutics for ectopic pregnancy. Mol. Hum. Reprod. 21, 126–135. https://doi.org/10.1093/molehr/gau084 (2015).
Pillai, R. N., Konje, J. C., Tincello, D. G. & Potdar, N. Role of serum biomarkers in the prediction of outcome in women with threatened miscarriage: A systematic review and diagnostic accuracy meta-analysis. Hum. Reprod. Update 22, 228–239. https://doi.org/10.1093/humupd/dmv054 (2016).
Zhao, Z. et al. Circulating microRNA miR-323-3p as a biomarker of ectopic pregnancy. Clin. Chem. 58, 896–905. https://doi.org/10.1373/clinchem.2011.179283 (2012).
Zhang, A. H., Sun, H. & Wang, X. J. Serum metabolomics as a novel diagnostic approach for disease: A systematic review. Anal. Bioanal. Chem. 404, 1239–1245. https://doi.org/10.1007/s00216-012-6117-1 (2012).
Amberg, A. et al. NMR and MS methods for metabolomics. Methods Mol. Biol. 1641, 229–258. https://doi.org/10.1007/978-1-4939-7172-5_13 (2017).
Johnson, C. H., Ivanisevic, J. & Siuzdak, G. Metabolomics: Beyond biomarkers and towards mechanisms. Nat. Rev. Mol. Cell Biol. 17, 451–459. https://doi.org/10.1038/nrm.2016.25 (2016).
Wishart, D. S. NMR metabolomics: A look ahead. J. Magn. Reson. 306, 155–161. https://doi.org/10.1016/j.jmr.2019.07.013 (2019).
Kapoor, S. & Narayanan, A. Leakage and the reproducibility crisis in machine-learning-based science. Patterns 4, 100804. https://doi.org/10.1016/j.patter.2023.100804 (2023).
Kirk, E. et al. Terminology for describing normally sited and ectopic pregnancies on ultrasound: ESHRE recommendations for good practice. Hum. Reprod. Open 2020, hoaa055. https://doi.org/10.1093/hropen/hoaa055 (2020).
Considine, E. C. & Salek, R. M. A tool to encourage minimum reporting guideline uptake for data analysis in metabolomics. Metabolites https://doi.org/10.3390/metabo9030043 (2019).
Sumner, L. W. et al. Proposed minimum reporting standards for chemical analysis chemical analysis working group (CAWG) metabolomics standards initiative (MSI). Metabolomics 3, 211–221. https://doi.org/10.1007/s11306-007-0082-2 (2007).
Kaplan, B. C. et al. Ectopic pregnancy: Prospective study with improved diagnostic accuracy. Ann. Emerg. Med. 28, 10–17. https://doi.org/10.1016/s0196-0644(96)70131-2 (1996).
Kohn, M. A. et al. Beta-human chorionic gonadotropin levels and the likelihood of ectopic pregnancy in emergency department patients with abdominal pain or vaginal bleeding. Acad. Emerg. Med. 10, 119–126. https://doi.org/10.1111/j.1553-2712.2003.tb00028.x (2003).
Bobdiwala, S. et al. The clinical performance of the M4 decision support model to triage women with a pregnancy of unknown location as at low or high risk of complications. Hum. Reprod. 31, 1425–1435. https://doi.org/10.1093/humrep/dew105 (2016).
Verhaegen, J. et al. Accuracy of single progesterone test to predict early pregnancy outcome in women with pain or bleeding: Meta-analysis of cohort studies. BMJ 345, e6077. https://doi.org/10.1136/bmj.e6077 (2012).
Ku, C. W. et al. Gestational age-specific normative values and determinants of serum progesterone through the first trimester of pregnancy. Sci. Rep. 11, 4161. https://doi.org/10.1038/s41598-021-83805-w (2021).
Elango, R. & Ball, R. O. Protein and amino acid requirements during pregnancy. Adv. Nutr. 7, 839s–844s. https://doi.org/10.3945/an.115.011817 (2016).
Hong, L. et al. Multi-omics reveals a relationship between endometrial amino acid metabolism and autophagy in women with recurrent miscarriage†. Biol. Reprod. 105, 393–402. https://doi.org/10.1093/biolre/ioab101 (2021).
Di Giulio, A. M. et al. Plasma amino acid concentrations throughout normal pregnancy and early stages of intrauterine growth restricted pregnancy. J. Matern. Fetal Neonatal Med. 15, 356–362. https://doi.org/10.1080/14767050410001725578 (2004).
Turkoglu, O. et al. Metabolomic identification of novel diagnostic biomarkers in ectopic pregnancy. Metabolomics 15, 143. https://doi.org/10.1007/s11306-019-1607-1 (2019).
Dona, A. C. et al. A guide to the identification of metabolites in NMR-based metabonomics/metabolomics experiments. Comput. Struct. Biotechnol. J. 14, 135–153. https://doi.org/10.1016/j.csbj.2016.02.005 (2016).
Alseekh, S. et al. Mass spectrometry-based metabolomics: A guide for annotation, quantification and best reporting practices. Nat. Methods 18, 747–756. https://doi.org/10.1038/s41592-021-01197-1 (2021).
Chen, C. et al. Altered metabolite levels and correlations in patients with colorectal cancer and polyps detected using seemingly unrelated regression analysis. Metabolomics https://doi.org/10.1007/s11306-017-1265-0 (2017).
Karaer, A., Avsar, F. A. & Batioglu, S. Risk factors for ectopic pregnancy: A case-control study. Aust. N. Z. J. Obstet. Gynaecol. 46, 521–527. https://doi.org/10.1111/j.1479-828X.2006.00653.x (2006).
Li, C. et al. Risk factors for ectopic pregnancy: A multi-center case-control study. BMC Pregnancy Childbirth 15, 187. https://doi.org/10.1186/s12884-015-0613-1 (2015).
Veleva, Z. et al. High and low BMI increase the risk of miscarriage after IVF/ICSI and FET. Hum. Reprod. 23, 878–884. https://doi.org/10.1093/humrep/den017 (2008).
de la Rochebrochard, E. & Thonneau, P. Paternal age and maternal age are risk factors for miscarriage: Results of a multicentre European study. Hum. Reprod. 17, 1649–1656. https://doi.org/10.1093/humrep/17.6.1649 (2002).
Maconochie, N., Doyle, P., Prior, S. & Simmons, R. Risk factors for first trimester miscarriage–results from a UK-population-based case-control study. BJOG 114, 170–186. https://doi.org/10.1111/j.1471-0528.2006.01193.x (2007).
Kolte, A. M. et al. Terminology for pregnancy loss prior to viability: A consensus statement from the ESHRE early pregnancy special interest group. Hum. Reprod. 30, 495–498. https://doi.org/10.1093/humrep/deu299 (2015).
National Guideline, A. in Ectopic Pregnancy and Miscarriage: Diagnosis and Initial Management (National Institute for Health and Care Excellence (UK). Copyright © NICE 2019, 2019).
Haug, K. et al. MetaboLights—An open-access general-purpose repository for metabolomics studies and associated meta-data. Nucleic Acids Res. 41, D781-786. https://doi.org/10.1093/nar/gks1004 (2013).
Klein, M. S. Affine transformation of negative values for NMR metabolomics using the mrbin R package. J. Proteome Res. 20, 1397–1404. https://doi.org/10.1021/acs.jproteome.0c00684 (2021).
Grosman, R. NMR Metabolic Profiling of Mosquito Species to Understand Insecticide Resistance (The University of Liverpool, 2019).
RCoreTeam. R: A language and environment for statistical computing. R Foundation for Statistical Computing (2021).
Dieterle, F., Ross, A., Schlotterbeck, G. & Senn, H. Probabilistic quotient normalization as robust method to account for dilution of complex biological mixtures. Application in 1H NMR metabonomics. Anal. Chem. 78, 4281–4290. https://doi.org/10.1021/ac051632c (2006).
Johnson, W. E., Li, C. & Rabinovic, A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics 8, 118–127. https://doi.org/10.1093/biostatistics/kxj037 (2007).
Gutiérrez, E. C. EvaCaamano/ExPeDiTe_publication: ExPeDiTE_DOI_Generated (v1.0.0). Zenodo, https://doi.org/10.5281/zenodo.10950303 (2024).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47. https://doi.org/10.1093/nar/gkv007 (2015).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 57, 289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x (1995).
Friedman, J., Hastie, T. & Tibshirani, R. Regularization paths for generalized linear models via coordinate descent. J. Stat. Softw. 33, 1–22 (2010).
Liaw, A. & Wiener, M. Classification and regression by RandomForest. Forest 23, 18–22 (2001).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer-Verlag, 2016).
Acknowledgements
We acknowledge the women who attended Liverpool Women’s NHS Foundation Trust and donated samples for research. We thank the research nurses for participant recruitment and sample collection, and technical staff Helen Cox, Samantha Williams, and Alan Carter for sample processing. The authors are grateful to Angela Lambert for plasma β-hCG and progesterone analysis. We thank Liverpool Shared Research Facilities for the support to complete statistical analyses. This work was funded by a Wellbeing of Women project grant (RG2137). C.J.H. is supported by the Wellbeing of Women (RG2137), SRI/Bayer and Wellcome Trust IFFS3. N.T. is supported by an Academic Clinical Lectureship from the National Institute of Health Research (NIHR). P.B. was supported by an NIHR Academic Clinical Fellowship. P.J.D. was supported by a Clinical Research Fellowship from the Liverpool Women’s Hospital NHS Foundation Trust. D.K.H. is supported by the Wellbeing of Women (RG2137) and A.M. and D.K.H. are supported by an MRC Clinical Research Training Fellowship (MR/V007238/1).
Author information
Authors and Affiliations
Contributions
D.K.H., M.M.P & A.W.H. conceived the study and secured funding. D.K.H., N.T., A.M. & J.A.D. acquired ethical approval. P.J.D., P.B. & A.M. collated participant demographics, medical histories, and clinical outcomes. C.J.H. & M.M.P. collected and processed NMR data. C.J.H. & A.S.D. collected and analysed plasma protein abundances. E.C.G. designed the statistical pipeline and built the relevant software. C.J.H., E.C.G. & M.M.P. performed statistical analyses. C.J.H. & E.C.G prepared figures. C.J.H., E.C.G. & D.K.H. wrote the first draft of the manuscript. All authors revised the manuscript critically and approved the final version.
Corresponding author
Ethics declarations
Competing interests
A.W.H. has received honoraria for consultancy for Ferring, Roche, Nordic Pharma, Gesynta, Joii and Abbvie. A.W.H. has received payment for presentations from Theramex and Gideon Richter. The remaining authors have no competing interests to report.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hill, C.J., Phelan, M.M., Dutton, P.J. et al. Diagnostic utility of clinicodemographic, biochemical and metabolite variables to identify viable pregnancies in a symptomatic cohort during early gestation. Sci Rep 14, 11172 (2024). https://doi.org/10.1038/s41598-024-61690-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-61690-3
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.