Abstract
Transgene-free plant genome editing in the T0 generation is highly desirable but challenging1,2. Here we achieved such a goal using a co-editing strategy via Agrobacterium-mediated transient expression of cytosine base editor to edit ALS encoding acetolactate synthase to confer herbicide chlorsulfuron resistance as a selection marker, Cas12a/CRISPR RNA for editing gene(s) of interest, and green fluorescent protein for selecting transgene-free transformants. The biallelic/homozygous transgene-free mutation rates for target genes among herbicide-resistant transformants ranged from 1.9% to 42.1% in tomato, tobacco, potato and citrus. This co-editing strategy is particularly useful for transgene-free genome editing of vegetatively propagated and perennial plant species in the T0 generation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The raw reads of genome resequencing for pummelo plants were deposited in the NCBI Bioproject database under accession number PRJNA931434. The reference genome of pummelo was downloaded from public citrus genome database CPBD: Citrus Pan-genome to Breeding Database (http://citrus.hzau.edu.cn/index.php). The raw reads of genome resequencing for tomato plants were deposited in the NCBI Bioproject database under accession number PRJNA931572. The reference genome of tomato was downloaded from public tomato genome database of International Tomato Genome Sequencing Project https://solgenomics.net/organism/Solanum_lycopersicum/genome). Source data are provided with this paper.
References
Zhang, Y. et al. Efficient and transgene-free genome editing in wheat through transient expression of CRISPR/Cas9 DNA or RNA. Nat. Commun. 7, 12617 (2016).
Woo, J. W. et al. DNA-free genome editing in plants with preassembled CRISPR–Cas9 ribonucleoproteins. Nat. Biotechnol. 33, 1162–1164 (2015).
Liang, Z. et al. Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nat. Commun. 8, 14261 (2017).
Svitashev, S., Schwartz, C., Lenderts, B., Young, J. K. & Cigan, A. M. Genome editing in maize directed by CRISPR-Cas9 ribonucleoprotein complexes. Nat. Commun. 7, 13274 (2016).
He, Y. B. & Zhao, Y. D. Technological breakthroughs in generating transgene-free and genetically stable CRISPR-edited plants. aBIOTECH 1, 88–96 (2020).
He, Y. B. et al. Programmed self-elimination of the CRISPR/Cas9 construct greatly accelerates the isolation of edited and transgene-free rice plants. Mol. Plant 11, 1210–1213 (2018).
Wang, X. et al. Genomic analyses of primitive, wild and cultivated citrus provide insights into asexual reproduction. Nat. Genet. 49, 765–772 (2017).
Huang, X., Wang, Y. & Wang, N. Base editors for citrus gene editing. Front. Genome Ed. 4, 852867 (2022).
Tyurin, A. A., Suhorukova, A. V., Kabardaeva, K. V. & Goldenkova-Pavlova, I. V. Transient gene expression is an effective experimental tool for the research into the fine mechanisms of plant gene function: advantages, limitations, and solutions. Plants 9, 1187 (2020).
Kapila, J., De Rycke, R., Van Montagu, M. & Angenon, G. An Agrobacterium-mediated transient gene expression system for intact leaves. Plant Sci. 122, 101–108 (1997).
Chen, L. Z. et al. A method for the production and expedient screening of CRISPR/Cas9-mediated non-transgenic mutant plants. Hortic. Res. 5, 13 (2018).
Danilo, B. et al. Efficient and transgene-free gene targeting using Agrobacterium-mediated delivery of the CRISPR/Cas9 system in tomato. Plant Cell Rep. 38, 459–462 (2019).
Veillet, F. et al. Transgene-free genome editing in tomato and potato plants using Agrobacterium-mediated delivery of a CRISPR/Cas9 cytidine base editor. Int. J. Mol. Sci. 20, 402 (2019).
Banfalvi, Z., Csakvari, E., Villanyi, V. & Kondrak, M. Generation of transgene-free PDS mutants in potato by Agrobacterium-mediated transformation. BMC Biotechnol. 20, 25 (2020).
He, Y., Mudgett, M. & Zhao, Y. Advances in gene editing without residual transgenes in plants. Plant Physiol. 188, 1757–1768 (2022).
Xu, W. et al. Discriminated sgRNAs-based surrogate system greatly enhances the screening efficiency of plant base-edited cells. Mol. Plant 13, 169–180 (2020).
Li, H. Y. et al. Multiplex precision gene editing by a surrogate prime editor in rice. Mol. Plant 15, 1077–1080 (2022).
Kuroiwa, K. et al. An iterative gene-editing strategy broadens eIF4E1 genetic diversity in Solanum lycopersicum and generates resistance to multiple potyvirus isolates. Plant Biotechnol. J. 21, 918–930 (2023).
Malabarba, J. et al. New strategies to overcome present CRISPR/Cas9 limitations in apple and pear: efficient dechimerization and base editing. Int. J. Mol. Sci. 22, 319 (2021).
Kuang, Y. J. et al. Base-editing-mediated artificial evolution of OsALS1 in planta to develop novel herbicide-tolerant rice germplasms. Mol. Plant 13, 565–572 (2020).
Wu, J. et al. Engineering herbicide-resistant oilseed rape by CRISPR/Cas9-mediated cytosine base-editing. Plant Biotechnol. J. 18, 1857–1859 (2020).
Zhang, R. et al. Generating broad-spectrum tolerance to ALS-inhibiting herbicides in rice by base editing. Sci. Chin. Life Sci. 64, 1624–1633 (2021).
Liang, Z., Wu, Y. Q., Ma, L. L., Guo, Y. J. & Ran, Y. D. Efficient genome editing in Setaria italica using CRISPR/Cas9 and base editors. Front. Plant Sci. 12, 815946 (2022).
Zhang, R. et al. Generation of herbicide tolerance traits and a new selectable marker in wheat using base editing. Nat. Plants 5, 480–485 (2019).
Bottero, E. et al. Generation of a multi-herbicide-tolerant alfalfa by using base editing. Plant Cell Rep. 41, 493–495 (2022).
Cheng, H. T. et al. Base editing with high efficiency in allotetraploid oilseed rape by A3A-PBE system. Plant Biotechnol. J. 19, 87–97 (2021).
Yu, Q. et al. AHAS herbicide resistance endowing mutations: effect on AHAS functionality and plant growth. J. Exp. Bot. 61, 3925–34 (2010).
Li, M. et al. ALS herbicide resistance mutations in Raphanus raphanistrum: evaluation of pleiotropic effects on vegetative growth and ALS activity. Pest Manag. Sci. 69, 689–95 (2013).
Légère, A. et al. Growth characterization of kochia (Kochia scoparia) with substitutions at Pro197 or Trp574 conferring resistance to acetolactate synthase-inhibiting herbicides. Weed Sci. 61, 267–276 (2013).
Tian, S. et al. Engineering herbicide-resistant watermelon variety through CRISPR/Cas9-mediated base-editing. Plant Cell Rep. 37, 1353–1356 (2018).
Tardif, F. et al. A mutation in the herbicide target site acetohydroxyacid synthase produces morphological and structural alterations and reduces fitness in Amaranthus powellii. N. Phytol. 169, 251–64 (2006).
Kwon, C. T. et al. Rapid customization of Solanaceae fruit crops for urban agriculture. Nat. Biotechnol. 38, 182–188 (2020).
Hu, Y. et al. Lateral organ boundaries 1 is a disease susceptibility gene for citrus bacterial canker disease. Proc. Natl Acad. Sci. USA 111, E521–E529 (2014).
Huang, X., Wang, Y. & Wang, N. Highly efficient generation of canker-resistant sweet orange enabled by an improved CRISPR/Cas9 system. Front. Plant Sci. 12, 769907 (2022).
Jia, H. G. et al. Genome editing of the disease susceptibility gene CsLOB1 in citrus confers resistance to citrus canker. Plant Biotechnol. J. 15, 817–823 (2017).
Jia, H. G. & Wang, N. Generation of homozygous canker-resistant citrus in the T0 generation using CRISPR–SpCas9p. Plant Biotechnol. J. 18, 1990–1992 (2020).
Jin, S. et al. Rationally designed APOBEC3B cytosine base editors with improved specificity. Mol. Cell 79, 728–740 (2020).
Kim, S., Kim, D., Cho, S. W., Kim, J. & Kim, J. S. Highly efficient RNA-guided genome editing in human cells via delivery of purified Cas9 ribonucleoproteins. Genome Res. 24, 1012–1019 (2014).
Randall, L. B. et al. Genome- and transcriptome-wide off-target analyses of an improved cytosine base editor. Plant Physiol. 187, 73–87 (2021).
O’Malley, R. C. & Ecker, J. R. Linking genotype to phenotype using the Arabidopsis unimutant collection. Plant J. 61, 928–940 (2010).
Omura, M. & Shimada, T. Citrus breeding, genetics and genomics in Japan. Breed. Sci. 66, 3–17 (2016).
Darqui, F. et al. The ALS gene as genetic target in CRISPR/Cas approaches: what have we learned so far? Mod. Concepts Dev. Agron. https://doi.org/10.31031/MCDA.2020.07.000656 (2020).
Turnbull, C., Lillemo, M. & Hvoslef-Eide, T. A. K. Global regulation of genetically modified crops amid the gene edited crop boom—a review. Front Plant Sci. 12, 630396 (2021).
Su, H. et al. Generation of the transgene-free canker-resistant Citrus sinensis using Cas12a/crRNA ribonucleoprotein in the T0 generation. Nat. Commun. 14, 3957 (2023).
Schindele, P. & Puchta, H. Engineering CRISPR/LbCas12a for highly efficient, temperature-tolerant plant gene editing. Plant Biotechnol. J. 18, 1118–1120 (2020).
Zhang, Y. X. et al. Expanding the scope of plant genome engineering with Cas12a orthologs and highly multiplexable editing systems. Nat. Commun. 12, 1944 (2021).
Jia, H. G., Omar, A., Orbović, V. & Wang, N. Biallelic editing of the LOB1 promoter via CRISPR/Cas9 creates canker-resistant ‘Duncan’ grapefruit. Phytopathology 112, 308–314 (2022).
Jia, H. G., Orbovic, V. & Wang, N. CRISPR-LbCas12a-mediated modification of citrus. Plant Biotechnol. J. 17, 1928–1937 (2019).
Jia, H. G., Wang, Y. C., Su, H., Huang, X. E. & Wang, N. LbCas12a–D156R efficiently edits LOB1 effector binding elements to generate canker-resistant citrus plants. Cells 11, 315 (2022).
Sun, H. J., Uchii, S., Watanabe, S. & Ezura, H. A highly efficient transformation protocol for Micro-Tom, a model cultivar for tomato functional genomics. Plant Cell Physiol. 47, 426–431 (2006).
Huang, X. E., Wang, Y. C., Xu, J. & Wang, N. Development of multiplex genome editing toolkits for citrus with high efficacy in biallelic and homozygous mutations. Plant Mol. Biol. 104, 297–307 (2020).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Poplin, R. et al. A universal SNP and small-indel variant caller using deep neural networks. Nat. Biotechnol. 36, 983–987 (2018).
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Liu, H. et al. CRISPR-P 2.0: an improved CRISPR-Cas9 tool for genome editing in plants. Mol. Plant 10, 530–532 (2017).
Bae, S., Park, J. & Kim, J. S. Cas-OFFinder: a fast and versatile algorithm that searches for potential off-target sites of Cas9 RNA-guided endonucleases. Bioinformatics 30, 1473–1475 (2014).
Acknowledgements
We thank Wang lab members for constructive suggestions and insightful discussions. This project was supported by funding from Florida Citrus Initiative Program, Citrus Research and Development Foundation, US Department of Agriculture National Institute of Food and Agriculture grants 2022-70029-38471, 2021-67013-34588, 2018-70016-27412 and 2016-70016-24833, FDACS Specialty Crop Block Grant Program, and Hatch project (FLA-CRC-005979) (N.W.).
Author information
Authors and Affiliations
Contributions
X.H., H.J. and N.W. conceptualized and designed the experiments. X.H., H.J. and Y.W. performed the experiments. J.X. and J.W. performed bioinformatics. X.H., H.J. and N.W. wrote the manuscript with input from all co-authors.
Corresponding author
Ethics declarations
Competing interests
N.W., H.J. and X.H. filed a PCT patent application based on the results reported in this paper. All other authors declare no competing financial interests.
Peer review
Peer review information
Nature Plants thanks Myeong-Je Cho, Xiaoou Dong, Pengcheng Wei, Lanqin Xia, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
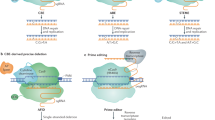
Extended Data Fig. 1 Generation of transgene-free, gene-edited plants in the first generation using the CBE-Cas12a-GFP construct.
The CBE-Cas12a-GFP construct consists of CBE, Cas12a, and GFP, each driven by its own promoter. CBE and its gRNA are used to base edit the ALS gene to confer resistance to herbicide chlorsulfuron; Cas12a and its crRNA are used for target gene editing; GPF is used for selecting transgene-free transformants. The construct is introduced into plants through Agrobacterium-mediated transient expression. Transformants are screened on media containing chlorsulfuron. Putative transgene-free, gene-edited plants are kept if they lack green fluorescence. Plants with fluorescence are discarded. Regenerants without GFP are subjected to genotyping and further analysis.
Extended Data Fig. 2 Establishment of herbicide-assisted transgene-free genome editing system.
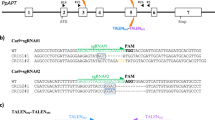
a, The CBE-Cas12a-GFP-SlALS1 construct used in the generation of transgene-free, SlALS1-edited tomato. The gRNA for SlALS1 is boxed. The targeted nucleotides (CC) are highlighted in yellow. CsVMV, Cassava vein mosaic virus promoter; U6, citrus U6 promoter; CmYLCV, Cestrum yellow leaf curling virus promoter; CBE, cytosine base editor; T, terminator. For GFP, Nos terminator; for SlALS gRNA, poly (T) terminator; for CBE and Cas12a, HSP 18.2 terminator. RB, T-DNA right border; LB, T-DNA left border. b, PCR amplification of GFP in the regenerated chlorsulfuron-resistant tomato lines with or without green fluorescence. c, SlALS1 gene genotyping of chlorsulfuron-resistant tomato regenerants through restriction enzyme digestion of PCR amplicons with StyI. PCR amplicons spanning the SlALS1 gRNA region were subjected to restriction enzyme digestion with StyI. Editing of the targeted nucleotides abolishes the StyI recognition site, resulting in resistance to StyI digestion. Bottom text: SlALS1 genotypes in the edited lines were confirmed by Sanger sequencing. b and c were repeated two times independently with similar results. Source data are provided as a Source data file.
Extended Data Fig. 3 Tomato calli and shoots selected on media containing chlorsulfuron herbicide.
a, Tomato calli were grown on the medium containing 110 nM chlorsulfuron. b, Close-up view of calli from the selection medium shown in a. c, Tomato calli and shoots selected on medium containing 110 nM chlorsulfuron.
Extended Data Fig. 4 Heritability test.
a, SlER genotypes of the progenies of sler-4. Seeds of the sler-4 line were germinated, and the seedlings were genotyped at SlER. b, PCR amplification of GFP and Cas12a for lines listed in a. At least 16 clones for each gene from each plant were subjected to Sanger sequencing. This experiment was repeated two times independently with similar results. Source data are provided as a Source data file.
Extended Data Fig. 5 Green fluorescence observation of seeds of transgenic and transgene-free lines.
Detection of green fluorescence in sler-4 and transgenic control seeds was conducted under a GFP filter.
Extended Data Fig. 6 Efficient transgene-free gene editing of tomato in the T0 generation with 2 crRNAs.
a, Construct scheme showing 1 crRNA targeting SlRbohD. b, Construct scheme showing 2 crRNAs targeting SlRbohD. c, The SlRbohD genotypes of the transgene-free, homozygous/biallelic edited lines without green fluorescence. d, Comparison of rate of transgene-free, homozygous/biallelic mutants using 1 crRNA and 2 crRNAs. e, PCR amplification of GFP and Cas12a in the lines shown in c. This experiment was repeated two times independently with similar results. Source data are provided as a Source data file.
Extended Data Fig. 7 Transgene-free, multiplex gene editing of tomato in the first generation.
a, Generation of transgene-free, biallelic/homozygous double mutants of tomato for SlEDS1 and SlPAD4. b, PCR amplification of GFP and Cas12a from the edited sleds1/slpad4 mutant lines from a. This experiment was repeated two times independently with similar results. Source data are provided as a Source data file. c, Generation of transgene-free, biallelic/homozygous double mutants for SlDMR6 and SlINVINH1. d, PCR amplification of GFP and Cas12a from the edited sldmr6/slinvinh1 mutant lines from c. This experiment was repeated two times independently with similar results. Source data are provided as a Source data file.
Extended Data Fig. 8 Transgene-free gene editing in the first generation (T0) in tobacco and potato.
a-c, Co-editing of NtALS and NtPDS in Nicotiana tabacum. a, Albino phenotype with or without green fluorescence. Regenerants were selected on herbicide chlorsulfuron-containing media. Upper: transgenic albino tobacco plant; lower: transgene-free albino tobacco plant. b, Confirmation of transgene-free gene editing. PCR amplification of GFP and Cas12a in WT, non-transgenic (NT), and transgenic (T) plants. This experiment was repeated two times independently with similar results. Source data are provided as a Source data file. c, Genotypes of NtALS, NtPDS genes in a transgene-free, albino tobacco line from a. d & e, Transgene-free gene editing in potato. d, PCR amplification of GFP and Cas12a from a regenerated potato line 9 and control transgenic plant. This experiment was repeated two times independently with similar results. Source data are provided as a Source data file. e, Genotype of line 9 at StDMR6. crRNAs are underlined. 1 crRNA was used for StDMR6 editing.
Extended Data Fig. 9 Analysis of line ep-8.
a, Identification of the construct integration site in the tomato genome through whole genome sequencing. Blue, tomato DNA sequence; red, construct DNA sequence. b Illustrations demonstrating the partial integration of the transformation construct into the tomato genome. Arrows indicate the primers used in the following PCR confirmation. c, PCR confirmation. This experiment was repeated two times independently with similar results. Source data are provided as a Source data file.
Supplementary information
Supplementary Information
Supplementary Tables 1–5, Figs. 1–18 and Files 1 and 2.
Source data
Source Data Fig. 1
Unprocessed gel.
Source Data Fig. 2
Unprocessed gel.
Source Data Extended Data Fig. 2
Unprocessed gel.
Source Data Extended Data Fig. 4
Unprocessed gel.
Source Data Extended Data Fig. 6
Unprocessed gel.
Source Data Extended Data Fig. 7
Unprocessed gel.
Source Data Extended Data Fig. 8
Unprocessed gel.
Source Data Extended Data Fig. 9
Unprocessed gel.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, X., Jia, H., Xu, J. et al. Transgene-free genome editing of vegetatively propagated and perennial plant species in the T0 generation via a co-editing strategy. Nat. Plants 9, 1591–1597 (2023). https://doi.org/10.1038/s41477-023-01520-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-023-01520-y
This article is cited by
-
Blueprint for non-transgenic edited plants
Nature Plants (2023)