Abstract
Soil organic carbon (SOC) is a primary regulator of the forest–climate feedback. However, its indicative capability for the soil CH4 sink is poorly understood due to the incomplete knowledge of the underlying mechanisms. Therefore, SOC is not explicitly included in the current model estimation of the global forest CH4 sink. Here, using in-situ observations, global meta-analysis, and process-based modeling, we provide evidence that SOC constitutes an important variable that governs the forest CH4 sink. We find that a CH4 sink is enhanced with increasing SOC content on regional and global scales. The revised model with SOC function better reproduces the field observation and estimates a 39% larger global forest CH4 sink (24.27 Tg CH4 yr−1) than the model without considering SOC effects (17.46 Tg CH4 yr−1). This study highlights the role of SOC in the forest CH4 sink, which shall be factored into future global CH4 budget quantification.
Similar content being viewed by others
Introduction
Upland soils are an important sink of atmospheric methane (CH4) owing to the activity of methanotrophs, contributing 5–10% of atmospheric CH4 removal1,2. Among upland ecosystems, forests play a pivotal role, accounting for up to 62% of the global soil CH4 sink3. Therefore, accurate quantification of the global CH4 sink provided by forest ecosystems is essential to improving our understanding of the global CH4 budget.
The magnitude of global forest CH4 sink remains poorly constrained due to our limited understanding of the process1,4. Previous models estimated the global forest CH4 sink ranges from 9 to 24 Tg CH4 yr−1 1,3,4,5,6,7,8, which highlights the necessity for reducing this uncertainty to better estimate the sink. Conventionally, methanotrophs are considered to utilize CH4 as their sole carbon and energy source. Accordingly, the existing estimations using various process-based modeling and meta-analysis approaches generally use soil factors related to gas diffusivity (CH4 and O2 availability) as major determinants of soil CH4 sink4,6,7,8. Along with soil gas diffusivity, other soil variables such as alternative carbon sources9,10 and nutrient availability11,12 have been suggested to play a pivotal role in determining the soil CH4 sink. Incorporation of physiological characteristics of methanotrophs into the process-based model indeed constrained the uncertainty of estimation7,13,14, suggesting that it is crucial to incorporate them into the model to improve our estimation capability. However, due to our incomplete understanding of the regulatory mechanisms behind these variables, current estimations do not adequately consider these variables in the model7,13,14. Thus, investigation of the regulatory mechanisms of the previously overlooked drivers and further consideration of these factors is needed to reduce the uncertainty of global forest CH4 sink estimation.
Soil organic matter (SOM) is a complex of decaying organic matter, degradation products such as humus, and soil microbes, and plays a pivotal role in terrestrial ecosystem functioning15,16,17. While SOM data has traditionally been used as a soil quality indicator in soil science, various disciplines are now acknowledging the significance of SOM attributes as indicators of ecosystem processes, including climate feedback. Therefore, diverse ecosystem process models now incorporate quantitative and qualitative data on SOM18. Previous models generally considered SOM to negatively affect the soil CH4 sink because it can provide carbon substrates to methanogens, thereby increasing CH4 emissions19. However, this assumption does not consider the diverse mechanisms of SOM-dependent regulation of soil variables that are associated with soil CH4 oxidation, constraining our estimation capability.
To elucidate the mechanisms underlying the SOM-dependent control of the forest CH4 sink and to further improve global forest CH4 sink estimation, three independent approaches—continuous in-situ measurement, global meta-analysis, and process-based modeling—were used in this study. First, we monitored in-situ CH4 uptake rate and edaphic characteristics, including SOM content, for 2 years in subtropical forests with different tree species (Japanese cedar, oak, mixed forest) and in temperate coniferous forests with different thinning intensities (light, moderate, heavy intensity, and control). Second, we performed a global meta-analysis to establish a relationship between the forest CH4 sink and soil organic carbon (SOC) content on a global scale. Finally, we revised a process-based forest CH4 sink model by considering the effect of SOC content to quantify the global forest CH4 sink.
Results and discussion
Field observations
Continuous measurements of CH4 fluxes and edaphic characteristics for two years from April 2018 to March 2020 revealed there are significant positive correlations between SOM content and CH4 uptake rate both in a subtropical forest with different tree species (Fig. 1a; R2 = 0.538, P < 0.001) and in a temperate coniferous forest with different thinning intensities (Fig. 1b; R2 = 0.258, P < 0.001). The positive relationship observed in temperate forest further suggests that the variability of CH4 uptake between the forests with the same dominant species but different soil characteristics can be partially explained by the SOM content. It is conceivable that the positive correlations observed between SOM content and CH4 uptake rate at the two examined sites may have been primarily influenced by other environmental variables that exhibit variability according to tree species or thinning intensity and that are closely associated with CH4 uptake. However, partial correlation analysis indicates that even after removing the influence of other variables such as air-filled porosity (AFP), dissolved organic carbon (DOC) content, and water-filled pore space, SOM content remains positively correlated with CH4 uptake rate (Supplementary Table 1). Previous studies have demonstrated that magnitudes of soil CH4 sinks exhibit variability among different tree species, which can be attributed to distinctions in the physical and chemical properties of litter20,21,22, and inorganic N content23. In addition to the existing literature, our study reveals a noteworthy positive correlation between SOM content and CH4 uptake rate in subtropical forests. This finding further supports the notion that SOM content is an influential factor in distinguishing soil CH4 sink capacity among different tree species. Moderate intensity plot exhibited the highest CH4 uptake rate in temperate forest, which can be attributed to the highest SOM content (Supplementary Fig. 1b) and understory biomass (Supplementary Fig. 2a). We found a strong positive relationship between SOM content and understory biomass (Supplementary Fig. 2b), indicating that understory attributed to higher SOM content24,25. These findings suggest that understory vegetation may indirectly promote soil CH4 uptake by increasing SOM content. In addition, understory vegetation can enhance soil CH4 uptake by stimulating oxygen and CH4 transportation to the surface soil layer through the root26.
Relationships between forest soil CH4 uptake rate and soil organic matter (SOM) content in (a) subtropical forest with different tree species (N = 72, P < 0.001) and (b) temperate coniferous forest with different thinning intensities (N = 258, P < 0.001). A linear mixed effect model was used and the error bands surrounding the regression lines represent the 95% confidence interval of the correlation.
On the basis of these field measurement data and statistical analysis, we hypothesize that the following mechanisms underpin the positive effect of SOM on forest CH4 uptake. First, SOM can provide alternative carbon substrates to facultative methanotrophs and enhance their activity by stimulating growth. Soil labile carbon forms such as formate, acetate, and methanol are suggested to increase CH4 oxidation activity by stimulating methanotrophic growth9,10. Sullivan et al. 27 performed an in vitro experiment in semiarid soils and reported an increase in CH4 oxidation rate with increasing DOC content. In accordance with previous studies, CH4 uptake rate had significant positive correlations with DOC content both in the subtropical forest and temperate forest at our study sites (Table 1). We also found significant positive correlations between SOM content and DOC content both in the subtropical forest and temperate forest (Table 1). These results indicate that SOM may indirectly stimulate soil CH4 uptake in both types of forests by providing alternative C compounds for methanotrophs.
The second hypothetical mechanism is based on the enhanced water-holding capacity of organic-rich soil28, which can alleviate water stress and stimulate CH4 oxidation. Generally, soil water content (SWC) has a unimodal relationship with soil CH4 uptake rate29,30. Under low soil moisture, water stress can promote ethylene production by plants, which inhibits CH4 oxidation in soil30,31. In addition, water stress on methanotrophs can result in reducing their activity29. On the other hand, high soil moisture inhibits methanotrophic activity by decreasing gas diffusivity30. A global meta-analysis showed that the forest CH4 sink increased with increasing mean annual precipitation, suggesting a higher CH4 uptake at sites with more precipitation on a global scale32. These authors suggested that the positive effect of water content induced by water-stress alleviation is larger than the negative effect of decreased CH4 and O2 availability in the forests. Several other studies indicated the prevalent role of precipitation in promoting biological oxidation of CH4 in forest soils19,33,34, suggesting that enhanced water-holding capacity may stimulate methanotrophic activity, with the exception of highly saturated periods. Although the correlation between SWC and CH4 uptake rate was not significant at our study sites, the highest CH4 uptake rate was observed in the oak forest (Supplementary Fig. 3a) and a plot with moderate intensity thinning (Supplementary Fig. 3b), where the highest SWC was observed in the subtropical forest (Supplementary Fig. 4a) and temperate forest (Supplementary Fig. 4b), respectively. Furthermore, we found significant positive correlations between SWC and CH4 uptake rate when the highly saturated periods were excluded, both in the subtropical forest (Supplementary Fig. 5c; R2 = 0.09; P < 0.05), and temperate forest (Supplementary Fig. 5d; R2 = 0.02; P < 0.05), indicating that soil moisture positively influences soil CH4 uptake, with the exception of periods of high soil water saturation. Significant positive correlations between SOM content and SWC both in the subtropical forest and temperate forest (Table 1) indicate that higher moisture content in organic matter–rich soil can be attributed to enhanced water-holding capacity. Collectively, these results reinforce our hypothesis that the enhanced water-holding capacity of organic-rich soil may stimulate CH4 oxidation activity in forest soils by alleviating water stress.
Lastly, SOM content increases soil gas transport capability by increasing pore space owing to the positive effect of organic matter on soil aggregation, tilth, and biopore development15. Indeed, we found a positive correlation between SOM content and AFP in the temperate forest (Table 1). Although the correlation was not significant in the subtropical forest, the highest AFP was observed in the oak forest, where the SOM content was highest, and the lowest AFP was observed in the Japanese cedar forest, where the SOM content was lowest (Supplementary Fig. 1a and Supplementary Fig. 6a). These results indicate that SOM enhances gas diffusivity in forest soils. AFP was positively correlated with soil CH4 uptake rate both in the subtropical forest and temperate forest (Table 1), suggesting that SOM may indirectly regulate CH4 uptake by controlling the soil pore space.
In addition to these three suggested mechanisms based on in-situ observations, we speculate that several other mechanisms may play a role in the relationship between SOM and CH4 oxidation rates. For example, increased microbial diversity in SOM-rich soil can increase methanotrophic activity. Recent microbial pure-culture studies revealed that the diversity of heterotrophic bacteria significantly increases the activity and growth rate of methanotrophs by generating products beneficial for methanotrophs or by removing inhibitory products35,36,37,38,39. For example, a culture study found the higher CH4 oxidation rate for cultured methanotrophs with diverse heterotrophs compared to samples incubated with methanotrophs only35. It was suggested that this mutual relationship was based on the fact that certain heterotrophs produce essential metabolites for methanotrophs, thus stimulating CH4 oxidation activity. As SOM generally increases soil microbial diversity40,41, a more diverse microbial community in SOM-rich soil can increase CH4 sink capacity by enhancing the positive interaction between methanotrophs and heterotrophic bacteria. Additionally, SOM can provide nutrients such as N and P to methanotrophs and alleviate nutrient limitations11,12,42,43,44,45.
Relationship between soil CH4 uptake rate and SOC content in global forests
To provide the global context for our in-situ measurements, we retrieved data on mean forest CH4 uptake rate and SOC content worldwide from 81 published articles (Fig. 2a). A significant positive correlation was found between forest mean CH4 uptake rate and SOC content (Fig. 2b; R2 = 0.323, P < 0.001), suggesting that SOC indeed positively influences forest CH4 uptake in global forests. In addition, by comparing four ranges of SOC content, we confirmed that CH4 uptake rate increases with the increasing SOC content (Fig. 2c): mean soil CH4 uptake rate was 0.79 ± 0.09 mg CH4 m−2 d−1 in a SOC content range from 0 to 20 g kg−1, but it was more than 3 times higher (2.98 ± 0.32 mg CH4 m−2 d−1) when SOC content ranged from 60 to 100 g kg−1. While SOC has been regarded to rather reduce the forest CH4 sink by affecting methanogenesis19,32, the relationship we found in the field observations and meta-analysis suggest that the positive effect of SOC on soil CH4 uptake outweighs the negative effect at both regional and global scales. These results further suggest the need to incorporate the positive effect of SOC into the estimation model.
a The location of the 204 sites across the globe that are included in the meta-analysis. The size of the dot represents the number of sites. b Relationship between reported forest CH4 uptake rate and soil organic carbon (SOC) content (N = 204, P < 0.001). Linear regression was used and the error bands surrounding the regression lines represent the 95% confidence interval of the correlation. c Forest CH4 uptake rate at different SOC content ranges. Differing letters denote statistically significant differences between SOC content ranges based on the Kruskal–Wallis one-way ANOVA (P < 0.05). The thick central line represents the median value, the boxed areas represent the interquartile range, and the whiskers show the maximum and minimum values.
Revised global forest CH4 sink estimation model
We incorporated the effect of SOC into the terrestrial ecosystem model (TEM) to quantify the global forest CH4 sink. Overall, a comparison between the observed and simulated forest CH4 uptake rates showed a 30% reduction of root mean square error (RMSE) values in the new model that included the function of SOC (TEM-SOC; Supplementary Fig. 7b; RMSE = 0.56) in comparison with the model without this function (TEM-DEF; Supplementary Fig. 7a; RMSE = 0.79; Liu et al.8). However, when different biomes were analyzed separately, estimation using the new model substantially reduced RMSE in temperate and boreal forests but increased it in the tropical forest. This result indicates that the simulated CH4 uptake rate better represents the observational data in temperate and boreal forests by considering the effect of SOC, but not in the tropical forest.
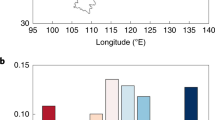
Overall, TEM-SOC estimates the global forest CH4 sink as 24.27 ± 1.64 Tg CH4 yr−1, which is 39% larger than the estimate by TEM-DEF (17.46 ± 5.09 Tg CH4 yr−1) (Fig. 3). As previous bottom-up estimation using a process-based model underestimated the global upland CH4 sink by 8 Tg CH4 yr−1 in comparison with the top-down estimation that uses the atmospheric inverse-modeling framework46, our revised model reconciles the disparity between the two different approaches. Compared to TEM-DEF, TEM-SOC estimated 38% and 341% higher CH4 sink in temperate forest (13.92 ± 1.44 Tg CH4 yr−1 versus 10.08 ± 3.28 Tg CH4 yr−1) and boreal forest (5.82 ± 1.27 Tg CH4 yr−1 versus 1.32 ± 0.44 Tg CH4 yr−1), respectively, whereas estimated 23% lower CH4 sink in tropical forest (4.65 ± 0.37 Tg CH4 yr−1 versus 6.06 ± 2.8 Tg CH4 yr−1) (Fig. 3c). The difference among biomes in the magnitude of responses to SOC incorporation was caused perhaps by different sensitivity of each biome to soil variables including SOC content32. In the regions characterized by predominantly low mean temperatures, such as boreal forests, it has been proposed that CH4 and O2 availability are not the primary limiting factors for CH4 uptake32,33. Rather, the biological activity of methanotrophs, which may be subject to physiological control by other environmental factors such as nutrient availability and pH, limits the process. Thus, in regions with low mean temperature, the role of soil variables that can promote the biological oxidation of CH4 physiologically becomes more critical than CH4 and O2 availability. The CH4 sink in temperate and boreal forests estimated by TEM-SOC was larger than that estimated by TEM-DEF because the biological activity of methanotrophs is limited in these biomes, and TEM-SOC considers the positive physiological effect of SOC on soil CH4 consumption. Meanwhile, the estimated CH4 sink in the tropical forest was lower in TEM-DEF than in TEM-SOC because the biological activity of methanotrophs is not limited owing to the high temperature, while the CH4 and O2 availability is more likely the limiting factor. Indeed, our global meta-analysis showed that the slope of the linear regression line between SOC content and soil CH4 uptake rate was steepest at high latitudes (40–70° N&S), followed by middle latitudes (20–40° N&S), and shallowest at low latitudes (0–20° N&S) (Supplementary Fig. 8), indicating that the positive effect of SOC on forest CH4 sink is weaker in warmer regions. The differences in the magnitudes of responses between biomes may also be induced by different SOC contents. Generally, boreal forests have higher SOC contents than tropical and temperate forests owing to the slow decomposition because of the cold temperature. Because of the high SOC content, the positive effect of SOC in boreal forests results in a substantial increase in soil CH4 sink, while this effect is less significant in tropical forests due to the relatively low SOC content. Process-based soil CH4 sink model estimates lower CH4 uptake from the boreal forest than the temperate and tropical forests most likely due to the low temperature in the boreal biome3,7. However, previous in-situ observation data consistently suggested that the mean soil CH4 uptake rate of boreal forests is similar to those of most other biomes32,34, indicating that previous process-based models underestimated the CH4 sink capacity of boreal forests.
The simulated global forest mean CH4 uptake rate of (a) the model without the function of SOC (TEM-DEF) and (b) the model with the function of SOC (TEM-SOC). c Estimated mean annual forest CH4 sink (Tg CH4 yr–1) results from TEM-DEF and TEM-SOC during 2000–2016 with different biome types. The error bars represent 1σ of TEM-DEF and TEM-SOC, which were determined by varying the optimized parameters from ensemble simulations (N = 50 for TEM-DEF and N = 20 for TEM-SOC).
Our study based on field measurements and global meta-analysis provides evidence on the pivotal role of SOC in the forest soil CH4 sink on both regional and global scales. Our revised model with SOC function improves the estimation and reconciles the disparity between bottom-up estimation and top-down modeling. Revealing the response of climate feedback to changing SOC dynamics is one of the major challenges for projecting future climate change. SOC stocks in terrestrial ecosystems including forests are sensitive to climate change and are projected to be reduced by global warming47,48,49,50,51, which may induce a positive feedback to climate change. The global soil CH4 sink is projected to increase in the future, most likely due to the increase in atmospheric CH4 concentration and temperature rise52. However, our study suggests that the reduction of the SOC stock in forest ecosystems by warming can partially offset the increasing soil CH4 sink, potentially inducing positive feedback to warming along with the increasing CO2 emission. Thus, it is important to incorporate the effect of SOC on the forest CH4 sink to better understand the impact of climate change and constrain the global CH4 budget.
Methods
Site description and soil sampling
The study sites were located in the Hannam experimental forest (subtropical forest; 33°33’N, 126°65’E) and in Gwangneung experimental forest (temperate forest; 37°76’ E, 127°17’E) in the Republic of Korea. The mean annual temperature was 17.0 °C and 11.7 °C in subtropical forest and temperate forest, respectively, and the mean annual precipitation was 2188 mm and 1364 mm during the observation period in subtropical forest and temperate forest, respectively. In subtropical forest, we selected 3 different tree species: Quercus acuta (oak), Cryptomeria japonica (Japanese cedar), and mixed forest. In temperate forest, we selected Pinus koraiensis dominant forest with 3 different thinning intensities: light intensity (700 trees ha–1), moderate intensity (600 trees ha–1), heavy intensity (500 trees ha–1), and control (800 trees ha–1). Soil samples were collected seasonally (spring, summer, autumn, and winter) in 2018 and 2019 at subtropical forest and monthly from April 2018 to March 2020 at temperate forest. Soil samples were collected to the depth of 0–10 cm after litter layer and O-horizon removal. Soil temperature was measured at the depth of 5 cm after soil sampling. Collected samples were placed in plastic zipper bags and transported to the lab on ice. Samples were stored at 4 °C and further analyses were conducted within 3 days.
Edaphic characteristics
SWC was measured by oven-drying at 105 °C for 24 h, and SOM content was determined by a loss-on-ignition method. Soil DOC content was extracted by mixing soil with distilled water, filtered through 0.45-µm filter, and then analyzed by a TOC analyzer (TOC-VCHP, Shimadzu, Kyoto, Japan). Soil core of 0–10 cm depth was collected to measure bulk density. Based on the measured bulk density, Air-filled porosity was calculated as below:
Where ρs is particle density (2.65 g cm–3) and θv is volumetric moisture content.
Highly saturated periods were defined as periods when the measurements were taken right after a rain event or snowmelt, and when the water-filled pore space was higher than 85% and 60% in subtropical forests and temperate forests, respectively. Briefly, April 2018 in the subtropical forest and December 2018 and February 2019 in the temperate forest were defined as highly saturated periods.
Soil CH4 fluxes measurement
Soil CH4 fluxes were measured every 3 months in the subtropical forest from April 2018 to December 2019 and monthly in the temperate forest from April 2018 to March 2020. Fluxes were measured using the closed static chamber method with triplicate measurement in each treatment. Changes in headspace CH4 concentration were measured using GasScouterTM G4301 Mobile Gas Concentration Analyzer (Picarro Inc., Santa Clara, CA, USA) from April 2018 to September 2019. Due to mechanical issues with the portable analyzer, we took the headspace gas sample every 10 min for 50 min and transferred it to a pre-evacuated glass vial from October 2019 to April 2020. We used a GC-FID (CP-3800 Varian, USA) to measure the CH4 concentration. CH4 fluxes were calculated from linear regression slopes (chamber headspace [CH4] vs. time) with a minimum R2 = 0.82.
Literature review and data collection
We collected 204 data points from 81 published papers using Web of Science and Elsevier’s Scopus academic database. We searched articles published between 1989 to 2021 using the keyword ‘(CH4 uptake OR CH4 sink) AND (forest)’. Reported variables including annual CH4 uptake rate, topsoil SOC or SOM content, mean annual precipitation, mean annual temperature, latitude and longitude were extracted from each paper. Collected SOM contents (N = 5) were converted to SOC content by using the simple equation: SOM = SOC × 1.72 (van Bemmelen factor53). The van Bemmelen factor has been widely used to convert SOM content to SOC content54.We also collected papers that present only total C content (N = 26) since inorganic C content in forest soil is negligible55,56. When the paper presented SOC values with depth profiles, values below the humus layer were collected. In the case of field manipulation experiments such as warming, elevated CO2, and fertilizer addition, the CH4 uptake rate of only the control plot was collected to prevent other controlling variables affecting the CH4 uptake rate of forests with natural conditions.
Process-based modeling of global forest CH4 soil sink
TEM is a process-based biogeochemistry model and its CH4, soil, thermal, and hydrological dynamics have been evaluated in previous studies7,14,57. The model simulates CH4 production, oxidation, and three transport processes including diffusion, ebullition, and plant-mediated transport between soils and the atmosphere. The version of TEM we used is from Liu et al.8, which is referred as to TEM-DEF, and we focused on CH4 oxidation process in non-inundated forest regions.
For TEM-DEF, the CH4 oxidation is calculated by the product of the maximum potential oxidation rate (OMAX) and limiting functions of methane concentration, soil temperature, soil moisture, redox potential, nitrogen deposition, and diffusion limited by high soil moisture (CM, TSOIL, ESM, ROX, NDP, and DMS, respectively) (Eq. 3). For the new model, TEM-SOC, we revised the CH4 oxidation function by incorporating the SOC effect on CH4 oxidation (f(SOC) in Eq. 4). We optimized Michaelis-Menten function between SOC and CH4 oxidation using our collected data (Supplementary Fig. 9).
We optimized five parameters related to maximum oxidation potential, soil temperature, and moisture sensitivity for CH4 oxidation related to both TEM-DEF and TEM-SOC for tropical, temperate, and boreal forest regions (Supplementary Table 2 and 3). All other parameters were set the same as in Liu et al.8. We used the Shuffled Complex Evolution Approach in R language (SCE-UA-R) to minimize the difference between simulated and observed CH4 oxidation rates58.
To make spatially- and temporally-varying estimates of CH4 emission and consumption, we used spatially explicit data of land cover, soil pH and textures, meteorology, and leaf area index. We used SOC data from Soilgrids database59. The model was applied at the spatial resolution of 0.5° latitude by 0.5° longitude for forest upland ecosystems with a daily time-step during 2000–2016. A year of spin-up was used for CH4 equilibrium in soils, and simulated ecosystem-specific CH4 oxidations were then area weighted for each grid cell. For each site, 20 ensembles were run using SCE-UA-R with 10,000 maximum loops per parameter ensemble, and all of them reached steady state before the end of the loops.
Statistical analysis
Linear mixed-effect models were used to assess the positive relationship between SOM content and soil CH4 uptake rate in the field. In the model, tree species (subtropical forest) or thinning intensity (temperate forest) was used as a random effect for the incorporation of between-species or between-thinning intensity dissimilarity. Before conducting regression analysis, SOM content and soil CH4 uptake rate was log-transformed to improve the normality of the data and validity of regression analysis. Correlations between soil characteristics (SWC, soil DOC content, and AFP) and SOM content as well as soil characteristics and CH4 uptake rate were assessed with Pearson’s product moment correlation coefficients. Linear mixed effect model was used to assess the effect of tree species or thinning intensity on forest soil CH4 uptake rate, SOM content, soil AFPS, and SWC. Tree species or thinning intensity was used as categorical variables, and plot and month were used as random effects. Tukey’s HSD tests were used to compare individual means. Partial correlation analysis was conducted to assess whether SOM content was positively correlated with soil CH4 uptake rate without the influences of other variables that vary by tree species or thinning intensity. In this analysis, environmental variables that (1) vary by tree species or thinning intensity and (2) are closely related to soil CH4 uptake rate were selected as control variables.
Using the data we collected from 81 previously published papers, linear regression analysis was conducted to test whether positive relationship between SOC content and soil CH4 uptake rate also exists in the global forests. Before conducting linear regression analysis, collected soil CH4 uptake rate and SOC content were log-transformed to improve the normality of the data and the validity of the analysis. To assess whether forest soil CH4 uptake increases along the SOC content range, we conducted Kruskal–Wallis one-way ANOVA.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Supporting Information is available online. The dataset used for meta-analysis can be found in Supplementary Data. The model results are available at the Purdue University Research Repository (PURR) under accession code https://doi.org/10.4231/8K7W-NF8460.
Code availability
The code is also archived and freely available at the Purdue University Research Repository (PURR) at https://doi.org/10.4231/8K7W-NF8460.
References
Kirschke, S. et al. Three decades of global methane sources and sinks. Nat. Geosci. 6, 813–823 (2013).
Le Mer, J. & Roger, P. Production, oxidation, emission and consumption of methane by soils: A review. Eur. J. Soil Biol. 37, 25–50 (2001).
Dutaur, L. & Verchot, L. V. A global inventory of the soil CH4 sink. Glob. Biogeochem. Cycles 21, 1–9 (2007).
Murguia-Flores, F., Arndt, S., Ganesan, A. L., Murray-Tortarolo, G. & Hornibrook, E. R. C. Soil methanotrophy model (MeMo v1.0): a process-based model to quantify global uptake of atmospheric methane by soil. Geosci. Model Dev. 11, 2009–2032 (2018).
Ridgwell, A. J., Marshall, S. J. & Gregson, K. Consumption of atmospheric methane by soils: a process-based model. Glob. Biogeochem. Cycles 13, 59–70 (1999).
Curry, C. L. Modeling the soil consumption at atmospheric methane at the global scale. Glob. Biogeochem. Cycles 21, 1–15 (2007).
Zhuang, Q. et al. Response of global soil consumption of atmospheric methane to changes in atmospheric climate and nitrogen deposition. Glob. Biogeochem. Cycles 27, 650–663 (2013).
Liu, L. et al. Uncertainty quantification of global net methane emissions from terrestrial ecosystems using a mechanistically based biogeochemistry model. J. Geophys. Res. Biogeosciences 125, 1–19 (2020).
Jensen, S., Priemé, A. & Bakken, L. Methanol improves methane uptake in starved methanotrophic microorganisms. Appl. Environ. Microbiol. 64, 1143–1146 (1998).
West, A. E. & Schmidt, S. K. Acetate stimulates atmospheric CH4 oxidation by an alpine tundra soil. Soil Biol. Biochem. 31, 1649–1655 (1999).
Xu, M. et al. Low-level nitrogen addition promotes net methane uptake in a boreal forest across the great Xing’an mountain region, China. For. Sci. 60, 973–981 (2014).
Veraart, A. J., Steenbergh, A. K., Ho, A., Kim, S. Y. & Bodelier, P. L. E. Beyond nitrogen: the importance of phosphorus for CH4 oxidation in soils and sediments. Geoderma 259–260, 337–346 (2015).
Oh, Y. et al. A scalable model for methane consumption in arctic mineral soils. Geophys. Res. Lett. 43, 5143–5150 (2016).
Oh, Y. et al. Reduced net methane emissions due to microbial methane oxidation in a warmer Arctic. Nat. Clim. Chang. 10, 317–321 (2020).
Franzluebbers, A. J. Soil organic matter stratification ratio as an indicator of soil quality. Soil Tillage Res. 66, 95–106 (2002).
Bachmann, J. et al. Physical carbon-sequestration mechanisms under special consideration of soil wettability. J. Plant Nutr. Soil Sci. 171, 14–26 (2008).
Naveed, M. et al. Spatial variability of microbial richness and diversity and relationships with soil organic carbon, texture and structure across an agricultural field. Appl. Soil Ecol. 103, 44–55 (2016).
Billings, S. A. et al. Soil organic carbon is not just for soil scientists: measurement recommendations for diverse practitioners. Ecol. Appl. 31, 1–19 (2021).
Yu, L., Huang, Y., Zhang, W., Li, T. & Sun, W. Methane uptake in global forest and grassland soils from 1981 to 2010. Sci. Total Environ. 607–608, 1163–1172 (2017).
Borken, W. & Beese, F. Methane and nitrous oxide fluxes of soils in pure and mixed stands of European beech and Norway spruce. Eur. J. Soil Sci. 57, 617–625 (2006).
Christiansen, J. R. & Gundersen, P. Stand age and tree species affect N2O and CH4 exchange from afforested soils. Biogeosciences 8, 2535–2546 (2011).
Aronson, E. L., Allison, S. D. & Helliker, B. R. Environmental impacts on the diversity of methane-cycling microbes and their resultant function. Front. Microbiol. 4, 0–15 (2013).
Reay, D. S., Nedwell, D. B., McNamara, N. & Ineson, P. Effect of tree species on methane and ammonium oxidation capacity in forest soils. Soil Biol. Biochem. 37, 719–730 (2005).
Yang, Y. et al. Understory vegetation plays the key role in sustaining soil microbial biomass and extracellular enzyme activities. Biogeosciences 15, 4481–4494 (2018).
Fang, X. M. et al. Litter addition and understory removal influenced soil organic carbon quality and mineral nitrogen supply in a subtropical plantation forest. Plant Soil 460, 527–540 (2021).
Plain, C., Ndiaye, F. K., Bonnaud, P., Ranger, J. & Epron, D. Impact of vegetation on the methane budget of a temperate forest. N. Phytol. 221, 1447–1456 (2019).
Sullivan, B. W., Selmants, P. C. & Hart, S. C. Does dissolved organic carbon regulate biological methane oxidation in semiarid soils? Glob. Chang. Biol. 19, 2149–2157 (2013).
Schulten, H. R. & Schnitzer, M. The chemistry of soil organic nitrogen: a review. Biol. Fertil. Soils 26, 1–15 (1997).
Schnell, S. & King, G. M. Responses of methanotrophic activity in soils and cultures to water stress. Appl. Environ. Microbiol. 62, 3203–3209 (1996).
Zhou, X., Dong, H., Chen, C., Smaill, S. J. & Clinton, P. W. Ethylene rather than dissolved organic carbon controls methane uptake in upland soils. Glob. Chang. Biol. 20, 2379–2380 (2014).
Zhou, X., Smaill, S. J., Gu, X. & Clinton, P. W. Manipulation of soil methane oxidation under drought stress. Sci. Total Environ. 757, 144089 (2021).
Gatica, G., Fernández, M. E., Juliarena, M. P. & Gyenge, J. Environmental and anthropogenic drivers of soil methane fluxes in forests: global patterns and among-biomes differences. Glob. Chang. Biol. 26, 6604–6615 (2020).
Fang, H. J. et al. Effects of multiple environmental factors on CO2 emission and CH4 uptake from old-growth forest soils. Biogeosciences 7, 395–407 (2010).
Liu, L., Estiarte, M. & Peñuelas, J. Soil moisture as the key factor of atmospheric CH4 uptake in forest soils under environmental change. Geoderma 355, 113920 (2019).
Ho, A. et al. The more, the merrier: heterotroph richness stimulates methanotrophic activity. ISME J. 8, 1945–1948 (2014).
Jeong, S. Y., Cho, K. S. & Kim, T. G. Density-dependent enhancement of methane oxidation activity and growth of Methylocystis sp. by a non-methanotrophic bacterium Sphingopyxis sp. Biotechnol. Rep. 4, 128–133 (2014).
Krause, S. M. B. et al. Lanthanide-dependent cross-feeding of methane-derived carbon is linked by microbial community interactions. Proc. Natl. Acad. Sci. USA. 114, 358–363 (2017).
Stock, M. et al. Exploration and prediction of interactions between methanotrophs and heterotrophs. Res. Microbiol. 164, 1045–1054 (2013).
Veraart, A. J. et al. Living apart together - Bacterial volatiles influence methanotrophic growth and activity. ISME J. 12, 1163–1166 (2018).
Maestre, F. T. et al. Increasing aridity reduces soil microbial diversity and abundance in global drylands. Proc. Natl. Acad. Sci. USA. 112, 15684–15689 (2015).
Bastida, F. et al. Soil microbial diversity–biomass relationships are driven by soil carbon content across global biomes. ISME J. 15, 2081–2091 (2021).
Rigler, E. & Zechmeister-Boltenstern, S. Oxidation of ethylene and methane in forest soils - Effect of CO2 and mineral nitrogen. Geoderma 90, 147–159 (1999).
Mohanty, S. R., Bodelier, P. L. E., Floris, V. & Conrad, R. Differential effects of nitrogenous fertilizers on methane-consuming microbes in rice field and forest soils. Appl. Environ. Microbiol. 72, 1346–1354 (2006).
Geng, J. et al. Soil nitrate accumulation explains the nonlinear responses of soil CO2 and CH4 fluxes to nitrogen addition in a temperate needle-broadleaved mixed forest. Ecol. Indic. 79, 28–36 (2017).
Peng, Y. et al. Unimodal response of soil methane consumption to increasing nitrogen additions. Environ. Sci. Technol. 53, 4150–4160 (2019).
Saunois, M. et al. The global methane budget 2000–2017. Earth Syst. Sci. Data 12, 1561–1623 (2020).
Davidson, E. A., Trumbore, S. E. & Amundson, R. Soil warming and organic carbon content. Nature 408, 789–790 (2000).
Knorr, W., Prentice, I. C., House, J. I. & Holland, E. A. Long-term sensitivity of soil carbon turnover to warming. Nature 433, 298–301 (2005).
Hagedorn, F., Mulder, J. & Jandl, R. Mountain soils under a changing climate and land-use. Biogeochemistry 97, 1–5 (2010).
Prietzel, J., Zimmermann, L., Schubert, A. & Christophel, D. Organic matter losses in German Alps forest soils since the 1970s most likely caused by warming. Nat. Geosci. 9, 543–548 (2016).
Nottingham, A. T., Meir, P., Velasquez, E. & Turner, B. L. Soil carbon loss by experimental warming in a tropical forest. Nature 584, 234–237 (2020).
Murguia-Flores, F., Ganesan, A. L., Arndt, S. & Hornibrook, E. R. C. Global uptake of atmospheric methane by soil from 1900 to 2100. Glob. Biogeochem. Cycles 35, 1–15 (2021).
Van Bemmelen, J. Über Die Bestimmung Des Wassers, Des Humms, Des Schwefels, Der in Den Colloïdalen Silikaten Gebundenen Kieselsäure, Des Mangans U. S. W. Im Ackerboden. Die Landwirthschaftlichen Vers.-Stationen 37, 279–290 (1890).
Heaton, L., Fullen, M. A. & Bhattacharyya, R. Critical analysis of the van bemmelen conversion factor used to convert soil organic matter data to soil organic carbon data: comparative analyses in a UK loamy sand soil análise crítica do fator de conversão van bemmelen usado para converter dados de mat. Espaço Aberto 6, 35–44 (2016).
Wang, Y., Li, Y., Ye, X., Chu, Y. & Wang, X. Profile storage of organic/inorganic carbon in soil: from forest to desert. Sci. Total Environ. 408, 1925–1931 (2010).
Alvarez, R. et al. Land use effects on soil carbon and nitrogen stocks and fluxes in the Pampas: impact on productivity. Soil Carbon: Types, Manag. Pract. Environ. Benefits (2014).
Zhuang, Q. et al. CO2 and CH4 exchanges between land ecosystems and the atmosphere in northern high latitudes over the 21st century. Geophys. Res. Lett. 33, 2–6 (2006).
Duan, Q. Y., Gupta, V. K. & Sorooshian, S. The anodic dissolution of gold in alkaline cyanide solutions. Influence of impurities in solution on rate of process at low overpotentials. J. Optim. Theroy Appl. 76, 501–521 (1993).
Hengl, T. et al. SoilGrids250m: global gridded soil information based on machine learning. PLoS ONE 12, e0169748 (2017).
Lee, J. et al. Soil organic carbon is a key determinant of CH4 sink in global forest soils. Purdue university research repository. https://doi.org/10.4231/8K7W-NF84 (2023).
Acknowledgements
This study was supported by the Korea Forest Service (2017096A001719BB01), Ministry of Science and ICT of Korea (NRF-2020M1A5A1110494, NRF-2020R1I1A2072824), Ministry of Environment of Korea (2022003640002), and the Ministry of Education of Korea (NRF-2019H1A2A1076239, NRF-2021R1A6A3A03039376, NRF-2019R1A6A 3A01091184, NRF- 2022R1I1A1A01071925).
Author information
Authors and Affiliations
Contributions
J.L. and H.K. conceived and co-designed the study. J.L. conducted field experiments and meta-analysis, and led the writing of the paper. H.K. contributed to writing. Y.O. and Q.Z. built the model. Y.O. conducted the model runs. J.Y., Y.Y., and J.K. contributed to formal analysis and visualization. S.T.L. and Y.O.S. contributed to field survey and writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Karen Lloyd, Simeon Smaill and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, J., Oh, Y., Lee, S.T. et al. Soil organic carbon is a key determinant of CH4 sink in global forest soils. Nat Commun 14, 3110 (2023). https://doi.org/10.1038/s41467-023-38905-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-023-38905-8
This article is cited by
-
Fertilization-induced reactive nitrogen gases and carbon dioxide emissions: insight to the carbon-nitrogen cycles
Sustainable Environment Research (2023)
-
Arctic soil methane sink increases with drier conditions and higher ecosystem respiration
Nature Climate Change (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.