Abstract
Mosquito-borne dengue (DENV) and Zika (ZIKV) viruses originated in Old World sylvatic (forest) cycles involving monkeys and canopy-living Aedes mosquitoes. Both viruses spilled over into human transmission and were translocated to the Americas, opening a path for spillback into Neotropical sylvatic cycles. Studies of the trade-offs that shape within-host dynamics and transmission of these viruses are lacking, hampering efforts to predict spillover and spillback. We infected a native, Asian host species (cynomolgus macaque) and a novel, American host species (squirrel monkey) with sylvatic strains of DENV-2 or ZIKV via mosquito bite. We then monitored aspects of viral replication (viremia), innate and adaptive immune response (natural killer (NK) cells and neutralizing antibodies, respectively), and transmission to mosquitoes. In both hosts, ZIKV reached high titers that translated into high transmission to mosquitoes; in contrast DENV-2 replicated to low levels and, unexpectedly, transmission occurred only when serum viremia was below or near the limit of detection. Our data reveal evidence of an immunologically-mediated trade-off between duration and magnitude of virus replication, as higher peak ZIKV titers are associated with shorter durations of viremia, and higher NK cell levels are associated with lower peak ZIKV titers and lower anti-DENV-2 antibody levels. Furthermore, patterns of transmission of each virus from a Neotropical monkey suggest that ZIKV has greater potential than DENV-2 to establish a sylvatic transmission cycle in the Americas.
Similar content being viewed by others
Introduction
Spillover of zoonotic, arthropod-borne viruses (arboviruses) into humans is accelerating, with outcomes ranging from dead-end infections to pandemics1. Because of the unparalleled mobility of humans, these viruses are being transported across the world, creating the potential for spillback into novel wildlife reservoirs2. Such translocation is not new: yellow fever virus (YFV), which originated in a sylvatic cycle in the forests of Africa involving non-human primates (NHPs) and arboreal mosquitoes, was carried via sailing ships to the neotropics centuries ago3,4. Soon after, YFV established urban transmission in the Americas and spilled back into a sylvatic cycle maintained in neotropical primates and mosquitoes that plagues South America to present day3,4. The four serotypes of dengue virus (DENV-1-4) underwent a similar journey, albeit their ancestral sylvatic cycles occur in Asia, but compelling evidence of a sylvatic cycle of DENV in the Americas has never been detected despite being actively sought5,6,7. Thus, when ZIKV was detected in the Americas in 2015, there was great concern, but equally great uncertainty, about its potential to establish a neotropical sylvatic cycle8,9,10. While multiple instances of ZIKV infection in neotropical primates have now been documented11,12, the potential for such spillback events to launch a sylvatic cycle remains unclear. Furthermore, the sylvatic cycles of DENV and ZIKV persist in Asia and Africa, where both continue to spill over into humans and are sometimes translocated to new continents, e.g.13,14,15,16,17,18,19.
Mathematical models using within-host viral dynamics to predict between-host transmission are key for predicting arbovirus spillover, spread, and spillback20. These models assume within-host trade-offs that influence virus transmission21, particularly the trade-off between instantaneous pathogen transmission and infection duration22,23. Host mortality is often invoked in theoretical studies as the mechanism that regulates infection duration, however most pathogens are cleared by the immune response. Indeed, for sylvatic DENV and ZIKV there is no evidence of disease in infected Old World monkeys24,25,26, although sylvatic ZIKV was first isolated in Uganda from a febrile sentinel rhesus macaque, an Asian species imported to Africa for viral surveillance16, and poses similar risks to the developing fetus in Old World monkeys as do human-endemic lineages of the virus27,28,29. Thus, transmission of these viruses likely depends on the trade-off between the magnitude of viral replication and immune clearance30.
Studies of replication-clearance trade-offs in arboviruses are scarce. The majority of publications on the intra-host dynamics of arboviruses during experimental infection of vertebrate reservoir hosts found an inverse relationship between the magnitude (peak titer) of infection and duration of infection31,32. However, most of these studies delivered virus to the host via needle injection, which differs from natural transmission via mosquito bite33,34,35. We formulated a dynamical model comparing the effect of a “tortoise” strategy of low magnitude, long-duration viremia and a “hare” strategy of short-duration, high-magnitude viremia and showed that arboviruses adopting a tortoise strategy had higher rates of persistence in both host and vector populations32. Ben-Shachar and Koelle36 investigated the replication-clearance trade-off in DENV using within-host simulation models based on data from a human cohort study in which mosquitoes fed on infected individuals, and found that a replication-clearance trade-off selects for intermediate DENV virulence. Importantly for the current study, they invoked natural killer (NK) cells to represent the innate immune response driving this effect.
In the current study, we investigated trade-offs between replication and clearance of sylvatic DENV and ZIKV, as well as specific immune responses associated with these trade-offs and potential differences in trade-offs between native and novel NHP hosts. This study tested three a priori hypotheses, of which the first is fundamental and the latter two consider mechanism and evolutionary context, respectively: Hypothesis 1 - sylvatic arboviruses experience a replication-clearance trade-off in both native and novel hosts, as evidenced by a positive relationship between the dose of virus delivered and peak virus titer, a positive relationship between virus titer and transmission to mosquitoes, and a negative relationship between peak virus titer and duration of viremia; Hypothesis 2 - NK cells can serve as a proxy for the innate immune responses that shape this trade-off, as evidenced by a negative relationship between NK cell mobilization immediately post-infection (pi) and peak virus titer as well as levels of neutralizing antibody, and Hypothesis 3 - sylvatic arboviruses have achieved an optimal replication-clearance trade-offs in native hosts but have not had evolutionary opportunity to reach this optimum in novel hosts, resulting in less transmission from novel hosts.
Results
Overview of experimental design
The putative native host in this study was the cynomolgus macaque, Macaca fascicularis. Rudnick and colleagues isolated sylvatic DENV from sentinel cynomolgus macaques held in the forest canopy in Malaysia and also showed that ≥ 50% of wild, forest-living cynomolgus macaques in Malaysia were seropositive for at least one serotype of DENV37,38,39. Other studies in Asia have also detected seropositive cynomolgus macaques (reviewed in ref. 40). The role of NHPs as hosts for ZIKV in Asia, and indeed the existence of an Asian sylvatic ZIKV cycle, is murkier because it is less studied. Wikan and Smith41 have proposed that the sparsity of human ZIKV infections is Asia is consistent with spillover from a sylvatic cycle. Of 234 wild cynomolgus macaques tested in Malaysia42, 1.3% were seropositive for ZIKV, and, in a singular case, an Australian traveler became infected with ZIKV after being bitten by a cynomolgus macaque in Indonesia43. Moreover, machine learning models have pinpointed cynomolgus macaques as having the highest risk of all primate species worldwide to serve as a host for ZIKV44.
The novel host was the neotropical squirrel monkey, Saimiri boliviensis boliviensis, which machine learning models have identified as a high risk for spillback of ZIKV44. The two sylvatic viruses were DENV-2 strain P8-1407 and ZIKV strain DakAR 41525; the former was isolated from a sentinel cynomolgus macaque in the forest canopy in Malaysia37,38,39 and the latter was isolated from an Aedes africanus, a primatophilic mosquito species that is generally confined to forest habitats45, in Senegal. Virus was delivered by infected Ae. albopictus mosquitoes, which are native to Asia but distributed across much of the world, a likely bridge vector for spillover and spillback of both DENV and ZIKV46, and a vector in outbreaks of sylvatic ZIKV in Gabon47.
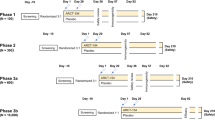
Figure 1 shows the experimental design for this study. To infect macaques, cartons of either 1 (low dose, n = 5 monkeys) or 10 (high dose, n = 5 monkeys) Ae. albopictus that had been intrathoracically (IT) inoculated with a sylvatic Malaysian strain of DENV-2, or 15 (high dose, n = 3 monkeys) Ae. albopictus IT inoculated with a sylvatic African strain of ZIKV, or 10 uninoculated, control Ae. albopictus (n = 3 monkeys) were placed upon a monkey’s ear for approximately 10 minutes. Engorged mosquitoes were separated, incubated for 2 days, and force-salivated to estimate dose of virus delivered. Squirrel monkeys were infected with either a high dose (15 mosquitoes) of a sylvatic Malaysian strain of DENV-2 (n = 10 monkeys) or a sylvatic African ZIKV strain (n = 10 monkeys) or control mosquitoes (n = 4 monkeys). Because the small size of squirrel monkeys constrained blood draw frequency, monkeys were assigned to one of two cohorts and bled prior to infection and on alternating days in the first week pi. NK cells, neutralizing antibody titers via plaque reduction neutralization titers (PRNTs), weight and temperature were measured on designated days prior to and post-infection (dpi). On designated dpi, uninfected Ae. albopictus were allowed to feed on each animal to monitor transmission.
Blood samples were used to measure viremia, natural killer (NK) cells, and neutralizing antibody as plaque reduction neutralization titer (PRNT). Monkey body temperature was monitored continuously via implanted transponder and weight was monitored on designated days. Infection and monitoring were similar in macaques and squirrel monkeys, save that: (i) the sample size of infected and uninfected mosquitoes was increased from 10 in DENV-2 infected and control macaques to 15 in ZIKV-infected macaques and all squirrel monkeys, (ii) stool was collected daily from macaques to screen for occult blood; stool was not collected from squirrel monkeys due to pair housing of animals, (iii) mosquitoes that fed on infected squirrel monkeys on dpi 3 and 4 were force-salivated after a 14 day incubation, but this was not done in macaques, and (iv) squirrel monkeys were euthanized at the conclusion of the experiment and necropsies were conducted; euthanasia was not performed on macaques. In one instance, blood sampling (dpi 7) and mosquito feeding (dpi 8) occurred on different days for macaques. Images used under license from Shutterstock.com.
All raw data from macaques and squirrel monkeys infected with DENV-2 or ZIKV, save for temperature and PRNT values against non-infecting viruses, are provided in Supplementary Datasets 1, 2, 3, and 4, respectively. Following infection with either DENV-2 or ZIKV, body temperatures of macaques48,49 and squirrel monkeys50 stayed within the normal range of each species (Fig. S1) and there were no differences among treatment groups, nor were there any differences among treatment groups in changes in body weight change (Fig. S2).
Sylvatic DENV-2 delivered by mosquito bite to both macaques and squirrel monkeys resulted in nearly 100% seroconversion with high neutralizing antibody titers
In IT-inoculated mosquitoes used to deliver DENV-2 to macaques, virus was not initially detectable via serial dilution and immunostaining of saliva samples in monolayers of mosquito cells. After saliva were subjected to one blind passage in mosquito cells, the virus was detected in a subset of salivations (Table 1). Thus, we used the number of mosquitoes salivating detectable virus as a proxy for the total dose of DENV delivered to macaques. For macaque, UG253A, the single mosquito that fed produced no detectable virus in the saliva, and UG253A never produced detectable viremia or seroconverted (defined here as a PRNT50 titer, the most permissive cutoff, ≥ 20; Supplementary Dataset 5). Hence UG253A was reassigned, post hoc, to the control treatment. Macaque SB393 received a single bite from a mosquito that did not salivate detectable virus, but this macaque seroconverted, demonstrating that it had become infected. Of the remaining eight macaques in the low and high-dose treatments, seven seroconverted, but one, BC407, did not, even though it received a bite from a mosquito with detectable virus in the saliva (Table 1). All statistical analyses of neutralizing antibody levels were conducted using PRNT80 values, a more stringent measure consistent with our previous study of sylvatic DENV-2 in African green monkeys24. The harmonic mean PRNT80 value (values <limit of detection (LOD) set at 19) for macaques was 69.7 (Table 1, Fig. 2A) Of the mosquitoes used to deliver DENV-2 to squirrel monkeys, the virus was detectable in the saliva of 30/45 IT inoculated mosquitoes by titer of collected saliva or passaged saliva (Table 2). All squirrel monkeys seroconverted (PRNT50 > LOD), with a harmonic mean PRNT80 value of 91.4 (Table 2, Fig. 2B). There were no notable findings in necropsy reports for squirrel monkeys infected with DENV-2.
A Top panel shows mean peak titer ± 1 SE of DENV-2 and ZIKV and bottom panel shows mean PRNT80 ± 1 SE, in cynomolgus macaques, N = 13 (10 DENV-2 and 3 ZIKV from a single independent experiment each) biologically independent animals in each panel. B Top panel shows higher mean peak titer ± 1 SE of ZIKV than DENV-2 (2-tailed t test, DF = 17, t = 7.18, P < 0.0001), and bottom panel shows lower mean PRNT80 ± 1 SE against ZIKV than DENV-2 (Mann–Whitney U test, DF = 1, chi-squared = 7.65, P = 0.006) in squirrel monkeys, N = 19 (10 DENV-2 and 9 ZIKV from a single independent experiment each) biologically independent animals in panel A and 18 (10 DENV-2 and 8 ZIKV from a single independent experiment each) biologically independent animals in panel (B), no adjustment was made for multiple tests. C Mean ± 1 SE of early % NK cells in macaques or squirrel monkeys do not differ between viruses or between infected and control animals (2-tailed ANOVA, DF = 4, F = 0.44, P = 0.78; note that ZIKV in cynomolgus macaques excluded from analysis due to small sample size), N = 105 total PBMCs per animal for 40 biologically independent animals from a single independent experiment for each virus in each monkey species. D Early % NK cells are negatively associated with PRNT80 values for macaques (triangles) and squirrel monkeys (circles) infected with DENV-2, shaded band shows 95% confidence interval around the line of best fit (linear model, 2-tailed ANOVA, DF = 1, F = 10.8, P = 0.005), N = 20 biologically independent animals, with 105 total PBMCs per animal used to estimate % NK cells. E negative relationship between peak viremia and duration of viremia for macaques (triangles) and squirrel monkeys (circles) infected with ZIKV, shaded band shows 95% confidence interval around the line of best fit (2-tailed ordinal logistic regression, DF = 1, chi-squared = 11.1, P = 0.0009, R2 = 0.17), N = 12 biologically independent animals. F Negative relationship between early % NK cells and peak viremia for macaques (triangles) and squirrel monkeys (circles) infected with ZIKV, shaded band shows 95% confidence interval around the line of best fit (2-tailed linear regression, DF = 1, F = 13.4, P = 0.004, adjusted R2 = 0.53), N = 12 biologically independent animals. No adjustment for multiple testing was made for any of the tests listed above. Images used under license from Shutterstock.com. Source data are provided as a Source Data file.
Sylvatic DENV-2 replicated to low levels in macaques and squirrel monkeys and showed unexpected patterns of transmission
We initially predicted, based on hypothesis 1, that increasing dose of virus would lead to increasing peak viremia, which in turn would be associated with greater instantaneous transmission to mosquitoes but a shorter duration of viremia. It was not possible to test most of these predictions in macaques infected with DENV-2, as viremia was detected from serum via cell culture-based assay in only three macaques, one from the low dose and two from the high dose treatment (Table 1, Fig. 3A). Furthermore, only three macaques transmitted DENV-2 to mosquitoes, all from the high dose treatment (Table 1, Fig. 3A, Supplementary Dataset 6). Intriguingly, virus was not detected from raw or passaged serum in any macaque on the day of transmission to mosquitoes or, in the case of macaques FR840 and BC167, at any point during the study. It was possible to analyze the number of infectious bites received and the likelihood of becoming viremic, as evidenced by detectable virus in raw or passaged serum or mosquitoes (n = 5 viremic macaques total). Consistent with Hypothesis 1, detectably viremic animals received a significantly greater number of infectious bites than animals that were not detectably viremic (logistic regression, DF = 1, n = 9, χ2 = 4.63, P = 0.03).
Sylvatic dengue virus serotype 2 (DENV-2) replicates to low levels and transmits rarely to Ae. albopictus in infected: (A) cynomolgus macaques and (B) squirrel monkeys. Points for mosquito infections that occurred from monkeys without detectable viremia are placed at arbitrary positions below the limit of detection (LOD) for visualization only. Viremia was monitored by direct titration of serum as well as one passage of serum followed by titration; transmission was monitored by feeding cartons of 10-15 uninfected mosquitoes on each monkey; note that usually fewer than the full complement of mosquitoes engorged. Source data are provided as a Source Data file. Images used under license from Shutterstock.com.
Only three squirrel monkeys produced viremia that could be detected from serum (Table 2, Fig. 3B). Two monkeys that produced viremia detectable in serum transmitted DENV-2 to mosquitoes, but only when their viremia was at its lowest point. Another four monkeys that did not produce detectable viremia nonetheless transmitted DENV-2 to mosquitoes (Table 2, Fig. 3B). Notably, for mosquitoes fed on both macaques and squirrel monkeys, DENV-2 could be detected more often in legs than in bodies (Supplementary Dataset 6). Contra Hypothesis 1, dose of DENV-2 delivered had no significant impact on the likelihood of squirrel monkeys becoming detectably viremic (logistic regression, N = 7, Table 2, DF = 1, χ2 = 1.0556, P = 0.30). Given the small number of monkeys for which peak titer could be quantified, we did not analyze the association of dose with peak titer. DENV-2 was not detectable in any of the salivations from the mosquitoes that fed on squirrel monkeys at 3 and 4 dpi.
Following infection with sylvatic DENV-2, higher NK cells on day 1 were associated with lower neutralizing antibody titers in both macaques and squirrel monkeys
We predicted, based on Hypothesis 2, that NK cells mobilized early in infection would show a negative relationship with peak virus titer and neutralizing antibody titer. To analyze variation in NK cells, we used the percent NK cells of the total number of peripheral blood mononuclear cells (PBMCs). After Bonferroni correction for multiple testing, in infected macaques, values for % NK cells at each dpi were significantly correlated with the following dpi, however values at D0 and D1 dpi were not significantly correlated. In infected squirrel monkeys, percent NK cells at earlier (1 or 2 dpi) and later (7 or 8 dpi) timepoints were significantly, positively correlated with each other (P = 0.005); earlier % NK cells were correlated with % NK cells prior to infection (P = 0.04), but later % NK cells were not (P = 0.21). Thus, we analyzed only early % NK cells (1 dpi for macaques or 1 and 2 dpi for squirrel monkeys, batched due to the cohort structure of the experiment).
Early % NK cells were not significantly different among control, low dose macaques (1 mosquito) and high dose macaques ( > 1 mosquito) macaques (ANOVA, DF = 2, F = 2.74, P = 0.11) (Fig. 2C), and Day 28 PRNT80 values were not significantly different between low and high dose macaques (t-test on log10 transformed data, DF = 7, t = 2.29, P = 0.056) (Table 1). Thus, we combined low and high dose macaques for analysis. Early % NK cells did not differ between control and high dose squirrel monkeys (ANOVA, DF = 1, F = 0.87, P = 0.37; note that all four control squirrel monkeys were used in comparisons with both DENV and ZIKV infected squirrel monkeys; Fig. 2C).
Due to the low level of replication of DENV-2, it was not possible to analyze peak titer for either macaques or squirrel monkeys. The effects of early % NK cells and host species, as well as the interaction between the two factors, on PRNT80 values in infected animals was tested using a linear model: the only significant effect was early % NK cells (ANOVA, DF = 1, F = 10.8, P = 0.005), and the relationship between early % NK cells and PRNT80 values was negative, as predicted (Fig. 2D).
Sylvatic DENV-2 dynamics, transmission, and neutralizing antibody were similar between native and novel hosts
Based on Hypothesis 3, we predicted that transmission of DENV-2 would be less efficient from squirrel monkeys than cynomolgus macaques. Squirrel monkeys and macaques received similar doses of DENV-2 (Supplementary Text S1.1, S1.2, S1.3). When considering only infected animals, neither early % NK cells (t test, DF = 17, t = −0.39, P = 0.70) nor PRNT80 values (t test, DF = 17, t = 0.35, P = 0.73) differed between macaques and squirrel monkeys. Viremia was too low (Fig. 2A, B) and transmission was too infrequent (Fig. 3A, B) from both macaques and squirrel monkeys for statistical comparison but did not support the claim that transmission is lower from a novel host. Intriguingly, DENV-2 transmission from both macaques and squirrel monkeys was associated with low or undetectable viremia (Fig. 4A).
Relationship between virus titer in non-human primate serum and transmission of (A) sylvatic dengue virus serotype 2 (DENV-2) or (B) sylvatic Zika virus (ZIKV) to Ae. albopictus (either body or leg infection). Curves fitted using a generalized additive model, allowing a variety of shapes, to unravel a possible pattern. Source data are provided as a Source Data file.
Sylvatic ZIKV delivered by mosquito bite to both macaques and squirrel monkeys resulted in 100% seroconversion, but antibody responses differed between the species
ZIKV was detectable in the saliva of 22/25 intrathoracially inoculated mosquitoes that fed on macaques. All three macaques seroconverted (Table 2, Supplementary Dataset 5) with a minimum mean harmonic PRNT80 of 320 (treating the > 640 value as 640) (Fig. 2A). ZIKV was detectable in the saliva of 58/70 IT inoculated mosquitoes that fed on squirrel monkeys. There was a tight relationship between number of mosquitoes salivating ZIKV and total dose delivered (DF = 1, F = 12.6, P = 0.008, adjusted R2 = 0.56, slope 0.14 (0.05; 0.22) in log10 units). All squirrel monkeys that received infectious mosquito bites seroconverted based on PRNT50 values (Table 2, Supplementary Dataset 5), but 63% produced PRNT80 levels below the LOD, with a harmonic PRNT80 of 23.6 (Fig. 2B). The small sample size of ZIKV-infected macaques (N = 3) precluded most statistical analyses, but as described below, these data could sometimes be combined with squirrel monkey data to test the central hypotheses of the manuscript. As described in Supplementary Text S2, two squirrel monkeys infected with ZIKV had to be euthanized prior to the end of the experiment following the recommendation of the head veterinarian on staff: 4683 due to cluster seizures and 4728 due to a worsening sore on the foot.
Sylvatic ZIKV dynamics were similar in macaques and squirrel monkeys and peak viremia was negatively associated with duration of viremia
All three ZIKV-infected macaques produced viremia that peaked between dpi 4 and 5 and lasted for 3 to 5 days (Table 1) and transmitted virus to a high proportion of mosquitoes during the window of viremia (Fig. 5A). All squirrel monkeys receiving bites from ZIKV-infected mosquitoes produced detectable viremia and infected mosquitoes on at least one dpi (Table 2, Fig. 5B, Fig. S3). ZIKV titer peaked between dpi 3 and 5 and viremia lasted between 3 to 7 days, although there is substantial uncertainty to these estimates given the sampling regimen.
ZIKV replicates robustly and transmits efficiently to Ae. albopictus in (A) cynomolgus macaques (n = 3 biologically independent animals) and (B) squirrel monkeys (N = 10 biologically independent animals) from a single independent experiment each. Control animals are not included in this figure. Blue lines indicate mean viremia, bands indicate ± 1 standard error (SE), and black dots indicate viremia for each individual monkey. Bars indicate mean % mosquitoes infected per monkey, and error bars indicate ± 1 standard error (SE). In panel A, the inset shows the dynamics of viremia and transmission during the window of viremia for each individual macaque. Viremia was monitored by direct titration of serum as well as one passage of serum followed by titration; transmission was monitored by feeding cartons of 15 uninfected mosquitoes on each monkey; note that usually fewer than 15 engorged. Source data are provided as a Source Data file. Images used under license from Shutterstock.com.
For analyses of the magnitude and duration of ZIKV viremia in squirrel monkeys, we chose to exclude 4683, the animal euthanized at 13 dpi (Supplementary Text S2), as we did not know whether pre-existing health problems may ultimately have led to euthanasia and may have impacted viral replication or immune parameters; however, we note below when inclusion of this animal altered determination of the significance of comparisons. Contrary to the predictions of Hypothesis 1, the total dose delivered did not affect the peak titer of ZIKV in squirrel monkeys (DF = 1, F = 0.02, P = 0.88). Since every cynomolgus macaque received a higher virus dose of ZIKV than any squirrel monkey (see below), we did not analyze data from both species combined (Supplementary Text S1.3). In agreement with the predictions of Hypothesis 1, peak virus titer was negatively associated with the duration of infection for ZIKV, albeit the range of duration was quite limited (ordinal logistic regression, DF = 1, chi-squared = 7.4, P = 0.007, R2 = 0.17). When these analyses were repeated with 4683 included, relationships were qualitatively similar, but the association between peak titer and duration of infection was no longer significant. Since both peak viremia and duration of viremia of ZIKV in macaques fell within the range of these parameters in squirrel monkeys, we also conducted an analysis of combined values across species and found a similar, negative relationship (ordinal logistic regression, DF = 1, chi-squared = 11.1, P = 0.0009, R2 = 0.17) (Fig. 2E).
Following infection with sylvatic ZIKV, higher NK cells on day 1 were associated with lower peak virus titer in both macaques and squirrel monkeys
Values for early % NK cells in ZIKV-infected macaques all fell within the range of values for control macaques (Fig. 2C). Similarly, early % NK cells did not differ between ZIKV-infected and control squirrel monkeys (DF = 11, t = −1.72, P = 0.11; Fig. 2C). Moreover, % NK cells in infected squirrel monkeys at early and later timepoints were significantly, positively correlated with each other (P = 0.005) but not with % NK cells prior to infection (P > 0.27 for both comparisons). Consistent with predictions of Hypothesis 2, higher early NK cells were associated with lower peak virus titer in squirrel monkeys (DF = 1, F = 12.6, P = 0.009, adjusted R2 = 0.59; this relationship was not significant when data from 4683 was included). Since the values for early % NK cells and peak viremia in macaques fell within the range of these parameters in squirrel monkeys, we also conducted an analysis of combined values across species and found a similar, negative relationship (DF = 1, F = 13.4, P = 0.004, adjusted R2 = 0.53) (Fig. 2F). It was not possible to analyze the association between early NK cells and neutralizing antibody titer in squirrel monkeys, as approximately half of the PRNT80 values were <LOD.
Sylvatic ZIKV transmission increased with increasing viremia, but novel host transmitted more efficiently than native hosts
In contrast to DENV-2, the likelihood of ZIKV transmission from both macaques and squirrel monkeys increased with virus titer (Fig. 4B, this analysis included 4683). Of mosquitoes that fed on dpi 3 and 4 on ZIKV-infected squirrel monkeys, one batch of mosquitoes died, but all other monkeys infected at least one mosquito that salivated detectable virus (Supplementary Dataset 6). The mean rate of virus dissemination to saliva from fed mosquitoes was 63% (±12%) (Supplementary Dataset 6).
The best dose-response fit to characterize the relationship between host viremia and disseminated ZIKV infection in mosquitoes (presence of virus in mosquito legs) was obtained using a logistic equation (Eq. S.1) and a betabinomial likelihood for both cynomolgus macaques and squirrel monkeys (Fig. 6A, Table S1). While the fits were not significantly different overall, the dose needed to infect 50% of mosquitoes (ID50) was lower for squirrel monkeys (2.53, 95% CI (1.96;3.15) than macaques (3.84 (3.35;4.31) log10pfu/ml, Fig. 6A). When using data on mosquito body infection, the fitted relationships were significantly different between species (Table S2, Fig. S4A). This is because, as with DENV-2, among mosquitoes that fed on ZIKV-infected squirrel monkeys, a much higher proportion of infections could be detected in mosquito legs than bodies. However, these proportions were relatively similar among mosquitoes fed on ZIKV-infected macaques (Supplementary Dataset 6). The parameters of fitted relationships can be found in Table S3.
A Relationship between virus titer in serum and transmission of sylvatic Zika virus (ZIKV) to legs of Ae. albopictus from cynomolgus macaques (light blue) or squirrel monkeys (dark blue). Points show raw data, with point size proportional to the number of mosquitoes tested in the batch (one day in one monkey). B–E Relationships between virus titer in serum and transmission of ZIKV to legs of Ae. albopictus from both non-human primate species combined (blue curve, repeated in B–E) or transmission to legs of Ae. aegypti from humans of (B) Dengue virus (DENV) serotype 1, (C, D) DENV serotype 2, (E) DENV serotype 4. DENV curves were fitted to data from Duong et al.51 for sub-panels (B, C, E), and to data from Long et al. 201952 for sub-panel (D). Infectious dose 50 (ID50) and confidence interval (CI) provided for each designated virus and mosquito species in log10 pfu/ml. Model selection was conducted with three competing functional forms, all with a sigmoidal shape. Curves represent the best fit, and shaded areas correspond to 95% confidence intervals of the fitting process. a CI of ID50 not provided as it was too wide to be insightful. b ID50 and CI not provided as they were not insightful. Note that data used for fitting of this DENV-2 curve was from mosquito heads rather than legs, both are markers of a disseminated infection. Source data are provided as a Source Data file.
Sylvatic DENV-2 and ZIKV dynamics differed dramatically from each other in mosquito vectors and monkey hosts
Aedes albopictus
Although Ae. albopictus were IT inoculated with the same dose of sylvatic DENV-2 and ZIKV, mean saliva titer was significantly lower in DENV-2 than ZIKV-infected mosquitoes (DENV 1.38 (1.24; 1.53), ZIKV 2.55 (2.36; 2.74) log10 pfu, P = 2.2e–4, Supplementary Text S1.2). In addition, the variability of saliva titers was also significantly lower in DENV-2 than ZIKV-infected mosquitoes (F = 12.7, P = 5e−4, Supplementary Text S1.2).
Macaques
Cynomolgus macaques received on average fewer bites from DENV-2 infected mosquitoes (mean 4.2, min–max (3–6) bites) than ZIKV-infected mosquitoes (7.3 (5–10) bites), Supplementary Text S1.1) and the saliva titers of DENV-2-infected mosquitoes were significantly lower than those of ZIKV-infected mosquitoes, as shown above. Thus, macaques received lower doses of DENV-2 than ZIKV (DENV-2 mean 1.92, min-max (1.78-2.08) log10 pfu; ZIKV 4.44 (4.41-4.48), Supplementary Text S1.3). Although we did not conduct a formal analysis due to the small sample size of ZIKV-infected macaques, ZIKV reached peak titers that were, on average, ≥3 logs higher than DENV-2 in macaques (Fig. 2A), yet early NK cells and neutralizing antibody titers were quite similar, with all values for ZIKV-infected macaques falling within the range of DENV-2 infected macaques (Fig. 2A, C).
Squirrel monkeys
Squirrel monkeys received on average fewer bites from DENV-2-infected mosquitoes (3.4 (2-6)) than ZIKV-infected mosquitoes (6.0 (1-11)) and the saliva titers of DENV-2-infected mosquitoes were significantly lower than those of ZIKV-infected mosquitoes (DENV-2 1.38 (1.24;1.53); ZIKV 2.55 (2.36;2.74) log10 pfu. Thus, squirrel monkeys received lower doses of DENV-2 than ZIKV (DENV-2 2.65 (1.60-3.37) log10 pfu; ZIKV 3.68 (2.81-4.20), Supplementary Text S1.3. Even though ZIKV peak titers in squirrel monkeys were > 3 logs higher than DENV-2 titers (t test, DF = 17, t = 7.18, P < 0.0001; Fig. 2B, Tables 1 and 2), PRNT80 values (with values < LOD set to 19) were significantly lower in ZIKV-infected than DENV-2-infected squirrel monkeys (Mann–Whitney U test, DF = 1, chi-squared = 7.65, P = 0.006) (Fig. 2B). Early % NK cells, in contrast, were similar between the two viruses (t test, DF = 17, t = 0.56, P = 0.58) (Fig. 2C). In a reduced dataset with similar range of doses for DENV and ZIKV ((2.3–3.3) log10 pfu; N = 5 DENV-2 and 4 ZIKV), peak viremia was still significantly higher in ZIKV- than DENV-2-infected squirrel monkeys (DF = 1, F = 37.83, P = 0.0005, adjusted R2 = 0.82).
Across hosts
Patterns of DENV-2 and ZIKV replication summed across hosts (Fig. S5) were dramatically different. DENV-2 sustained low but prolonged viremia while ZIKV peaked quickly and was quickly cleared. The transmission also differed, as highlighted when fitting generalized additive models to each dataset (Fig. 4, Supplementary Text S3). The smooth term associated with virus titer was non-significant for DENV (P = 0.118) and significant for ZIKV (p < 2e−16). The deviance explained by both models was 67.4% and 85.2%, respectively.
Relationship between viremia titer and transmission is similar between ZIKV in NHPs and DENV in humans
Using a common fit for both species, we compared the relationship for disseminated infection to the ones obtained using data generated by Duong et al.51 and Long et al.52 in which DENV viremia (serotypes 1, 2, and 4) in infected humans and transmission to Ae. aegypti mosquitoes (measured as infection of mosquito legs for Duong et al., mosquito heads for Long et al.) were quantified (Fig.6B–E). The ID50 for ZIKV transmission from NHPs to Ae. albopictus was substantially lower than for DENV transmission from humans to Ae. aegypti (Fig. 6B–E). We conducted a similar comparison to data on DENV transmission (all four serotypes) from infected humans to Ae. aegypti generated by Nguyen et al.53 and Long et al.52, focusing on transmission measured as infection of mosquito bodies. In this case, there was much overlap between ZIKV transmission from NHPs and DENV transmission from humans (Fig. S4B). Fitted relationships for DENV with the corresponding data are shown in Fig. S6.
Discussion
This study investigated whether the replication and clearance of arboviruses are locked into an immunologically-mediated trade-off, whether NK cells may be associated with this trade-off, and whether the nature of the trade-off differs between native and novel NHP hosts. If higher virus replication does drive faster clearance, we predicted that increasing viral dose would result in higher peak virus titer, that transmission would increase with virus titer, and that peak titer would be inversely related to duration of viremia. To test these predictions, we exposed cynomolgus macaques and squirrel monkeys to the bites of mosquitoes infected with either sylvatic DENV-2 or sylvatic ZIKV and monitored viremia as well as forward transmission to uninfected mosquitoes. Virus dose was estimated by summing the amount of virus in forced salivations from each mosquito that had fed upon a particular animal. While forced salivations likely underestimate the absolute amount of virus delivered54,55, they should reflect the relative dose of virus delivered. Our previous study of mosquito delivery of ZIKV to mice found a highly significant increase in neutralizing antibody titer with increasing virus dose estimated via forced salivations56.
Eight macaques and ten squirrel monkeys bitten by sylvatic DENV-2-infected mosquitoes seroconverted and produced robust neutralizing antibody titers. However, viremia was detected via cell culture assay in only three macaques and three squirrel monkeys, and only on one or a few days per animal. Intriguingly, nine animals in total transmitted DENV-2 to mosquitoes, but transmission usually occurred when the virus was not detected in the blood. This “decoupling” of transmission from DENV viremia has also been observed in studies in which Ae. aegypti were fed on viremic humans: Lambrechts et al.57 found that, at the same level of viremia, humans infected with DENV-2 transmitted to more mosquitoes in earlier days after onset of illness than later days, and Duong et al.51 found that people infected with DENV transmitted more efficiently to mosquitoes when they were asymptomatic and pre-symptomatic than when they were symptomatic, independent of viremia level. A number of host or viral products could be responsible for curbing transmission, such as blood biochemistry, immune factors, or viral proteins. Our data suggest that whatever is responsible is rather labile, as, in some monkeys (for example, macaque FR423A and squirrel monkey 4516), viremia could be detected from blood on the days before and after the monkey transmitted virus to mosquitoes but not on the day of transmission itself. With respect to our central hypothesis, it was not possible to analyze peak titer in either species due to the small number of monkeys with quantifiable viremia; we did find that a higher number of infectious mosquito bites increased likelihood of infection of macaques but not squirrel monkeys.
In contrast to DENV-2, sylvatic ZIKV reached high titers in both host species and transmission to mosquitoes was positively related to virus titer. In infected mosquitoes, ZIKV disseminated efficiently to the saliva. Previous studies have shown similar levels of infection, dissemination and salivation of ZIKV strain 41525 in multiple strains of both Ae. albopictus and Ae. aegypti using multiple infection paradigms58,59,60, suggesting that our results for this ZIKV strain are generalizable across domestic and peridomestic Aedes vectors. Although both host species supported high levels of viremia and transmission of ZIKV, cynomolgus macaques raised robust neutralizing antibody titers that were equivalent to those produced against DENV-2, while squirrel monkeys raised neutralizing antibody titers that were significantly lower than those of DENV-2, and often below the LOD when a stringent cutoff (PRNT80) was used. Similarly, studies on Ross River virus (reviewed in61) showed that experimentally infected bats produced no detectable viremia but nonetheless infected mosquitoes, while corellas (Cacatoes correla) became viremic but did not seroconvert. In combination, these findings highlight the complexity of using serological data to discern the role of species in virus transmission cycles62,63.
A trade-off between magnitude and duration of infection was evident both within and between viruses. For ZIKV infections, maximum virus titer was associated with a shorter duration of infection. Comparing between viruses, ZIKV titers were significantly higher than DENV-2 titers in both cynomolgus macaques and squirrel monkeys, but the monkeys infected with ZIKV were viremic for only three to seven days while the monkeys with detectable DENV-2 viremia were viremic over twelve to fourteen days. Because higher titers of ZIKV were expectorated into mosquito saliva, it is not entirely possible to determine whether this difference in dynamics is due to the virus per se or to the higher dose of ZIKV delivered, although analysis of a subset of the monkeys in which dose was equivalent still found higher peak titers of ZIKV. However, since the ZIKV produced higher virus levels in saliva than DENV after equivalent amounts of virus were IT injected into mosquitoes, and since these results are consistent with a previous study by Chaves et al.64 in which Ae. aegypti were infected with both viruses via membrane feeding, we consider the high dose of ZIKV intrinsic to the phenotype of this strain. Moreover, the low levels of DENV-2 replication in this study are consistent with results from our previous study of the replication of an African sylvatic DENV-2 strain in African green monkeys (Chlorocebus sabaeus) following needle delivery of a high dose (105 pfu) of the virus24. Interestingly, the effect of virus titer on transmission to mosquitoes was different for DENV-2 and ZIKV, with DENV-2 transmission occurring when viremia was low or just above the LOD, while ZIKV transmission was positively associated with titer. Thus, it seems that sylvatic DENV-2 follows a “tortoise” strategy in squirrel monkeys, while sylvatic ZIKV follows a “hare” strategy32.
A previous mathematical model of the replication-clearance trade-off in DENV utilized NK cells as a proxy for the innate immune response to the virus36. We predicted that if NK cells are coupled to the immune responses shaping this trade-off, then higher mobilization of NK cells early in infection should suppress peak virus titer, which should in turn lower neutralizing antibody titers. For sylvatic DENV-2, peak titer could not be analyzed, but as the percentage NK cells out of total PBMCs on early dpi increased, PRNT80 values measured at 28 dpi decreased in both species, consistent with our prediction. The relationship of peak ZIKV viremia with PRNT80 values could not be analyzed, but peak ZIKV titers were negatively associated with higher early NK cells in both host species. Thus, there is substantial support that NK cells may play a direct role in shaping the replication of these viruses65 or may be linked to another pathway that enacts this function.
Third, we predicted that transmission from novel hosts would be low relative to native hosts, reflecting the co-adaptation of viruses with their native hosts. Infection of both cynomolgus macaques and squirrel monkeys with sylvatic DENV-2 via mosquito bite resulted in muted virus replication. Previous studies utilizing needle delivery of human-endemic DENV have reported peak virus titers about three orders of magnitude lower in cynomolgus macaques than neotropical primates31. Prima facie, these latter data would suggest that transmission is actually more efficient from neotropical monkeys than Old World monkeys. However, our startling finding that sylvatic DENV transmission to mosquitoes occurred predominantly at undetectable or barely detectable viremia indicates a more complex relationship between viremia and transmission. Nonetheless, transmission of sylvatic DENV-2 from both macaques and squirrel monkeys was infrequent. This paucity of transmission could be the byproduct of a mismatch in the genetic interaction between sylvatic DENV-2 and Ae. albopictus Galveston66 or could be an artifact of the passage history of this virus strain. Moreover, it is possible that sylvatic mosquito species, such as Aedes niveus species, are more susceptible to sylvatic DENV. Further study of transmission to sylvatic African, Asian and American mosquitoes would certainly be welcome; but few of these species are maintained in colonies. Alternatively, the low level of transmission could suggest that cynomolgus macaques are not actually a reservoir host for this virus. The paradigm that Asian NHPs are the reservoir of sylvatic DENV, rather than an amplifying host or a dead-end host67, rests on scanty evidence, including serosurveys and isolation of sylvatic DENV from sentinel cynomolgus macaques37,38,39. On the other hand, the lack of pathogenesis of sylvatic DENV-2 in macaques, the long duration of viremia, and indeed the low magnitude of viremia itself may be evidence that macaques are reservoir hosts68,69. Going forward, we will attempt to parameterize mathematical models with these data to assess whether or not cynomolgus macaques could be expected to sustain transmission of sylvatic DENV.
We found that ZIKV replicated to approximately a log higher titer in squirrel monkeys than macaques and, controlling for titer, transmitted to mosquitoes more efficiently from squirrel monkeys than macaques. Similarly, a Puerto Rican strain of ZIKV delivered by needle replicated to peak titers that were 2–3 orders of magnitude higher in neotropical marmosets and tamarins compared to rhesus and cynomolgus macaques70. Dudley et al.33 found that, compared to needle delivery, mosquito bite delivery of a Puerto Rican strain of ZIKV to rhesus macaques delayed the onset of viremia and delayed peak of viremia from day 3 to day 5 pi, but for both methods of delivery, almost none of the Ae. aegypti fed upon these monkeys became infected. We reported similarly low transmission from cynomolgus macaques infected with a Cambodian strain of ZIKV via needle delivery and fed upon by Ae. aegypti71. Thus, opposite our initial hypothesis, evidence to date suggests that neotropical primates support similar or higher levels of DENV and ZIKV transmission than their Old World counterparts. Moreover, similar to previous findings by Carrington et al.72, we detected little difference in replication dynamics of DENV-2 and ZIKV after delivery by mosquito bite and studies in which the viruses were delivered by needle.
Finally, our data offer insights into two other central questions: why has DENV never spilled back into a sylvatic cycle in the Americas and what is the likelihood that ZIKV will do so? The rarity of transmission of sylvatic DENV-2 from squirrel monkeys to Aedes albopictus suggests that the virus cannot gain a foothold in neotropical NHPs and sylvatic mosquitoes, albeit testing more strains of mosquitoes and monkeys, as well as modeling, are still needed. In contrast, the viremia-transmission curves of sylvatic ZIKV in squirrel monkeys were similar to those derived from studies of DENV-1-4 in humans51,52,53. While additional modeling will be needed to draw inferences about the epidemic potential of Ae. albopictus for ZIKV in neotropical monkeys, as was done by Lequime et al. for a human population73, our empirical data suggest that the establishment of a neotropical, enzootic cycle is substantially more likely for ZIKV than DENV.
Methods
Ethics statement
Our study complies with all relevant ethical guidelines and all community standards for containment of infected arthropod vectors; all procedures conducted on non-human primates were approved via UTMB Institutional Animal Care and Use Committee (IACUC) protocol 1912100, approved on December 1, 2019.
Viruses and cell lines
African green monkey kidney cells (Vero, catalog number CCL-81) and larval Ae. albopictus cells (C6/36, catalog number CRL-1660) were purchased from the American Type Culture Collection (ATCC, Bethesda, MD, USA). Vero cells were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM, ThermoFisher Scientific, Waltham, MA, USA) supplemented with 5% (v/v) heat-inactivated fetal bovine serum (FBS, Atlanta Biologicals, Flowery Branch, GA, USA), 1% (v/v) Penicillin–Streptomycin (P/S, ThermoFisher Scientific, Waltham, MA, USA; 100 U/mL and 100 μg/mL, respectively) in a humidified 37 °C incubator with 5% CO2. C6/36 cells were maintained in DMEM supplemented with 10% (v/v) heat-inactivated FBS, 10% (v/v) tryptose phosphate broth (TBP, Sigma-Aldrich, St. Louis, MO, USA), 1% (v/v) P/S in a humidified 28 °C incubator with 5% CO2.
The following viruses with the following passage histories were utilized to infect monkeys: sylvatic DENV-2 strain P8-1407 (suckling mouse (SM) 3 passages, C6/36 5 passages) and sylvatic ZIKV strain DakAR 41525 (AP-61 1 passage, C6/36 2 passages, Vero 6 passages). The DENV-2 stock was 3 C6/36 passages removed from a stock sequenced by Vasilakis et al.74 (Genbank ID KU95559.1), and the ZIKV stock was 3 C6/36 passages removed from the stock sequenced by Ladner et al.75 (Genbank ID EF105379.1). A diagnostic region of the envelope gene of each working stock of virus (nucleotides 1435–1744 for DENV-2 and 2163–2498 for ZIKV) were sequenced and showed no changes relative to the reference sequence. Pre-study blood samples were screened via PRNT assays against DENV-1 Hawaii, DENV-2 NGC (macaques) or DENV-2 16681 (squirrel monkeys), DENV-3 H87, DENV-4 Dominica, ZIKV PRVABC-59, and YFV 17D; all were negative (Supplementary Dataset 5; post-infection PRNTs were screened against DENV-2 NGC (macaques), DENV-2 16681 (squirrel monkeys) or ZIKV PRVABC-59 (both species), as appropriate. The switch between DENV-2 strains was necessitated by a freezer failure, but in our experience, should have little impact on PRNT values.
Virus quantification from cell culture supernatants, NHP sera and mosquitoes
Virus stocks and NHP sera were titered in Vero cells while mosquito samples were titered in C6/36 cells. All NHP sera were titered starting at a 1:10 dilution and were also passaged once in Vero cells for 5–7 days as follows: generally, 500 μL of 1:10 diluted NHP serum in Vero cell media was used to infect one well of confluent Vero cells in 6-well plates, plates were gently rocked for two hours before adding media to a total volume of 3 mL and then plates were incubated for 4-6 days, cell supernatants were clarified by centrifugation, stabilized in 1X SPG (final concentration: 218 mM sucrose, 6 mM L-glutamic acid, 3.8 mM monobasic potassium phosphate, and 7.2 mM dibasic potassium phosphate, pH 7.2), and stored at -80˚C. For a relatively small number of serum samples with volumes <50 μL, dilutions of 1:20, 1:40 and 1:100 were used as necessary. Resulting cell supernatants were titered in Vero cells. Fifty µl of saliva samples were titered and were also passaged once in one well of a 96-well plate of confluent C6/36 cells for 5-7 days, and the resulting cell supernatants were titered in C6/36 cells.
Viral quantification of cell-free viral stocks for infection and PRNT assays were conducted using standard infectious assay71. Briefly, viral stocks underwent 10-fold serial dilutions using dilution media (DMEM, 2% FBS, and 1% P/S) in sterile 96-well tissue culture plates. Dilutions were added to 85-95% confluent monolayers of either Vero or C6/36 cells, as specified, in 12 or 24 well tissue culture plates. Dilutions were allowed to adsorb onto monolayers for 1 hour in a humidified 37 °C (Vero) or 28 °C (C6/36) incubator with 5% CO2 and were rocked every 15 minutes to prevent drying of the monolayers. Following adsorption, monolayers were overlayed using a solution of DMEM containing 3% FBS, 1% Pen–Strep, 1.25 μL/mL amphotericin B, and 0.8% weight/vol methylcellulose. The overlayed plates were incubated for 5 days in a humidified 37 °C incubator with 5% CO2; then, each well was washed twice with PBS and fixed for a minimum of 30 min in an ice-cold solution of methanol:acetone, 1:1, vol/vol. The fixative was removed, and the plates were air dried. Following complete air drying, the plates were washed with PBS and then blocked with 3% FBS in PBS, followed by an overnight incubation with mouse hyperimmune serum against DENV-2 NGC (MIAF T-35432, generated 12/10/2014) or mouse hyperimmune serum against ZIKV strain MR-766 (MIAF T-36846 generated 07/19/2017) or pan-flavivirus monoclonal antibody 4G2. All three antibodies were used at a 1:2000 dilution; MIAF T-35432 and MIAF T-36846 were obtained from the World Reference Center on Emerging Viruses and Arboviruses (WRCEVA) at University of Texas Medical Branch while 4G2 was obtained from VWR (Radnor, PA, catalog number 76005-718). The plates were then washed with PBS, followed by incubation with a goat anti-mouse secondary antibody conjugated to horseradish peroxidase (KPL, Gaithersburg, MD, USA, catalog number 5220-0341) diluted 1:2000 in blocking solution. The plates were washed with PBS, after which an aminoethylcarbazole solution (Enzo Diagnostics, Farmingdale, NY, USA) prepared according to the manufacturer′s protocol was added, and the plates were incubated in the dark. Development was halted by washing in tap water, and the plates were allowed to air dry at room temperature before scoring. Virus quantification from monkey sera and mosquito samples was conducted using standard infectious assays, which are extremely similar to those described above save that titration was conducted in 24-well plates, plates were fixed in 90% methanol at room temperature, incubated with primary antibodies for one hour, and developed using TrueBlue (SeraCare, Milford, MA, USA).
Mosquito strains, maintenance, infection, and forced salivation
Aedes albopictus were field collected in Galveston Texas in the summer of 2018 and utilized to establish a continuous colony. All mosquitoes utilized over the course of these experiments were derived from this original colony. For infection of cynomolgus macaques with DENV-2 and control, Ae. albopictus Galveston F12 were used, while for infection of cynomolgus macaques with ZIKV and squirrel monkeys with both DENV-2 and ZIKV, Ae. albopictus Galveston F14 were used. All mosquitoes were maintained and/or utilized in Biosafety Level 2 facilities (Arthropod or Animal Containment) following standard procedures76. After hatching, larvae were maintained in water in plastic containers, and then transferred after eclosion into 0.5 L cardboard cups overlayed with mesh across the lids, and cartons were held in Tupperware containers within insect incubators set to 28 ± 1 °C with 80 ± 10% RH and a 16:8 light:dark cycle. Mosquitoes were provided ad libitum access 10% sucrose-soaked cotton rounds, save that one day prior to feeding on NHPs these were replaced with water-soaked rounds.
For mosquitoes used to infect monkeys, females at 4 days post-eclosion were IT inoculated with between 200 to 500 pfu of either DENV or ZIKV in a volume of 0.2 μL via a Nanoject II (Drummond Scientific, Broomall, PA, USA) and held in cartons as described. Ten days later, mosquitoes were cold-anesthetized and sorted into cartons based on treatment and individual NHP. Mosquitoes were then allowed to feed on monkeys as described below, engorged mosquitoes were separated and housed for 2-3 days. In high-dose treatments, engorgement rates per carton were always <100%; in low-dose treatments, if the single mosquito did not feed, which occurred twice, the feeding was repeated the following day with a new, IT-infected mosquito. To harvest saliva, mosquitoes were cold anesthetized and immobilized on mineral oil, after which legs and wings were removed and the proboscis was inserted into sterile 10 μL pipette tips containing 10 μL of FBS for 30 minutes. After this, the saliva within the FBS was ejected into a microfuge tube containing 100 μL of DMEM containing 2% FBS, 1% P/S and 2.5 μg/mL of amphotericin B (hereafter referred to as mosquito collection media, MCM).
For mosquitoes used to detect transmission, following feeding, all mosquitoes were cold-anesthetized and engorged mosquitoes were separated and housed for 14 days. They were then cold anesthetized on ice and the legs of individual mosquitoes were removed; bodies and legs were placed in separate microfuge tubes containing sterilized steel ball bearings and 500 μL of MCM. In mosquitoes fed on squirrel monkeys at dpi 3 and 4, saliva was also collected as described above.
Non-human primate origins, maintenance, and monitoring
All procedures conducted on non-human primates were approved via UTMB Institutional Animal Care and Use Committee (IACUC) protocol 1912100, approved on December 1, 2019. Mauritius-origin adolescent Macaca fascicularis of both sexes (between 2.4 to 5.4 years of age) weighing between 2.75-5.4 kg were purchased from Worldwide Primates, Inc (Miami, FL, USA). All animals were negative by serology for macacine alphaherpesvirus 1, simian immunodeficiency virus (SIV), simian type D retrovirus (SRV), simian T-lymphotropic virus 1 (STLV-1). Following a minimum 14-day quarantine, all animals were surgically implanted with DST micro-T temperature loggers (Star-Oddi, Garðabær, Iceland) set to record temperature every 15 minutes. Macaques were single housed in open metal caging that allowed for visual but not physical contact with other animals in the room. Standard primate chow was provided twice daily, with enrichment in the form of fruits and vegetables added once daily. All animals received a minimum of twice daily health checks.
Saimiri boliviensis boliviensis (Black capped squirrel monkeys) of both sexes over an adolescent to adult age range (between 4 to 14 years of age) were purchased from the MD Anderson Center (Bastrop, TX, USA). Following a minimum 14-day quarantine, all animals were surgically implanted with DST micro-T temperature loggers (Star-Oddi, Garðabær, Iceland) set to record temperature every 15 minutes. Squirrel monkeys were pair housed in open metal caging that allowed for visual but not physical contact with animals in other cages. Standard primate chow was provided twice daily, with enrichment in the form of fruits and vegetables added once daily. All animals received a minimum of twice daily health checks.
Non-human primate infection, blood sampling and mosquito feeding
All experimental procedures requiring any manipulation of the monkeys were conducted following an overnight fast and under anesthesia, via intramuscular injection of 5-20 (cynomolgus macaques) or 10-15 (squirrel monkey) mg/kg ketamine. Once anesthetized, animals were removed from caging, and physical examination and weights were taken. To maintain core body temperature in squirrel monkeys following anesthesia, all procedures took place atop a preheated heating blanket. To infect monkeys, a 0.5 L cardboard cup containing either 1, 10 or 15 (Fig. 1) sucrose-starved Ae. albopictus Galveston previously injected with DENV-2 or ZIKV or 10 or 15 uninjected control Ae. albopictus was placed on the anesthetized animal’s ear and the mosquitoes were allowed to feed for 7-10 minutes. On subsequent time points following infection, cartons of 10 (DENV-2-infected and control cynomolgus macaques) or 15 (all other experiment arms) sucrose-starved naïve Aedes albopictus were allowed to feed on the ear of the anesthetized animals.
Concurrent with mosquito feeding, blood was collected via venipuncture of the femoral vein. To measure viremia and PRNTs, blood was aliquoted into serum separator tubes (SSTs, BD Biosciences) and centrifuged at 2000g for 5 min to pellet cells and clarified serum was aliquoted into fresh tubes. The others were retained for viremia and PRNT analysis. For the quantification of NK cells, whole blood from venipuncture was added to lithium heparin microtainer tubes (BD Biosciences) and gently inverted.
Natural killer cell quantification
Lithium heparin microtainer tubes containing 0.2–0.3 ml blood were taken into a biosafety cabinet and 200 μL of heparinized whole blood from each NHP was added to an individually labeled round-bottomed polystyrene tube (Corning/Falcon, Corning, NY, USA). Nonspecific antigen binding to Fc receptors was inhibited via 10 minutes of blocking with Human TruStain FcX (BioLegend, San Diego, CA, USA). Cells were subsequently stained with mouse anti-human CD20 (clone 2H7, Alexa-700, catalog number 560631, lot number 0209371, 1:16 dilution), mouse anti-human CD3 (clone SP34-2, Alexa-700, catalog number catalog number 557917, lot number 9185577, 1:16 dilution), mouse anti-human CD14 (clone M5E2, Alexa-700, catalog number 557923, lot number 0023216, 1:8 dilution), mouse anti-human CD16 (clone 3G8, PE, catalog number 556619, lot number 9107538, 1:18 dilution) (all antibodies purchased from BD Biosciences and used at the manufacturer recommended dilution), and Live/Dead Fixable Blue (Invitrogen, Waltham, MA, USA) in a volume of 100 μL for 1 hour in the dark with gentle agitation every 15 minutes. Subsequently, erythrocytes were lysed with BD FACS Lysing Solution (BD Biosciences) diluted in molecular biology grade water (Corning, Corning, NY, USA) for 12 min followed by immediate centrifugation at 500 g for 5 minutes at room temperature. The supernatant was immediately aspirated, and the cellular pellet was reconstituted in FACS Buffer (0.2% bovine serum albumin, 2 mM EDTA in phosphate-buffered saline). Samples were centrifuged at 350 g for 5 minutes at room temperature. FACS buffer was removed, and pellets were fixed in freshly prepared 2% methanol-free formaldehyde (diluted from 16% Ultra Pure EM grade formaldehyde (Polysciences, Warrington, PA, USA) for a minimum of 1 h prior to analysis. Analyses were conducted using a BD LSRFortessa Cell Analyzer (BD, Franklin Lakes, NJ, USA), and 105 total cells were analyzed. Natural killer cells were defined as the live cell population that were CD16+ and CD3-CD14-CD20- (see gating strategy in Fig. S7). This population is functionally analogous to human CD56dim NK cells, characterized by high cytolytic activity and low cytokine production77. Data analysis was conducted utilizing FlowJo software (FlowJo, Ashland, OR, USA).
Plaque reduction neutralization titers
PRNTs were performed in Vero cells in a 12-well tissue culture plate format78. Pre-exposure serum and 28-day post-exposure serum samples were heat inactivated in a 56 °C water bath for 60 minutes to inactivate complement. Serum samples subsequently underwent 2-fold serial dilution series in DMEM with 2% FBS and 1% P/S (1:10 to 1:320). An equal volume (75 µl) of the designated virus at a titer of 800 focus forming units (FFU) per milliliter was added to each serum dilution (dilution series range 1:20-1:640). Serum/virus mixtures were incubated for one hour at 37 °C, at which point 100 μL of mixture was added to a nearly confluent (85-95%) monolayer of Vero cells and allowed to adsorb for one hour at 37 °C. Each well was then overlayed DMEM containing 3% FBS, 1% Pen–Strep, 1.25 μL/mL amphotericin B, and 0.8% weight/vol methylcellulose and incubated 37 °C for 5 days. Fixation and immunostaining procedures took place as described above. PRNT titers were represented as the reciprocal of the highest dilution of serum that inhibited a specified percentage foci when compared to a virus only control.
Statistical analysis and model fitting
To test a priori predictions within and between each species-virus combination, we used standard two-tailed parametric tests, non-parametric tests or generalized linear models, as appropriate. A priori power analyses were conducted to assess our ability to detect changes in peak titer and duration of titer between low and high-dose infections in macaques, resulting in a sample size of 10 in the infected group. As one macaque was transferred to the control group, and as analyses were primarily focused on NK cells and neutralizing antibody titers due to the paucity of viremia, we conducted a post hoc test of power, and found that, with the effect sizes and standard deviations reported, we had approximately 75% power to detect a significant difference in comparisons among 2 or 3 groups and lower power of approximately 20% for linear regressions. Sample sizes were smaller for infected squirrel monkeys (N = 8 per virus) and power was concomitantly lower. For models testing interactions, if the interactions term was non-significant with an effect size η2 < 0.01, we relied on a type II ANOVA for the significance of main effects, following recommendations by Smith and Cribbie79.
The number of infectious bites initially received per NHP, as well as the viral titers contained in biting mosquitoes’ saliva, are the components driving the initial dose delivered to NHPs, which can then drive their viral dynamics and immune response. Differences between experiments regarding those factors were assessed using generalized linear models (Supplementary Text S1).
To quantify a possible relationship between host infectious titer (log10 pfu/ml) and probability to infect mosquitoes, we first used a generalized additive model. The probability of mosquito infection was broadly defined, measured by a positive mosquito body or leg. We used a binomial error distribution and constrained the number of knots to 6 to avoid overfitting. Separate relationships were fitted for DENV-2 and ZIKV, and transmission from both NHP species was considered at once.
We then refined the fitting of dose-response relationships between ZIKV infectious titers (log10 pfu/ml) of squirrel monkeys and the probability of infecting mosquitoes. Separate relationships were fitted depending on whether mosquito infection was measured by the presence of virus in their abdomens or their legs (Supplementary Text S4). In order to compare these ZIKV dose-response relationships to existing results on dengue virus transmission from humans to Aedes aegypti, we retrieved data from Nguyen et al.53 (for abdomen infections) and Duong et al.51 (for leg infections), through the supplementary material of ten Bosch et al.80. As those data were in units RNA copies/ml, we applied a conversion factor, specific to each dengue serotype from Blaney et al.81 (Supplementary Text S4), to transform it into pfu/ml, and applied the same fitting procedure. We also retrieved data from Long et al.52 in which DENV-2 infection of mosquito bodies and heads was measured, and human viral load was in FFU/ml. We considered infection of both mosquito heads and legs as a marker of dissemination.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All raw data from macaques infected with DENV-2 or ZIKV and squirrel monkeys infected with DENV-2 or ZIKV, save for temperature and PRNT values against non-infecting viruses, are provided in Supplementary Datasets 1, 2, 3, and 4. For additional data, see README and data subfolder in GitHub repository https://github.com/helenececilia/hanley_2024_sylvatic_DENV_ZIKV_trade_offs. Source data are provided with this paper.
Code availability
All analyses were performed in R and all scripts, data, and outputs are available in a citable GitHub repository82, available at https://github.com/helenececilia/hanley_2024_sylvatic_DENV_ZIKV_trade_offs/. R version 4.3.1 was used, and analyses relied on packages glmmTMB 1.1.7, DHARMa 0.4.6, multcomp 1.4-25, car 3.1-2, bbmle 1.0-25, mgcv 1.9-0, emdbook 1.3.13, tmvtnorm 1.5.
References
Swei, A., Couper, L. I., Coffey, L. L., Kapan, D. & Bennett, S. Patterns, drivers, and challenges of vector-borne disease emergence. Vector Borne Zoonotic Dis. 20, 159–170 (2020).
Guth, S., Hanley, K. A., Althouse, B. M. & Boots, M. Ecological processes underlying the emergence of novel enzootic cycles: Arboviruses in the neotropics as a case study. PLoS Negl. Trop. Dis. 14, e0008338 (2020).
Barrett, A. D. T. The reemergence of yellow fever. Science 361, 847–848 (2018).
Bryant, J. E., Holmes, E. C. & Barrett, A. D. Out of Africa: a molecular perspective on the introduction of yellow fever virus into the Americas. PLoS Pathog. 3, e75 (2007).
Hanley, K. A. et al. Fever versus fever: the role of host and vector susceptibility and interspecific competition in shaping the current and future distributions of the sylvatic cycles of dengue virus and yellow fever virus. Infect. Genet Evol. 19, 292–311 (2013).
Turell, M. J. et al. Lack of evidence of sylvatic transmission of dengue viruses in the Amazon rainforest near Iquitos, Peru. Vector Borne Zoonotic Dis. 19, 685–689 (2019).
Valentine, M. J. et al. No evidence for sylvatic cycles of chikungunya, dengue and Zika viruses in African green monkeys (Chlorocebus aethiops sabaeus) on St. Kitts, West Indies. Parasit. Vectors 13, 540 (2020).
Aliota, M. T. et al. Zika in the Americas, year 2: What have we learned? What gaps remain? A report from the Global Virus Network. Antivir. Res. 144, 223–246 (2017).
Althouse, B. M. et al. Potential for Zika Virus to establish a sylvatic transmission cycle in the Americas. PLoS Negl. Trop. Dis. 10, e0005055 (2016).
Vasilakis, N. & Weaver, S. C. Flavivirus transmission focusing on Zika. Curr. Opin. Virol. 22, 30–35 (2017).
Favoretto, S. R. et al. Zika virus in peridomestic neotropical primates, Northeast Brazil. EcoHealth 16, 61–69 (2019).
Terzian, A. C. B. et al. Evidence of natural Zika virus infection in neotropical non-human primates in Brazil. Sci. Rep. 8, 16034 (2018).
Cardosa, J. et al. Dengue virus serotype 2 from a sylvatic lineage isolated from a patient with dengue hemorrhagic fever. PLoS Negl. Trop. Dis. 3, e423 (2009).
Diallo, D. et al. Concurrent amplification of Zika, chikungunya, and yellow fever virus in a sylvatic focus of arboviruses in Southeastern Senegal, 2015. BMC Microbiol. 20, 181 (2020).
Franco, L. et al. First report of sylvatic DENV-2-associated dengue hemorrhagic fever in West Africa. PLoS Negl. Trop. Dis. 5, e1251 (2011).
Gubler, D. J., Vasilakis, N. & Musso, D. History and emergence of Zika virus. J. Infect. Dis. 216, S860–S867 (2017).
Pyke, A. T. et al. Complete genome sequence of a highly divergent dengue virus type 2 strain, imported into Australia from Sabah, Malaysia. Genome Announc. 5, e00546–17 (2017).
Pyke, A. T. et al. Highly divergent dengue virus type 1 genotype sets a new distance record. Sci. Rep. 6, 22356 (2016).
Teoh, B. T., Sam, S. S., Abd-Jamil, J. & AbuBakar, S. Isolation of ancestral sylvatic dengue virus type 1, Malaysia. Emerg. Infect. Dis. 16, 1783–1785 (2010).
Childs, L. M. et al. Linked within-host and between-host models and data for infectious diseases: a systematic review. PeerJ 7, e7057 (2019).
Visher, E. et al. The three Ts of virulence evolution during zoonotic emergence. Proc. Biol. Sci. 288, 20210900 (2021).
Bull, J. J. & Lauring, A. S. Theory and empiricism in virulence evolution. PLoS Pathog. 10, e1004387 (2014).
Frank, S. A. Models of parasite virulence. Q. Rev. Biol. 71, 37–78 (1996).
Hanley, K. A. et al. Infection dynamics of sylvatic dengue virus in a natural primate host, the African Green Monkey. Am. J. Trop. Med Hyg. 91, 672–676 (2014).
Koide, F. et al. Development of a Zika Virus infection model in cynomolgus macaques. Front. Microbiol. 7, 2028 (2016).
Rayner, J. O. et al. Comparative pathogenesis of Asian and African-Lineage Zika virus in Indian rhesus macaque’s and development of a non-human primate model suitable for the evaluation of new drugs and vaccines. Viruses 10, 229 (2018).
Crooks, C. M. et al. African-Lineage Zika virus replication dynamics and maternal-fetal Interface infection in pregnant rhesus macaques. J. Virol. 95, e0222020 (2021).
Koenig, M. R. et al. Infection of the maternal-fetal interface and vertical transmission following low-dose inoculation of pregnant rhesus macaques (Macaca mulatta) with an African-lineage Zika virus. PLoS ONE 18, e0284964 (2023).
Raasch, L. E. et al. Fetal loss in pregnant rhesus macaques infected with high-dose African-lineage Zika virus. PLoS Negl. Trop. Dis. 16, e0010623 (2022).
Alizon, S. & van Baalen, M. Transmission-virulence trade-offs in vector-borne diseases. Theor. Popul Biol. 74, 6–15 (2008).
Althouse, B. M. et al. Viral kinetics of primary dengue virus infection in non-human primates: a systematic review and individual pooled analysis. Virology 452-453, 237–246 (2014).
Althouse, B. M. & Hanley, K. A. The tortoise or the hare? Impacts of within-host dynamics on transmission success of arthropod-borne viruses. Philos. Trans. R. Soc. Lond. B: Biol. Sci. 370, 20140299 (2015).
Dudley, D. M. et al. Infection via mosquito bite alters Zika virus tissue tropism and replication kinetics in rhesus macaques. Nat. Commun. 8, 2096 (2017).
Le Coupanec, A. et al. Aedes mosquito saliva modulates Rift Valley fever virus pathogenicity. PLoS Negl. Trop. Dis. 7, e2237 (2013).
Schneider, B. S. et al. Aedes aegypti saliva alters leukocyte recruitment and cytokine signaling by antigen-presenting cells during West Nile virus infection. PLoS ONE 5, e11704 (2010).
Ben-Shachar, R. & Koelle, K. Transmission-clearance trade-offs indicate that dengue virulence evolution depends on epidemiological context. Nat. Commun. 9, 2355 (2018).
Rudnick, A. Studies of the ecology of dengue in Malaysia: a preliminary report. J. Med. Entomol. 2, 203–208 (1965).
Rudnick, A. Ecology of dengue virus. Asian J. Infect. Dis. 2, 156–160 (1978).
Rudnick, A. & Lim, T. W. Dengue virus ecology in Malaysia. Inst. Med. Res. Malays. Bull. 23, 51–152 (1986).
Valentine, M. J., Murdock, C. C. & Kelly, P. J. Sylvatic cycles of arboviruses in non-human primates. Parasit. Vectors 12, 463 (2019).
Wikan, N. & Smith, D. R. Zika virus from a Southeast Asian perspective. Asian Pac. J. Trop. Med. 10, 1–5 (2017).
Chua, C. L. et al. Little Evidence of Zika Virus infection in wild long-tailed macaques, Peninsular Malaysia. Emerg. Infect. Dis. 25, 374–376 (2019).
Leung, G. H., Baird, R. W., Druce, J. & Anstey, N. M. Zika Virus infection in Australia following a monkey bite in Indonesia. Southeast Asian J. Trop. Med. Public Health 46, 460–464 (2015).
Han, B. A. et al. Confronting data sparsity to identify potential sources of Zika virus spillover infection among primates. Epidemics 27, 59–65 (2019).
Diallo, D. et al. Zika virus emergence in mosquitoes in southeastern Senegal, 2011. PLoS One 9, e109442 (2014).
Pereira-Dos-Santos, T., Roiz, D., Lourenco-de-Oliveira, R. & Paupy, C. A systematic review: Is Aedes albopictus an efficient bridge vector for zoonotic arboviruses? Pathogens 9, 266 (2020).
Grard, G. et al. Zika virus in Gabon (Central Africa)–2007: a new threat from Aedes albopictus? PLoS Negl. Trop. Dis. 8, e2681 (2014).
Fortman, J., Hewett, T. & Bennett, B. The Laboratory Nonhuman Primate (CRC Press 2002).
Laffins, M. M., Mellal, N., Almlie, C. L. & Regalia, D. E. Evaluation of infrared thermometry in cynomolgus Macaques (Macaca fascicularis). J. Am. Assoc. Lab Anim. Sci. 56, 84–89 (2017).
Brady, A. G. Research techniques for the squirrel monkey (Saimiri sp.). ILAR J. 41, 10–18 (2000).
Duong, V. et al. Asymptomatic humans transmit dengue virus to mosquitoes. Proc. Natl Acad. Sci. USA 112, 14688–14693 (2015).
Long, K. C. et al. Feasibility of feeding Aedes aegypti mosquitoes on dengue virus-infected human volunteers for vector competence studies in Iquitos, Peru. PLoS Negl. Trop. Dis. 13, e0007116 (2019).
Nguyen, M. N. et al. Host and viral features of human dengue cases shape the population of infected and infectious Aedes aegypti mosquitoes. Proc. Natl Acad. Sci. USA 110, 9072–9077 (2013).
Gloria-Soria, A., Brackney, D. E. & Armstrong, P. M. Saliva collection via capillary method may underestimate arboviral transmission by mosquitoes. Parasit. Vectors 15, 103 (2022).
Miller, M. R. et al. Characterizing and quantifying arbovirus transmission by Aedes aegypti using forced salivation and analysis of bloodmeals. Insects 12, 304 (2021).
Hanley, K. A., Azar, S. R., Campos, R. K., Vasilakis, N. & Rossi, S. L. Support for the transmission-clearance trade-off hypothesis from a study of Zika virus delivered by mosquito bite to mice. Viruses 11, 1072 (2019).
Lambrechts, L. et al. Direct mosquito feedings on dengue-2 virus-infected people reveal dynamics of human infectiousness. PLoS Negl. Trop. Dis. 17, e0011593 (2023).
Azar, S. R. et al. Differential vector competency of Aedes albopictus populations from the Americas for Zika virus. Am. J. Trop. Med Hyg. 97, 330–339 (2017).
Roundy, C. M. et al. Variation in Aedes aegypti mosquito competence for Zika virus transmission. Emerg. Infect. Dis. 23, 625–632 (2017).
Weger-Lucarelli, J. et al. Vector competence of American mosquitoes for three strains of Zika virus. PLoS Negl. Trop. Dis. 10, e0005101 (2016).
Skinner, E. B. et al. Species traits and hotspots associated with Ross River virus infection in nonhuman vertebrates in South East Queensland. Vector Borne Zoonotic Dis. 21, 50–58 (2021).
Gilbert, A. T. et al. Deciphering serology to understand the ecology of infectious diseases in wildlife. Ecohealth 10, 298–313 (2013).
Viana, M. et al. Assembling evidence for identifying reservoirs of infection. Trends Ecol. Evol. 29, 270–279 (2014).
Chaves, B. A. et al. Coinfection with Zika virus (ZIKV) and dengue virus results in preferential ZIKV transmission by vector bite to vertebrate host. J. Infect. Dis. 218, 563–571 (2018).
Bjorkstrom, N. K., Strunz, B. & Ljunggren, H. G. Natural killer cells in antiviral immunity. Nat. Rev. Immunol. 22, 112–123 (2022).
Lambrechts, L. et al. Genetic specificity and potential for local adaptation between dengue viruses and mosquito vectors. BMC Evol. Biol. 9, 160 (2009).
Althouse, B. M. et al. Role of monkeys in the sylvatic cycle of chikungunya virus in Senegal. Nat. Commun. 9, 1046 (2018).
Mandl, J. N. et al. Reservoir host immune responses to emerging zoonotic viruses. Cell 160, 20–35 (2015).
Mandl, J. N. et al. Distinctive TLR7 signaling, type I IFN production, and attenuated innate and adaptive immune responses to yellow fever virus in a primate reservoir host. J. Immunol. 186, 6406–6416 (2011).
Berry, N. et al. High susceptibility, viral dynamics and persistence of South American Zika virus in New World monkey species. Sci. Rep. 9, 14495 (2019).
Azar, S. R. et al. ZIKV demonstrates minimal pathologic effects and mosquito infectivity in viremic cynomolgus macaques. Viruses 10, 661 (2018).
Carrington, L. B. et al. Virological and immunological outcomes in rhesus monkeys after exposure to dengue virus-infected Aedes aegypti mosquitoes. Am. J. Trop. Med Hyg. 103, 112–119 (2020).
Lequime, S. et al. Modeling intra-mosquito dynamics of Zika virus and its dose-dependence confirms the low epidemic potential of Aedes albopictus. PLoS Pathog. 16, e1009068 (2020).
Vasilakis, N. et al. Evolutionary processes among sylvatic dengue type 2 viruses. J. Virol. 81, 9591–9595 (2007).
Ladner, J. T. et al. Complete genome sequences of five Zika virus isolates. Genome Announc. 4, e00377-16 (2016).
Higgs, S. Care, maintenance and experimental infection of mosquitoes. In Disease Vectors (ed. Marquhardt, W. C.) (Elsevier, 2005).
Hong, H. S., Rajakumar, P. A., Billingsley, J. M., Reeves, R. K. & Johnson, R. P. No monkey business: why studying NK cells in non-human primates pays off. Front. Immunol. 4, 32 (2013).
Vasilakis, N. et al. Antigenic relationships between sylvatic and endemic dengue viruses. Am. J. Trop. Med Hyg. 79, 128–132 (2008).
Smith, C. E. & Cribbie, R. Factorial ANOVA with unbalanced data: A fresh look at the types of sums of squares. J. Data Sci. 12, 385–404 (2021).
Ten Bosch, Q. A. et al. Contributions from the silent majority dominate dengue virus transmission. PLoS Pathog. 14, e1006965 (2018).
Blaney, J. E. Jr, Matro, J. M., Murphy, B. R. & Whitehead, S. S. Recombinant, live-attenuated tetravalent dengue virus vaccine formulations induce a balanced, broad, and protective neutralizing antibody response against each of the four serotypes in rhesus monkeys. J. Virol. 79, 5516–5528 (2005).
Cecilia, H. Trade-offs shaping transmission of sylvatic dengue 2 and Zika viruses in native and novel monkey hosts, hanley_2024_sylvatic_DENV_ZIKV_trade_offs, https://doi.org/10.5281/zenodo.10513981 (2024).
Acknowledgements
The authors thank the members of the support staff at UTMB and NMSU, who worked diligently in the face of COVID-19 restrictions during the pandemic. We thank Andrew Montoya at NMSU for his assistance in titering mosquitoes. This study was funded by NIH grant R01AI145918 to K.A.H., N.V., B.M.A., and S.L.R. Squirrel monkeys acquired from the UT MD Anderson Cancer Center, Michael E. Keeling Center for Comparative Medicine and Research were supported by NIH grant 5P40OD010938-36 prior to sale to UTMB.
Author information
Authors and Affiliations
Contributions
Conceptualization: K.A.H., N.V., B.M.A., S.L.R.; Implementation of animal studies: S.R.A., S.L.R., N.V., N.I.O.d.S., R.Y., K.H.; Optimization and implementation of assays: S.R.A., B.A.M., J.T.G., S.L.R., N.V., N.I.O.d.S., R.Y., W.Y., K.H.; Data analysis: H.C., K.A.H., B.M.A.; Writing original draft: K.A.H., H.C., S.R.A., N.V.; Review and editing: K.A.H., H.C., B.M.A., N.V., S.R.A., S.L.R., B.A.M., J.T.G., N.I.O.d.S., R.Y., W.Y.; Supervision: K.A.H., N.V., S.L.R.; Funding acquisition: K.A.H., N.V., B.M.A., S.L.R.; All authors read and approved the final manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hanley, K.A., Cecilia, H., Azar, S.R. et al. Trade-offs shaping transmission of sylvatic dengue and Zika viruses in monkey hosts. Nat Commun 15, 2682 (2024). https://doi.org/10.1038/s41467-024-46810-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-46810-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.