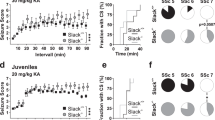
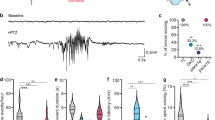
Abstract
Most seizures stop spontaneously; however, the molecular mechanisms that terminate seizures remain unknown. Observations that seizures reduced brain pH and that acidosis inhibited seizures indicate that acidosis halts epileptic activity. Because acid-sensing ion channel 1a (ASIC1a) is exquisitely sensitive to extracellular pH and regulates neuron excitability, we hypothesized that acidosis might activate ASIC1a, which would terminate seizures. Disrupting mouse ASIC1a increased the severity of chemoconvulsant-induced seizures, whereas overexpressing ASIC1a had the opposite effect. ASIC1a did not affect seizure threshold or onset, but shortened seizure duration and prevented seizure progression. CO2 inhalation, long known to lower brain pH and inhibit seizures, required ASIC1a to interrupt tonic-clonic seizures. Acidosis activated inhibitory interneurons through ASIC1a, suggesting that ASIC1a might limit seizures by increasing inhibitory tone. Our results identify ASIC1a as an important element in seizure termination when brain pH falls and suggest both a molecular mechanism for how the brain stops seizures and new therapeutic strategies.
This is a preview of subscription content, access via your institution
Access options






Similar content being viewed by others
References
Noebels, J.L. The biology of epilepsy genes. Annu. Rev. Neurosci. 26, 599–625 (2003).
Spencer, S.S. & Spencer, D.D. Implications of seizure termination location in temporal lobe epilepsy. Epilepsia 37, 455–458 (1996).
Bragin, A., Penttonen, M. & Buzsaki, G. Termination of epileptic afterdischarge in the hippocampus. J. Neurosci. 17, 2567–2579 (1997).
Chapman, A.G., Meldrum, B.S. & Siesjo, B.K. Cerebral metabolic changes during prolonged epileptic seizures in rats. J. Neurochem. 28, 1025–1035 (1977).
Freund, T.F., Buzsaki, G., Prohaska, O.J., Leon, A. & Somogyi, P. Simultaneous recording of local electrical activity, partial oxygen tension and temperature in the rat hippocampus with a chamber-type microelectrode. Effects of anaesthesia, ischemia and epilepsy. Neuroscience 28, 539–549 (1989).
Kloiber, O., Bockhorst, K., Hoehn-Berlage, M. & Hossmann, K.A. Effect of hypoxia on bicuculline seizures of rat: NMR spectroscopy and bioluminescence imaging. NMR Biomed. 6, 333–338 (1993).
Yamada, K. et al. Protective role of ATP-sensitive potassium channels in hypoxia-induced generalized seizure. Science 292, 1543–1546 (2001).
Kirchner, A., Veliskova, J. & Velisek, L. Differential effects of low glucose concentrations on seizures and epileptiform activity in vivo and in vitro. Eur. J. Neurosci. 23, 1512–1522 (2006).
Haglund, M.M. & Schwartzkroin, P.A. Role of Na-K pump potassium regulation and IPSPs in seizures and spreading depression in immature rabbit hippocampal slices. J. Neurophysiol. 63, 225–239 (1990).
Somjen, G.G. Acidification of interstitial fluid in hippocampal formation caused by seizures and by spreading depression. Brain Res. 311, 186–188 (1984).
Wang, R.I. & Sonnenschein, R.R. pH of cerebral cortex during induced convulsions. J. Neurophysiol. 18, 130–137 (1955).
Kaila, K. & Chesler, M. Activity-evoked changes in extracellular pH. in pH and Brain Function (ed. K. Kaila & B.R. Ransom) 309 (Wiley-Liss, New York, 1998).
Lennox, W.G. The effect on epileptic seizures of varying the composition of the respired air. J. Clin. Invest. 4, 23–24 (1929).
Mitchell, W.G. & Grubbs, R.C. Inhibition of audiogenic seizures by carbon dioxide. Science 123, 223–224 (1956).
Woodbury, D.M., Rollins, L.T., Henrie, J.R., Jones, J.C. & Sato, T. Effects of carbon dioxide and oxygen on properties of experimental seizures in mice. Am. J. Physiol. 184, 202–208 (1956).
Velisek, L., Dreier, J.P., Stanton, P.K., Heinemann, U. & Moshe, S.L. Lowering of extracellular pH suppresses low-Mg2+–induces seizures in combined entorhinal cortex–hippocampal slices. Exp. Brain Res. 101, 44–52 (1994).
Traynelis, S.F. & Cull-Candy, S.G. Proton inhibition of N-methyl-d-aspartate receptors in cerebellar neurons. Nature 345, 347–350 (1990).
Somjen, G.G. & Tombaugh, G.C. pH modulation of neuronal excitability and central nervous system functions. in pH and Brain Function (ed. K. Kaila & B.R. Ransom) 373–393 (Wiley-Liss, New York, 1998).
Dulla, C.G. et al. Adenosine and ATP link PCO2 to cortical excitability via pH. Neuron 48, 1011–1023 (2005).
Waldmann, R., Champigny, G., Bassilana, F., Heurteaux, C. & Lazdunski, M. A proton-gated cation channel involved in acid sensing. Nature 386, 173–177 (1997).
García-Añoveros, J., Derfler, B., Neville-Golden, J., Hyman, B.T. & Corey, D.P. BNaC1 and BNaC2 constitute a new family of human neuronal sodium channels related to degenerins and epithelial sodium channels. Proc. Natl. Acad. Sci. USA 94, 1459–1464 (1997).
Lingueglia, E. et al. A modulatory subunit of acid sensing ion channels in brain and dorsal root ganglion cells. J. Biol. Chem. 272, 29778–29783 (1997).
Benson, C.J. et al. Heteromultimerics of DEG/ENaC subunits form H+-gated channels in mouse sensory neurons. Proc. Natl. Acad. Sci. USA 99, 2338–2343 (2002).
Wemmie, J.A. et al. Acid-sensing ion channel 1 is localized in brain regions with high synaptic density and contributes to fear conditioning. J. Neurosci. 23, 5496–5502 (2003).
Alvarez de la Rosa, D. et al. Distribution, subcellular localization and ontogeny of ASIC1 in the mammalian central nervous system. J. Physiol. (Lond.) 546, 77–87 (2003).
Yermolaieva, O., Leonard, A.S., Schnizler, M.K., Abboud, F.M. & Welsh, M.J. Extracellular acidosis increases neuronal cell calcium by activating acid-sensing ion channel 1a. Proc. Natl. Acad. Sci. USA 101, 6752–6757 (2004).
Zha, X-M., Wemmie, J.A. & Welsh, M.J. ASIC1a is a postsynaptic proton receptor that influences the density of dendritic spines. Proc. Natl. Acad. Sci. USA 103, 16556–16561 (2006).
Wemmie, J.A. et al. The acid-activated ion channel ASIC contributes to synaptic plasticity, learning and memory. Neuron 34, 463–477 (2002).
Xiong, Z.G. et al. Neuroprotection in ischemia: blocking calcium-permeable acid-sensing ion channels. Cell 118, 687–698 (2004).
Gruol, D.L., Barker, J.L., Huang, L.Y., MacDonald, J.F. & Smith, T.G., Jr. Hydrogen ions have multiple effects on the excitability of cultured mammalian neurons. Brain Res. 183, 247–252 (1980).
Vukicevic, M. & Kellenberger, S. Modulatory effects of acid-sensing ion channels (ASICs) on action potential generation in hippocampal neurons. Am. J. Physiol. Cell Physiol. 287, C682–C690 (2004).
Bolshakov, K.V. et al. Characterization of acid-sensitive ion channels in freshly isolated rat brain neurons. Neuroscience 110, 723–730 (2002).
Dubberke, R., Vasilets, L.A. & Schwarz, W. Inhibition of the Na+, K+ pump by the epileptogenic pentylenetetrazole. Pflugers Arch. 437, 79–85 (1998).
Fisher, R.S. Animal models of the epilepsies. Brain Res. Brain Rs. Rev. 14, 245–278 (1989).
Cole, T.B., Robbins, C.A., Wenzel, H.J., Schwartzkroin, P.A. & Palmiter, R.D. Seizures and neuronal damage in mice lacking vesicular zinc. Epilepsy Res. 39, 153–169 (2000).
Escoubas, P. et al. Isolation of a tarantula toxin specific for a class of proton-gated Na+ channels. J. Biol. Chem. 275, 25116–25121 (2000).
Coryell, M.W. et al. Targeting ASIC1a reduces innate fear and alters neuronal activity in the fear circuit. Biol. Psychiatry 62, 1140–1148 (2007).
Wemmie, J.A. et al. Overexpression of acid-sensing ion channel 1a in transgenic mice increases fear-related behavior. Proc. Natl. Acad. Sci. USA 101, 3621–3626 (2004).
Mares, P. & Kubova, H. Electrical stimulation-induced models of seizures. in Models of Seizures and Epilepsy (ed. A. Pitkänen, P.A. Schwartzkroin & S.L. Moshé) 153–159 (Elsevier Academic Press, New York, 2006).
Buckmaster, P.S. & Wong, E.H. Evoked responses of the dentate gyrus during seizures in developing gerbils with inherited epilepsy. J. Neurophysiol. 88, 783–793 (2002).
Freund, T.F. & Buzsaki, G. Interneurons of the hippocampus. Hippocampus 6, 347–470 (1996).
Tschirgi, R.D. & Taylor, J.L. Slowly changing bioelectric potentials associated with the blood-brain barrier. Am. J. Physiol. 195, 7–22 (1958).
Meduna, L.J. The physiological basis of carbon dioxide treatment. in Carbon Dioxide Therapy (ed. L.J. Meduna) 3–19 (Charles C Thomas, Springfield, Illinois, 1958).
Waxman, S.G. Channel, neuronal and clinical function in sodium channelopathies: from genotype to phenotype. Nat. Neurosci. 10, 405–409 (2007).
Friese, M.A. et al. Acid-sensing ion channel 1 contributes to axonal degeneration in autoimmune inflammation of the central nervous system. Nat. Med. 13, 1483–1489 (2007).
Immke, D.C. & McCleskey, E.W. Lactate enhances the acid-sensing Na+ channel on ischemia-sensing neurons. Nat. Neurosci. 4, 869–870 (2001).
Racine, R.J. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr. Clin. Neurophysiol. 32, 281–294 (1972).
Bernard, C. Hippocampal slices: designing and interpreting studies in epilepsy research. in Models of Seizures and Epilepsy (eds. A. Pitkänen, P. Schwartzkroin & S. Moshé) 59–72 (Elsevier Academic Press, New York, 2006).
Anderson, W.W., Swartzwelder, H.S. & Wilson, W.A. The NMDA receptor antagonist 2-amino-5-phosphonovalerate blocks stimulus train-induced epileptogenesis, but not epileptiform bursting, in the rat hippocampal slice. J. Neurophysiol. 57, 1–21 (1987).
Kay, A.R. & Wong, R.K. Isolation of neurons suitable for patch-clamping from adult mammalian central nervous systems. J. Neurosci. Methods 16, 227–328 (1986).
Acknowledgements
We would like to thank J.M. Haenfler and A. Wunsch for excellent technical assistance, D.R. Langbehn and B. Zimmerman for assisting with statistical analysis, C.K. Kovach for assistance with EEG equipment and analysis, and F. Abboud and V. Snitsarev for assistance with EEG computational analysis. A.E.Z. was supported by the University of Iowa Interdisciplinary Training Program in Pain Research (US National Institute of Neurological Disorders and Stroke T32NS045549). M.J.W. is an Investigator of the Howard Hughes Medical Institute. J.A.W. was supported by US National Institute of Neurological Disorders and Stroke grant 1R21NS058309-01A1, a Veteran's Administration Advanced Career Development Award and a Research Initiatives grant from the University of Iowa Roy J. and Lucille A. Carver College of Medicine.
Author information
Authors and Affiliations
Contributions
A.E.Z. helped to generate the hypotheses for ASIC1a involvement in seizures, carried out the slice and in vivo electrophysiology/behavioral experiments, assessed seizure threshold, measured brain pH in response to seizures and CO2, assisted in studies using acutely dissociated neurons, wrote initial manuscript drafts and worked closely with other authors in editing the figures and manuscript. M.K.S. initiated, conceived, performed and interpreted electrophysiological experiments in dissociated neurons and assisted in preparation of the manuscript. M.A.S. helped to carry out and interpret the EEG experiments. G.W.A. assisted with seizure threshold studies. M.A.H. provided EEG equipment and helped with EEG interpretation. M.J.W. provided important overall direction and initiation for the project, contributed to experimental design and data interpretation, provided funding and wrote the manuscript. J.A.W. initiated development of the project, led experimental direction, interpreted data, carried out pilot experiments, provided funding and wrote the manuscript.
Corresponding authors
Rights and permissions
About this article
Cite this article
Ziemann, A., Schnizler, M., Albert, G. et al. Seizure termination by acidosis depends on ASIC1a. Nat Neurosci 11, 816–822 (2008). https://doi.org/10.1038/nn.2132
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2132
This article is cited by
-
PLPP/CIN-mediated NF2 S10 dephosphorylation distinctly regulates kainate-induced seizure susceptibility and neuronal death through PAK1-NF-κB-COX-2-PTGES2 signaling pathway
Journal of Neuroinflammation (2023)
-
Triggering of Major Brain Disorders by Protons and ATP: The Role of ASICs and P2X Receptors
Neuroscience Bulletin (2023)
-
Sepsis modulates cortical excitability and alters the local and systemic hemodynamic response to seizures
Scientific Reports (2022)
-
Seizures and risks for recurrence in critically ill patients: an observational cohort study
Journal of Neurology (2022)
-
Pregabalin for Recurrent Seizures in Critical Illness: A Promising Adjunctive Therapy, Especially for cyclic Seizures
Neurocritical Care (2022)