Abstract
This study aimed to define cardiovascular and heart rate variability (HRV) changes following head-up tilt (HUT) in children/adolescents with chronic fatigue syndrome (CFS) in comparison to age- and gender-matched controls. Twenty-six children/adolescents with CFS (11–19 y) and controls underwent 70-degree HUT for a maximum of 30 min, but returned to horizontal earlier at the participant's request with symptoms of orthostatic intolerance (OI) that included lightheadedness. Using electrocardiography and beat-beat finger blood pressure, a positive tilt was defined as OI with 1) neurally mediated hypotension (NMH); bradycardia (HR <75% of baseline), and hypotension [systolic pressure (SysP) drops >25 mm Hg)] or 2) postural orthostatic tachycardia syndrome (POTS); HR increase >30 bpm, or HR >120 bpm (with/without hypotension). Thirteen CFS and five controls exhibited OI generating a sensitivity and specificity for HUT of 50.0% and 80.8%, respectively. POTS without hypotension occurred in seven CFS subjects but no controls. POTS with hypotension and NMH occurred in both. Predominant sympathetic components to HRV on HUT were measured in CFS tilt–positive subjects. In conclusion, CFS subjects were more susceptible to OI than controls, the cardiovascular response predominantly manifest as POTS without hypotension, a response unique to CFS suggesting further investigation is warranted with respect to the pathophysiologic mechanisms involved.
Similar content being viewed by others
Main
Myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS) is, as the name suggests, a persistent tiredness, but has a clinical definition refined in the 1994 Centers for Disease Control and Prevention (CDC) criteria (1). These criteria consist of severe and disabling fatigue with a combination of symptoms lasting for longer than 6 mo, associated with four of the following: cognitive difficulties, pharyngitis, tender lymphadenopathy, muscle pain, joint pain, headache, nonrefreshing sleep, and postexercise malaise. For adolescents and children, 3 mo of symptoms has been suggested as diagnostic and encompassed by the CDC definition (2). Reliable prevalence data are limited, but the latest estimates range from 0.5% to 1.29% (3,4), with a 2.5:1 female-to-male ratio (5). CFS is an important illness in adolescence, and if the diagnostic accuracy can be enhanced, the resultant improvement in prognosis is then achievable. One way forward is to examine the cardiovascular response in this group of patients.
Abnormalities in cardiovascular control of blood pressure and autonomic regulation of the heart may contribute to some physical symptoms of CFS (6). Cardiovascular changes can be examined using the head-up tilt (HUT) test, a noninvasive orthostatic test that in some can yield syncope (i.e. fainting) or near syncope with symptoms of orthostatic intolerance (OI), i.e. lightheadedness, nausea, blurred vision, sweating, headache. Cardiovascular changes accompany or are preceded by OI on HUT. Two forms have been linked with CFS in research studies: neurally mediated hypotension (NMH) in which there is an abrupt drop in blood pressure and postural orthostatic tachycardia syndrome (POTS) in which there is an excessive tachycardia with HUT. Stewart et al. (7) investigated responses to 80-degree HUT in 26 CFS patients (11–19 y) and 13 healthy controls. All but one CFS subject and four of 13 controls experienced OI. Another study reported a predisposition to OI symptoms with standing in 57% of young cases (8). These studies suggest a strong link between OI and child/adolescent CFS, but few CFS studies in this age group exist, and few accurately define the normal healthy adolescent response to HUT. As young age has been positively correlated with postural tachycardia and syncope during HUT (9), it is essential to define disease (CFS) as an abnormal HUT that cannot be explained by the normal physiologic response of an adolescent.
Autonomic regulation of the heart can be assessed by measures of heart rate variability (HRV) reflecting the continuous oscillation of the R-R interval around its mean (10). Information about HRV in CFS patients (11,12) has commonly been obtained using linear methods such as spectral analysis that analyzes the frequencies of R-R interval waveforms with respect to time. Linear methods yield conflicting results with regard to CFS (13–16). They rely on stationary and long recordings, not differentiating between increasing and decreasing heart rates, and provide no information on time direction (17). To calculate variations related to fluctuating interbeat intervals with HUT, nonlinear methods should also be applied.
The aims of the present study were to 1) define the cardiovascular responses in a moderate sample of children and adolescents with CFS using a standard 70-degree HUT, 2) provide an age- and sex-matched comparison of healthy children and adolescents to define the normal physiologic response in this age group, 3) characterize the HRV changes with respect to HUT and CFS patients with OI employing linear and nonlinear techniques.
METHODS
Twenty-six children/adolescents with a confirmed diagnosis of CFS (1) and 26 controls matched by age (±3 mo), sex, and body mass index status completed the study. Median age was 15 y 2 mo (range, 10 y 11 mo to 19 y 0 mo), the and female-to-male ratio was 1.6:1. Participants gave informed written consent. The Otago Ethics Committee approved the protocol.
CFS.
Twenty-nine possible CFS subjects were recruited through General Practices and Pediatric Outpatient Departments. They were free of acute illness and neurally active or vasoactive medication at the time of study. All underwent physical examination and full blood chemistry analysis (including full blood count, thyroid function, and erythrocyte sedimentation rate). A pediatrician took a full history that included duration and symptomology as covered in the CDC criteria (1) as well as any precipitants, drug history, or other exposures. Two screening questions about depression (previously validated) (18) and family history of psychological conditions were asked. The examination ruled out other causes of fatigue so that only 26 meeting the CDC criteria went on to participate, thus excluding three with psychiatric, autoimmune, and gastrointestinal disorders.
Controls.
Control participants were recruited by invitation through local schools. They were free of illness and neurally active or vasoactive medications at the time of study. All underwent a brief examination and history to rule out significant symptoms suggestive of CFS. Included in the history taking were numbers of previous syncopal or near syncopal events and palpitations.
Setup.
Children/adolescents were studied at 0900 (following an overnight fast) in a quiet room with temperature at 19°–21°C. An electrocardiogram (ECG) was recorded continuously as were thoracic and abdominal movements (Respitrace model 150; Respitrace Co.), hemoglobin oxygen saturation, and pulse waveform (Nellcor N-200, Nellcor, Hayward, CA). A digital pulse pressure provided continuous noninvasive beat-beat blood pressure recorded by photoplethysmography (Portapres Model-2). An arm sling supported the Portapres weight and kept the forearm horizontal. All signals were relayed through an analog-to-digital converter (PowerLab, ADInstruments Pty Ltd.).
HUT protocol.
A motorized tilt table, with footboard for weight bearing and body straps for stability, was used. Baseline recordings were obtained for 15 min while subjects were supine. Subjects were then tilted 70 degrees head up for a maximum of 30 min or returned to horizontal earlier on request with symptoms of presyncope consistent with OI and, in one case, syncope. Subjects then remained horizontal for 15 min. A positive tilt was defined as: 1) NMH when OI was associated with bradycardia (HR <75% of baseline) and hypotension (systolic pressure (SysP) drops >25 mm Hg); 2) POTS in which OI was associated with an HR increase >30 bpm or reached >120 bpm during the first 10 min of the tilt with or without hypotension. HR and SysP data were averaged at time points of 1) baseline (15 min before tilt); 2) early response (3–5 min into tilt); 3) mid-response (average of 5 min midway, time adjusted according to duration of tolerance); 4) pre-return (1 min before event/if no event, 1 min before return to horizontal).
Measurement of HRV.
ECG data recorded at 500 Hz were analyzed using the HRV extension to PowerLab on five consecutive minutes of data: at baseline (pre-tilt), mid-tilt, and mid-recovery (5–10 min). For recordings ending well before the 30 min of HUT, the mid-tilt period was adjusted such that if 5 min was not available, at least 256 consecutive cycles were analyzed midway.
Linear methods.
1) Two time domain measures quantified total variation in HR: the root mean square of successive differences in R-R intervals (rMSSD) and the proportion of the number of interval differences between successive R-R intervals >50 ms (pNN50). 2) Power spectral analysis expressed dominant frequencies within a series of waveforms, quantifying beat-beat variability representing high-frequency (HF) bands (HF: 0.15–0.4 Hz) indicative of parasympathetic modulation (19), low-frequency (LF) bands (LF: 0.04–0.15 Hz) indicative of sympathetic and parasympathetic modulation and the LF/HF ratio, an index of sympathovagal balance (10,20). LF and HF power is presented in normalized units representing the relative power of each in proportion to the total power minus the very low frequency component (≤0.04 Hz).
Nonlinear method.
R-R intervals plotted as a function of the previous R-R interval create a scattergram (Poincaré plot). Quantitative data from Poincaré plots yield 1) changes in overall variability [standard deviation of the continuous beat-beat intervals (SDRR)] reflecting both parasympathetic and sympathetic modulation of HR (21,22); 2) beat-beat changes reflecting parasympathetic modulation [SD of the change between instantaneous beat-beat intervals (SDΔRR)] (21). SDRR and SDΔRR are calculated in absolute and then normalized units by dividing the SD by the average R-R interval. A higher ratio (SDRR/SDΔRR) is assumed to measure sympathetic predominance (23,24).
Statistical analysis.
The significance of positive responses to HUT between groups was determined using McNemar's test for paired binary data. Values given at one time point were compared using a paired t test, and over continuous time points, mixed linear modeling was used controlling for any remaining variability in age, sex, or body mass index. Kaplan-Meier survival curves and Cox proportional hazards models were used to compare the between-group risk of developing OI, tachycardia, or hypotension to HUT. Further statistical comparisons between control and CFS groups excluded control children with a positive tilt response to eliminate any possibility of these children having a different sympathetic profile on HUT than the negative responding controls (25,26). A p value <0.05 was considered statistically significant.
RESULTS
Participants.
CFS characteristics of the 26 cases are given (Table 1). Nonrefreshing sleep and postexercise malaise were the most common symptoms occurring “often” or always” in 76% and 93%, respectively, followed by headaches (50%), impaired memory (38.6%), tender lymph nodes (34.6%), muscle pain (36.4%), sore throat (26%), and joint pain (26%). Previous syncope was reported in 10 (38%) cases, one of whom also had many previous episodes of presyncope and two of whom “often” felt presyncopal. Seven reported experiencing palpitations. Among possible triggers to the illness, glandular fever was the most commonly assumed in 14 (56%); 10 were confirmed positive for Epstein-Barr virus by antibody detection. Three identified a sore throat as the precipitant; two, a sore stomach; one, gingivitis; and eight, an unknown virus. Cases reported school absenteeism for a median 25% (range, 6%–100%) of the previous year; two were unable to attend school at all. All reported levels of physical activity well below previously, 10 reported difficulties with walking and 7 reported reduced muscle power at rest. All but one reported daytime sleepiness and frequent daytime rests, but no one was bedridden. All blood test results were within normal limits.
In the control group, the most common CFS-related symptom was headache occurring “often” or “always” in five (20%). Two reported nonrefreshing sleep and, five daytime sleepiness, but none experienced postexercise malaise or any of the weakening physical symptoms of CFS. Six (23%) reported past incidences of syncope and two reported presyncope. Two subjects experienced palpitations occasionally. Two had a history of glandular fever and the median time off school in the preceding year was 1% (range, 0%–10%).
Response to HUT.
Table 1 also summarizes the tilt responses in which 13 of 26 (50%) of CFS and five of 26 (20%) control subjects exhibited OI (p = 0.04), yielding sensitivity and specificity for the test of 50.0% and 80.8%, respectively. In CFS, responses were predominantly POTS without hypotension occurring in seven (p = 0.01). NMH was a feature in two CFS subjects (one resulting in syncope) and one control subject. POTS with hypotension was featured in four CFS and four control children/adolescents. Six of the 13 CFS patients with a positive tilt had had previous syncope or symptoms of presyncope, as did two of the five controls.
Kaplan-Meier survival curves in relation to onset of tachycardia, hypotension, and OI are shown (Fig. 1). A significant difference between groups was the time to onset of tachycardia, with the hazard ratio calculated at 3.2 [95% confidence interval (CI): 1.03–10.2) indicating that the risk of a CFS patient developing tachycardia with HUT was 3.2 times higher than controls, assuming a constant ratio over time. Hazard ratios for the onset of hypotension or OI were not significant at 1.24 (95% CI: 0.38–4.1) and 2.6 (95% CI: 0.9–7.4), respectively.
Analyses of HR data at baseline, comparing the 21 controls (Fig. 2) and 13 CFS patients with a negative response (Fig. 3) showed no significant differences between groups (72 bpm, 95% CI: 67–75 vs 70 bpm, 95% CI: 63–76, respectively). However, the pattern of change over time was significantly different for HR averaged over the mid-tilt; HR increase from baseline was significantly higher in CFS (22 bpm, 95% CI: 17–26) than controls (17 bpm, 95% CI: 13–20). There was no significant difference for SysP at any time point.
Control subjects. (A) Mean HR data and 95% CI for the 21 control subjects with a negative response to HUT plotted from baseline (0 min) to completion of HUT (maximum 30 min). (B) Corresponding SysP data. (C) Individual HR plots from the five children/adolescents with a positive response to HUT. POTS, solid lines; NMH, dashed lines. D: Corresponding SysP data. Downward markers indicate time points at which symptoms of OI appeared.
CFS subjects. (A) Mean HR data and 95% CI for the 13 subjects with a negative response to HUT plotted from baseline (0 min) to completion of HUT (maximum 30 min). (B) Corresponding SysP data. (C) Individual HR plots from the six children/adolescents with a positive response to HUT meeting criteria for POTS with hypotension (solid lines) and NMH (dashed lines). (D) Corresponding SysP data. (E) Individual plots of HR from the six children/adolescents with a positive response to HUT meeting criteria for POTS without hypotension. (F) Corresponding SysP data. Downward markers indicate time points at which symptoms of OI appeared.
HRV.
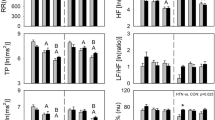
Table 2 summarizes the HRV variables for three subject groups divided according to their tilt response. At baseline, there were no significant group differences in any HRV variables despite a trend for the nonlinear data (SDRR and SDΔRR) to be reduced in CFS (tilt positive) compared with healthy controls (p = 0.06 for both). All variables changed significantly at mid-tilt: R-R interval and time domain measures (rMSSD and pNN50) decreased significantly; spectral data showed this as a shift to sympathetic over parasympathetic dominance as LF/HF increased significantly, also apparent with the nonlinear method (significantly higher SDRR/SDΔRR). The shift was highest in the CFS tilt–positive group detected by the relative changes in SDRR/SDΔRR from baseline to mid-tilt of 1.6-fold in healthy controls to 2.0-fold in CFS with a positive tilt.
At mid-tilt, group differences emerged with shorter R-R intervals in both CFS tilt–negative and CFS tilt–positive groups compared with controls, plus a concomitant decrease in rMSSD and pNN50. Nonlinear methods revealed reduced HRV in the CFS tilt–positive group in comparison with healthy controls, represented as enhanced sympathetic activity (increase SDRR/SDΔRR). Spectral analyses did not detect these group differences.
DISCUSSION
The main findings of this study were that children/adolescents with CFS exhibited more episodes of OI than their age- and gender-matched controls, predominantly manifested as POTS without hypotension. In POTS with hypotension, the tachycardiac event always preceded the hypotensive event. Therefore, in those meeting criteria for POTS without hypotension, it is possible that hypotension may have ensued had we been able to extend the duration of HUT. Tachycardia on HUT follows a reflex decrease in parasympathetic nerve activity and an increase in sympathetic nerve activity to the heart. An excessive tachycardia occurs in POTS appearing as the most sensitive and earliest index of OI (27), most likely representing a failure of the peripheral vasculature to constrict appropriately under stress (28). Other possible physiologic abnormalities involve a redistributive form of hypovolemia resulting from specific pooling within the splanchnic and thoracic vascular beds and defective muscle pumps (29). Reduced hydration status may be an important contributor.
The cardiovascular response of a few participants met the criteria for NMH (two CFS and one control). This is assumed to be a centrally mediated response to HUT signaling the heart to slow, inducing reflex vasodilatation leading to sudden hypotension and bradycardia (30). POTS may be an immature precursor to NMH possibly related to the balance of epinephrine and norepinephrine release in the system. If POTS is an immature precursor of NMH, it is worth speculating that demonstration of POTS without hypotension in the CFS cases with OI could indicate a mild form of autonomic depression manifested as absent or a delayed hypotension. A limitation of our study was the small number of controls meeting the criteria for POTS with hypotension (four), thus not a useful sample size for comparative analyses.
Our findings are in agreement with a previous study by Stewart et al. (7) in relation to more CFS than control children/adolescents exhibiting a positive response to HUT. However, unlike their study in which 96% of CFS subjects and 31% of controls responded positively, only 50% of our CFS subjects and 20% of controls responded this way. It is not clear why some had perfectly normal responses to the tilt, despite clear CFS symptomatology. Perhaps it represents the variable nature of the disease and/or the fact that we used a more conservative protocol than Stewart et al. (7). Thus, the HUT was shorter (30 min versus 40 min), a slighter tilt angle was used (70 degrees versus 80 degrees) and no i.v. line was used; use of the latter has been attributed to adding a false-positive rate (31). However, both studies were conducted after an overnight fast, which may have increased the susceptibility of some subjects to OI on HUT.
Predominant cardiovascular responses to HUT in CFS patients were those meeting the criteria for POTS without hypotension. This agrees with the findings of Stewart et al. (7). No controls responded this way, suggesting this aspect deserves further investigation in respect of the pathophysiologic mechanisms, but without more detailed history with regard to any orthostatic symptoms before the onset of illness, we cannot rule out the possibility that POTS in some may be the cause rather than a manifestation of the illness, as previously suggested (32). POTS, like CFS, may follow a viral infection.
As far as we are aware, this is the only matched case-control study of CFS in children/adolescents. Compared with adults, adolescents are more prone to syncope; 15% to 50% will have at least one episode by the end of adolescence (33). However, the study was limited by having only one control for each case; two or more controls for each would improve the power of the study. Although 12 of 26 CFS subjects and eight of 26 controls had previous episodes of syncope/presyncope, only six CFS subjects and two controls demonstrated OI under our laboratory conditions. Stewart et al. (7) excluded control children with a history of syncope, but we purposely did not do this so as to get a broad a range of orthostatic responses, representative of a normal healthy population. This approach led to a better picture of the prevalence of OI in cases relative to healthy controls and thus a better picture of any possible clinical value of the test.
An important finding was that nonlinear methods more readily detected enhanced sympathetic responses to HUT in CFS tilt–positive subjects than traditional linear methods. A previous study of adolescents that used the same tilt protocol and linear techniques reported sympathetic predominance in CFS (12); subjects were chosen by design to have OI and more responded with hypotension, perhaps reflecting a group with more severe autonomic imbalance. Evidence of enhanced sympathetic activity in adolescent CFS is strengthened by a recent study using nonlinear methods (time-varying spectrums) to demonstrate this even under conditions of low-grade HUT (20 degrees) (34). It needs to be emphasized, however, that attaching precise physiologic interpretation of any HRV index has its limitations because the autonomic nervous system is highly integrated. In addition, despite vast numbers of pharmacologic studies characterizing changes in HRV indices with autonomic modulation, there are both criticism and debate in the literature regarding these (35,36).
Clearly, illness in the cases studied here primarily followed a viral insult/infection. The host response to such an insult may reduce hydration and lead to a lowering of blood volume. Gravitational deconditioning as a result of inactivity and prolonged bed rest that follows illness onset may play a role in the pathophysiologic mechanism of OI and autonomic disturbances in CFS (15,37). Prolonged bed rest causes a redistribution of blood flow from the periphery to intravascular compartments and a reduction in plasma volume. Atrophy of the leg muscles allowing increased venous pooling and loss of venomotor tone also occurs. These factors may combine to tip the balance in favor of OI on experimental upright tilt in some CFS patients. Although no cases were bedridden at the time of study, reduced activity and daytime bed rest were common. However, a limitation was that we did not include a physical or ambulatory index to investigate correlations with cardiovascular responses. Alternative explanations for the mechanisms include a postviral neuropathy (15), decreased baroreceptor reflex responsiveness involving reduced gain (12,38), abnormalities in afferent or efferent pathways, and/or abnormalities in integration of cardiovascular control at the brainstem.
There are no published controlled trials of treatment of children/adolescents with CFS. The management approach deals with individual symptoms. While symptoms persist for many months or years, prognosis for complete recovery is better than for adults (39,40). Undoubtedly, children/adolescents with CFS suffer many symptoms that are disturbing, and OI in itself can be extremely debilitating. The findings of the present study add to the body of evidence that there is likely a shift in favor of increased sympathovagal balance of the autonomic nervous system on HUT that could account for some of the symptoms of CFS in those with a positive tilt response. Our findings did not allow further delineation of the mechanisms involved in autonomic disturbances, but rather fill a void in the literature providing comprehensive matched case-control data over this young age range and using a standard tilt protocol. We suggest further research should take a prospective approach to capture CFS early by enrolling children/adolescents at the stage of glandular fever diagnosis. By thoroughly detailing medical events before and surrounding the onset and progression of illness, many of the unknown factors regarding both autonomic and immunologic disturbances in those who go on to develop CFS could be delineated.
Abbreviations
- CFS:
-
chronic fatigue syndrome
- HRV:
-
heart rate variability
- HUT:
-
head-up tilt
- NMH:
-
neurally mediated hypotension
- OI:
-
orthostatic intolerance
- POTS:
-
postural orthostatic tachycardia syndrome
- SysP:
-
systolic pressure
References
Fukuda K, Straus SE, Hickie I, Sharpe MC, Dobbins JG, Komaroff A 1994 The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann Intern Med 121: 953–959
Jason LA, Bell DS, Row K, Van Hoof EL, Jordan K, Lapp C, Gurwitt A, Miike T, Torres-Harding S, De Meirleir K 2006 A pediatric case definition for myalgic encephalomyelitis and chronic fatigue syndrome. J Chronic Fatigue Syndr 13: 1–44
Farmer A, Fowler T, Scourfield J, Thapar A 2004 Prevalence of chronic disabling fatigue in children and adolescents. Br J Psychiatry 184: 477–481
Rimes KA, Goodman R, Hotopf M, Wessely S, Meltzer H, Chalder T 2007 Incidence, prognosis, and risk factors for fatigue and chronic fatigue syndrome in adolescents: a prospective community study. Pediatrics 119: e603–e609
Marshall GS 1999 Report of a workshop on the epidemiology, natural history, and pathogenesis of chronic fatigue syndrome in adolescents. J Pediatr 134: 395–405
Rowe PC 2002 Orthostatic intolerance and chronic fatigue syndrome: new light on an old problem. J Pediatr 140: 387–389
Stewart JM, Gewitz MH, Weldon A, Arlievsky N, Li K, Munoz J 1999 Orthostatic intolerance in adolescent chronic fatigue syndrome. Pediatrics 103: 116–121
Tanaka H, Matsushima R, Tamai H, Kajimoto Y 2002 Impaired postural cerebral hemodynamics in young patients with chronic fatigue with and without orthostatic intolerance. J Pediatr 140: 412–417
de Jong-de Vos van Steenwijk CC, Wieling W, Johannes JM, Harms MP, Kuis W, Wesseling KH 1995 Incidence and hemodynamic characteristics of near-fainting in healthy 6- to 16-year old subjects. J Am Coll Cardiol 25: 1615–1621.
1996 Heart rate variability: standards of measurement, physiological interpretation, and clinical use. Taskforce of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 93: 1043–1065
Pagani M, Lucini D, Mela GS, Langewitz W, Malliani A 1994 Sympathetic overactivity in subjects complaining of unexplained fatigue. Clin Sci 87: 655–661
Stewart JM 2000 Autonomic nervous system dysfunction in adolescents with postural orthostatic tachycardia syndrome and chronic fatigue syndrome is characterized by attenuated vagal baroreflex and potentiated sympathetic vasomotion. Pediatr Res 48: 218–226
Stewart J, Weldon A, Arlisevky N, Li K, Munoz J 1998 Neurally mediated hypotension and autonomic dysfunction measured by heart rate variability during head-up tilt testing in children with chronic fatigue syndrome. Clin Auton Res 8: 221–230
De Becker P, Dendale P, De Meirleir K, Campine I, Vandenborne K, Hagers Y 1998 Autonomic testing in patients with chronic fatigue syndrome. Am J Med 105: 22S–26S
Freeman R, Komaroff AL 1997 Does the chronic fatigue syndrome involve the autonomic nervous system?. Am J Med 102: 357–364
Yataco A, Talo H, Rowe P, Kass DA, Berger RD, Calkins H 1997 Comparison of heart rate variability in patients with chronic fatigue syndrome and controls. Clin Auton Res 7: 293–297
Braun C, Kowallik P, Freking A, Hadeler D, Kniffki KD, Meesmann M 1998 Demonstration of nonlinear components in heart rate variability of healthy persons. Am J Physiol 275: H1577–H1584
Arroll B, Khin N, Kerse N 2003 Screening for depression in primary care with two verbally asked questions: cross sectional study. BMJ 327: 1144–1146
Pagani M, Lombardi F, Guzzetti S, Rimoldi O, Furlan R, Pizzinelli P, Sandrone G, Malfatto G, Dell'Orto S, Piccaluga E 1986 Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog. Circ Res 59: 178–193
Cevese A, Gulli G, Polati E, Gottin L, Grasso R 2001 Baroreflex and oscillation of heart period at 0.1 Hz studied by alpha-blockade and cross-spectral analysis in healthy humans. J Physiol 531: 235–244
Tulppo MP, Makikallio TH, Takala TE, Seppanen T, Huikuri HV 1996 Quantitative beat-to-beat analysis of heart rate dynamics during exercise. Am J Physiol 271: H244–H252
De Vito G, Galloway SD, Nimmo MA, Maas P, McMurray JJ 2002 Effects of central sympathetic inhibition on heart rate variability during steady-state exercise in healthy humans. Clin Physiol Funct Imaging 22: 32–38
Hirose M, Imai H, Ohmori M, Matsumoto Y, Amaya F, Hosokawa T, Tanaka Y 1998 Heart rate variability during chemical thoracic sympathectomy. Anesthesiology 89: 666–670
Woo MA, Stevenson WG, Moser DK, Trelease RB, Harper RM 1992 Patterns of beat-to-beat heart rate variability in advanced heart failure. Am Heart J 123: 704–710
Furlan R, Jacob G, Snell M, Robertson D, Porta A, Harris P, Mosqueda-Garcia R 1998 Chronic orthostatic intolerance: a disorder with discordant cardiac and vascular sympathetic control. Circulation 98: 2154–2159
Mosqueda-Garcia R, Furlan R, Fernandez-Violante R, Desai T, Snell M, Jarai Z, Ananthram V, Robertson RM, Robertson D 1997 Sympathetic and baroreceptor reflex function in neurally mediated syncope evoked by tilt. J Clin Invest 99: 2736–2744
Streeten DH 1990 Pathogenesis of hyperadrenergic orthostatic hypotension. Evidence of disordered venous innervation exclusively in the lower limbs. J Clin Invest 86: 1582–1588
Hoeldtke RD, Davis KM 1991 The orthostatic tachycardia syndrome: evaluation of autonomic function and treatment with octreotide and ergot alkaloids. J Clin Endocrinol Metab 73: 132–139
Stewart JM 2004 Chronic orthostatic intolerance and the postural tachycardia syndrome (POTS). J Pediatr 145: 725–730
Kosinski D, Grubb BP, Temesy-Armos P 1995 Pathophysiological aspects of neurocardiogenic syncope: current concepts and new perspectives. Pacing Clin Electrophysiol 18: 716–724
Newman D, Lurie K, Rosenqvist M, Washington C, Schwartz J, Scheinman MM 1993 Head-up tilt testing with and without isoproterenol infusion in healthy subjects of different ages. Pacing Clin Electrophysiol 16: 715–721
Stewart JM, Gewitz MH, Weldon A, Munoz J 1999 Patterns of orthostatic intolerance: the orthostatic tachycardia syndrome and adolescent chronic fatigue. J Pediatr 135: 218–225
Prodinger RJ, Reisdorff EJ 1998 Syncope in children. Emerg Med Clin North Am 16: 617–626
Wyller VB, Saul JP, Amlie JP, Thaulow E 2007 Sympathetic predominance of cardiovascular regulation during mild orthostatic stress in adolescents with chronic fatigue. Clin Physiol Funct Imaging 27: 231–238
Eckberg DL 1997 Sympathovagal balance: a critical appraisal. Circulation 96: 3224–3232
Parati G, Mancia G, Rienzo MD, Castiglioni P, Taylor JA, Studinger P 2006 Point:counterpoint: cardiovascular variability is/is not an index of autonomic control of circulation. J Appl Physiol 101: 676–682
De Lorenzo F, Xiao H, Mukherjee M, Harcup J, Suleiman S, Kadziola Z, Kakkar VV 1998 Chronic fatigue syndrome: physical and cardiovascular deconditioning. QJM 91: 475–481
Peckerman A, LaManca JJ, Qureishi B, Dahl KA, Golfetti R, Yamamoto Y, Natelson BH 2003 Baroreceptor reflex and integrative stress responses in chronic fatigue syndrome. Psychosom Med 65: 889–895
Arav-Boger R, Spirer Z 1995 Chronic fatigue syndrome: pediatric aspects. Isr J Med Sci 31: 330–334
Smith MS, Carter BD 2003 Chronic fatigue syndrome in adolescence. In: Johnson LA, Fennell PA, Taylors RR (eds) Handbook of Chronic Fatigue Syndrome. John Wiley & Sons, New York, pp 693–712
Acknowledgements
The authors thank Andrew Gray for his statistical assistance. They are grateful to the children and adolescents who participated in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by a University of Otago research grant.
Rights and permissions
About this article
Cite this article
Galland, B., Jackson, P., Sayers, R. et al. A Matched Case Control Study of Orthostatic Intolerance in Children/Adolescents With Chronic Fatigue Syndrome. Pediatr Res 63, 196–202 (2008). https://doi.org/10.1203/PDR.0b013e31815ed612
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/PDR.0b013e31815ed612
This article is cited by
-
An anthropogenic model of cardiovascular system adaptation to the Earth’s gravity as the conceptual basis of pathological anthropology
Journal of Physiological Anthropology (2021)
-
Heart rate and heart rate variability comparison between postural orthostatic tachycardia syndrome versus healthy participants; a systematic review and meta-analysis
BMC Cardiovascular Disorders (2019)
-
Blood pressure variability and closed-loop baroreflex assessment in adolescent chronic fatigue syndrome during supine rest and orthostatic stress
European Journal of Applied Physiology (2011)