Abstract
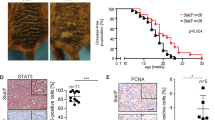
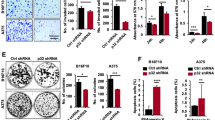
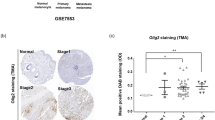
Melanomas are very aggressive neoplasms with notorious resistance to therapeutics. It was recently proposed that the remarkable phenotypic plasticity of melanoma cells allows for the rapid development of both resistance to chemotherapeutic drugs and invasive properties. Indeed, the capacity of melanoma cells to form distant metastases is the main cause of mortality in melanoma patients. Therefore, the identification of the mechanism controlling melanoma phenotype is of paramount importance. In the present report, we show that deletion of microphthalmia-associated transcription factor (MITF), the master gene in melanocyte differentiation, is sufficient to increase the metastatic potential of mouse and human melanoma cells. MITF silencing also increases fibronectin and Snail, two mesenchymal markers that might explain the increased invasiveness in vitro and in vivo. Furthermore, ablation of this population by Forskolin-induced differentiation or MITF-forced expression significantly decreases tumour and metastasis formation, suggesting that eradication of low-MITF cells might improve melanoma treatment. Moreover, we demonstrate that a hypoxic microenvironment decreases MITF expression through an indirect, hypoxia-inducible factor 1 (HIF1)α-dependant transcriptional mechanism, and increases the tumourigenic and metastatic properties of melanoma cells. We identified Bhlhb2, a new factor in melanoma biology, as the mediator of hypoxia/HIF1α inhibitory effect on MITF expression. Our results reveal a hypoxia–HIF1α–BHLHB2–MITF cascade controlling the phenotypic plasticity in melanoma cells and favouring metastasis development. Targeting this pathway might be helpful in the design of new anti-melanoma therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Agarwal KC, Parks Jr RE . (1983). Forskolin: a potential antimetastatic agent. Int J Cancer 32: 801–804.
Alonso SR, Tracey L, Ortiz P, Perez-Gomez B, Palacios J, Pollan M et al. (2007). A high-throughput study in melanoma identifies epithelial-mesenchymal transition as a major determinant of metastasis. Cancer Res 67: 3450–3460.
Bedogni B, Welford SM, Cassarino DS, Nickoloff BJ, Giaccia AJ, Powell MB . (2005). The hypoxic microenvironment of the skin contributes to Akt-mediated melanocyte transformation. Cancer Cell 8: 443–454.
Bedrosian I, Faries MB, Guerry Dt, Elenitsas R, Schuchter L, Mick R et al. (2000). Incidence of sentinel node metastasis in patients with thin primary melanoma (< or =1 mm) with vertical growth phase. Ann Surg Oncol 7: 262–267.
Berra E, Benizri E, Ginouves A, Volmat V, Roux D, Pouyssegur J . (2003). HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1alpha in normoxia. EMBO J 22: 4082–4090.
Bertolotto C, Abbe P, Hemesath TJ, Bille K, Fisher DE, Ortonne JP et al. (1998). Microphthalmia gene product as a signal transducer in cAMP-induced differentiation of melanocytes. J Cell Biol 142: 827–835.
Bhawal UK, Sato F, Arakawa Y, Fujimoto K, Kawamoto T, Tanimoto K et al. (2011). Basic helix-loop-helix transcription factor DEC1 negatively regulates cyclin D1. J Pathol 224: 420–429.
Botton T, Puissant A, Bahadoran P, Annicotte JS, Fajas L, Ortonne JP et al. (2009). In vitro and in vivo anti-melanoma effects of ciglitazone. J Invest Dermatol 129: 1208–1218.
Busca R, Ballotti R . (2000). Cyclic AMP a key messenger in the regulation of skin pigmentation. Pigment Cell Res 13: 60–69.
Carreira S, Goodall J, Aksan I, La Rocca SA, Galibert MD, Denat L et al. (2005). Mitf cooperates with Rb1 and activates p21Cip1 expression to regulate cell cycle progression. Nature 433: 764–769.
Carreira S, Goodall J, Denat L, Rodriguez M, Nuciforo P, Hoek KS et al. (2006). Mitf regulation of Dia1 controls melanoma proliferation and invasiveness. Genes Dev 20: 3426–3439.
Cheli Y, Guiliano S, Botton T, Rocchi S, Hofman V, Hofman P et al. (2011). Mitf is the key molecular switch between mouse or human melanoma initiating cells and their differentiated progeny. Oncogene 30: 2307–2318.
Cheli Y, Ohanna M, Ballotti R, Bertolotto C . (2010). Fifteen-year quest for microphthalmia-associated transcription factor target genes. Pigment Cell Melanoma Res 23: 27–40.
Cho Y, Noshiro M, Choi M, Morita K, Kawamoto T, Fujimoto K et al. (2009). The basic helix-loop-helix proteins differentiated embryo chondrocyte (DEC) 1 and DEC2 function as corepressors of retinoid X receptors. Mol Pharmacol 76: 1360–1369.
Choi SM, Cho HJ, Cho H, Kim KH, Kim JB, Park H . (2008). Stra13/DEC1 and DEC2 inhibit sterol regulatory element binding protein-1c in a hypoxia-inducible factor-dependent mechanism. Nucleic Acids Res 36: 6372–6385.
Corsetti RL, Allen HM, Wanebo HJ . (2000). Thin < or =1 mm level III and IV melanomas are higher risk lesions for regional failure and warrant sentinel lymph node biopsy. Ann Surg Oncol 7: 456–460.
Du J, Widlund HR, Horstmann MA, Ramaswamy S, Ross K, Huber WE et al. (2004). Critical role of CDK2 for melanoma growth linked to its melanocyte-specific transcriptional regulation by MITF. Cancer Cell 6: 565–576.
Ehata S, Hanyu A, Hayashi M, Aburatani H, Kato Y, Fujime M et al. (2007). Transforming growth factor-beta promotes survival of mammary carcinoma cells through induction of antiapoptotic transcription factor DEC1. Cancer Res 67: 9694–9703.
Eichhoff OM, Zipser MC, Xu M, Weeraratna AT, Mihic D, Dummer R et al. (2010). The immunohistochemistry of invasive and proliferative phenotype switching in melanoma: a case report. Melanoma Res 20: 349–355.
Fenouille N, Robert G, Tichet M, Puissant A, Dufies M, Rocchi S et al. (2011). The p53/p21(Cip1/Waf1) pathway mediates the effects of SPARC on melanoma cell cycle progression. Pigment Cell Melanoma Res 24: 219–232.
Ginouves A, Ilc K, Macias N, Pouyssegur J, Berra E . (2008). PHDs overactivation during chronic hypoxia ‘desensitizes’ HIFalpha and protects cells from necrosis. Proc Natl Acad Sci USA 105: 4745–4750.
Giuliano S, Cheli Y, Ohanna M, Bonet C, Beuret L, Bille K et al. (2010). Microphthalmia-associated transcription factor controls the DNA damage response and a lineage-specific senescence program in melanomas. Cancer Res 70: 3813–3822.
Gupta PB, Kuperwasser C, Brunet JP, Ramaswamy S, Kuo WL, Gray JW et al. (2005). The melanocyte differentiation program predisposes to metastasis after neoplastic transformation. Nat Genet 37: 1047–1054.
Hoek KS, Eichhoff OM, Schlegel NC, Dobbeling U, Kobert N, Schaerer L et al. (2008). In vivo switching of human melanoma cells between proliferative and invasive states. Cancer Res 68: 650–656.
Hoek KS, Goding CR . (2010). Cancer stem cells versus phenotype-switching in melanoma. Pigment Cell Melanoma Res 23: 746–759.
Honma S, Kawamoto T, Takagi Y, Fujimoto K, Sato F, Noshiro M et al. (2002). Dec1 and Dec2 are regulators of the mammalian molecular clock. Nature 419: 841–844.
King R, Googe PB, Weilbaecher KN, Mihm Jr MC, Fisher DE . (2001). Microphthalmia transcription factor expression in cutaneous benign, malignant melanocytic, and nonmelanocytic tumors. Am J Surg Pathol 25: 51–57.
Ledda MF, Adris S, Bravo AI, Kairiyama C, Bover L, Chernajovsky Y et al. (1997). Suppression of SPARC expression by antisense RNA abrogates the tumorigenicity of human melanoma cells. Nat Med 3: 171–176.
Loercher AE, Tank EM, Delston RB, Harbour JW . (2005). MITF links differentiation with cell cycle arrest in melanocytes by transcriptional activation of INK4A. J Cell Biol 168: 35–40.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY et al. (2008). The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133: 704–715.
Martin TA, Goyal A, Watkins G, Jiang WG . (2005). Expression of the transcription factors snail, slug, and twist and their clinical significance in human breast cancer. Ann Surg Oncol 12: 488–496.
Nakashima A, Kawamoto T, Honda KK, Ueshima T, Noshiro M, Iwata T et al. (2008). DEC1 modulates the circadian phase of clock gene expression. Mol Cell Biol 28: 4080–4092.
Olbryt M, Jarzab M, Jazowiecka-Rakus J, Simek K, Szala S, Sochanik A . (2006). Gene expression profile of B 16(F10) murine melanoma cells exposed to hypoxic conditions in vitro. Gene Exp 13: 191–203.
Radisky DC . (2005). Epithelial-mesenchymal transition. J Cell Sci 118: 4325–4326.
Robert G, Gaggioli C, Bailet O, Chavey C, Abbe P, Aberdam E et al. (2006). SPARC represses E-cadherin and induces mesenchymal transition during melanoma development. Cancer Res 66: 7516–7523.
Roesch A, Fukunaga-Kalabis M, Schmidt EC, Zabierowski SE, Brafford PA, Vultur A et al. (2010). A temporarily distinct subpopulation of slow-cycling melanoma cells is required for continuous tumor growth. Cell 141: 583–594.
Sanchez-Martin M, Rodriguez-Garcia A, Perez-Losada J, Sagrera A, Read AP, Sanchez-Garcia I . (2002). SLUG (SNAI2) deletions in patients with Waardenburg disease. Hum Mol Genet 11: 3231–3236.
Shaw HM, McCarthy WH, McCarthy SW, Milton GW . (1987). Thin malignant melanomas and recurrence potential. Arch Surg 122: 1147–1150.
Spira AI, Carducci MA . (2003). Differentiation therapy. Curr Opin Pharmacol 3: 338–343.
Wellbrock C, Marais R . (2005). Elevated expression of MITF counteracts B-RAF-stimulated melanocyte and melanoma cell proliferation. J Cell Biol 170: 703–708.
Acknowledgements
We thank Giorgetti-Peraldi Sophie, Regazzetti Claire and Karine Bille for their help. Bhlhb2 reporter plasmid and expression vector were a kind gift from Dr S Egan (Toronto, Canada). This work was supported by the ‘Fondation de France’, and Societé Française de Dermatologie. YC has been supported by ARC's fellowships. The authors greatly acknowledge the C3M Imaging core facility (microscopy and imaging platform Côte d'Azur, MICA) and the C3M animal room facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Cheli, Y., Giuliano, S., Fenouille, N. et al. Hypoxia and MITF control metastatic behaviour in mouse and human melanoma cells. Oncogene 31, 2461–2470 (2012). https://doi.org/10.1038/onc.2011.425
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.425
Keywords
This article is cited by
-
Dormancy of cutaneous melanoma
Cancer Cell International (2024)
-
Whole genome sequencing revealed genetic diversity, population structure, and selective signature of Panou Tibetan sheep
BMC Genomics (2023)
-
The microphthalmia-associated transcription factor is involved in gastrointestinal stromal tumor growth
Cancer Gene Therapy (2023)
-
Transglutaminase Type 2-MITF axis regulates phenotype switching in skin cutaneous melanoma
Cell Death & Disease (2023)
-
Bone Morphogenic Protein Signaling and Melanoma
Current Treatment Options in Oncology (2021)