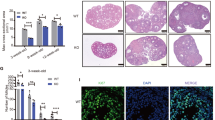
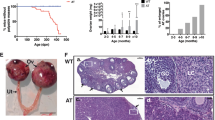
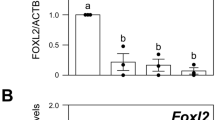
Abstract
Some mutations in FOXL2 result in premature ovarian failure accompanied by blepharophimosis, ptosis, epicanthus inversus syndrome type I disease, and FOXL2-null mice exhibit developmental defects in granulosa cells. Recently, FOXL2 c.402C>G, a new somatic mutation that leads to a p.C134W change, was found in the majority of adult-type ovarian granulosa cell tumors (GCTs). In this study, we investigated the possible mechanisms by which the C134W mutation contributes to the development of GCTs. Wild-type (WT) and mutant FOXL2 displayed differential apoptotic activities. Specifically, WT FOXL2 induced significant granulosa cell death, but the mutant exhibited minimal cell death. The FOXL2-induced apoptotic response was greatly dependent on caspase 8, BID and BAK because the depletion of any of these three proteins inhibited FOXL2 from eliciting the full apoptotic response. Activation of caspase 8 and subsequent increased production of truncated BID, and oligomerization of BAK, and release of cytochrome c were all associated with the apoptosis induced by WT FOXL2 expression. In contrast, the mutant FOXL2 was unable to elicit the full array of apoptotic signaling responses. In addition, we found differential TNF-R1 (tumor necrosis factor-receptor 1) and Fas (CD95/APO-1) upregulation between the WT and the mutant, and the silencing of TNF-R1 or Fas and the blockage of the death signaling mediated by TNF-R1 or Fas using TNF-Fc or Fas-Fc, respectively, resulted in significant attenuations of FOXL2-induced apoptosis. Moreover, granulosa cells that expressed either WT FOXL2 or mutant exhibited distinct cell death sensitivities on activation of death receptors and deprivation of serum. Thus, the differential activities of FOXL2 and its mutant may partially account for the pathophysiology of GCT development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Arnoult D . (2008). Apoptosis-associated mitochondrial outer membrane permeabilization assays. Methods 44: 229–234.
Benayoun BA, Caburet S, Dipietromaria A, Georges A, D'Haene B, Pandaranayaka PJ et al. (2010). Functional exploration of the adult ovarian granulosa cell tumor-associated somatic FOXL2 mutation p.Cys134Trp (c.402C>G). PLoS One 5: e8789.
Chang DW, Xing Z, Pan Y, Algeciras-Schimnich A, Barnhart BC, Yaish-Ohad S et al. (2002). c-FLIP(L) is a dual function regulator for caspase-8 activation and CD95-mediated apoptosis. EMBO J 21: 3704–3714.
Chipuk JE, Bouchier-Hayes L, Green DR . (2006). Mitochondrial outer membrane permeabilization during apoptosis: the innocent bystander scenario. Cell Death Differ 13: 1396–1402.
Crisponi L, Deiana M, Loi A, Chiappe F, Uda M, Amati P et al. (2001). The putative forkhead transcription factor FOXL2 is mutated in blepharophimosis/ptosis/epicanthus inversus syndrome. Nat Genet 27: 159–166.
Dewson G, Kratina T, Sim HW, Puthalakath H, Adams JM, Colman PM et al. (2008). To trigger apoptosis, Bak exposes its BH3 domain and homodimerizes via BH3:groove interactions. Mol Cell 30: 369–380.
Ganju N, Eastman A . (2003). Zinc inhibits Bax and Bak activation and cytochrome c release induced by chemical inducers of apoptosis but not by death-receptor-initiated pathways. Cell Death Differ 10: 652–661.
Green DR, Kroemer G . (2005). Pharmacological manipulation of cell death: clinical applications in sight? J Clin Invest 115: 2610–2617.
Guicciardi ME, Gores GJ . (2009). Life and death by death receptors. FASEB J 23: 1625–1637.
Hail Jr N . (2005). Mitochondria: a novel target for the chemoprevention of cancer. Apoptosis 10: 687–705.
Jabbour AM, Heraud JE, Daunt CP, Kaufmann T, Sandow J, O'Reilly LA et al. (2009). Puma indirectly activates Bax to cause apoptosis in the absence of Bid or Bim. Cell Death Differ 16: 555–563.
Kang BP, Frencher S, Reddy V, Kessler A, Malhotra A, Meggs LG . (2003). High glucose promotes mesangial cell apoptosis by oxidant-dependent mechanism. Am J Physiol Renal Physiol 284: F455–F466.
Kim MS, Hur SY, Yoo NJ, Lee SH . (2010). Mutational analysis of FOXL2 codon 134 in granulosa cell tumour of ovary and other human cancers. J Pathol 221: 147–152.
Kornblau SM, Singh N, Qiu Y, Chen W, Zhang N, Coombes KR . (2010). Highly phosphorylated FOXO3A is an adverse prognostic factor in acute myeloid leukemia. Clin Cancer Res 16: 1865–1874.
Lee K, Pisarska MD, Ko JJ, Kang Y, Yoon S, Ryou SM et al. (2005). Transcriptional factor FOXL2 interacts with DP103 and induces apoptosis. Biochem Biophys Res Commun 336: 876–881.
Li J, Yuan J . (2008). Caspases in apoptosis and beyond. Oncogene 27: 6194–6206.
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES et al. (1997). Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91: 479–489.
Li P, Nijhawan D, Wang X . (2004). Mitochondrial activation of apoptosis. Cell 116: S57–S59.
Mahmood Z, Shukla Y . (2010). Death receptors: targets for cancer therapy. Exp Cell Res 316: 887–899.
Muzio M . (1998). Signalling by proteolysis: death receptors induce apoptosis. Int J Clin Lab Res 28: 141–147.
Muzio M, Chinnaiyan AM, Kischkel FC, O'Rourke K, Shevchenko A, Ni J et al. (1996). FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85: 817–827.
Naka K, Hoshii T, Muraguchi T, Tadokoro Y, Ooshio T, Kondo Y et al. (2010). TGF-beta-FOXO signalling maintains leukaemia-initiating cells in chronic myeloid leukaemia. Nature 463: 676–680.
Nishi Y, Yanase T, Mu Y, Oba K, Ichino I, Saito M et al. (2001). Establishment and characterization of a steroidogenic human granulosa-like tumor cell line, KGN, that expresses functional follicle-stimulating hormone receptor. Endocrinology 142: 437–445.
Obexer P, Geiger K, Ambros PF, Meister B, Ausserlechner MJ . (2007). FKHRL1-mediated expression of Noxa and Bim induces apoptosis via the mitochondria in neuroblastoma cells. Cell Death Differ 14: 534–547.
Paik JH, Kollipara R, Chu G, Ji H, Xiao Y, Ding Z et al. (2007). FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell 128: 309–323.
Park M, Shin E, Won M, Kim JH, Go H, Kim HL et al. (2010). FOXL2 interacts with steroidogenic factor-1 (SF-1) and represses SF-1-induced CYP17 transcription in granulosa cells. Mol Endocrinol 24: 1024–1036.
Perez D, White E . (2000). TNF-alpha signals apoptosis through a bid-dependent conformational change in Bax that is inhibited by E1B 19K. Mol Cell 6: 53–63.
Pisarska MD, Bae J, Klein C, Hsueh AJ . (2004). Forkhead l2 is expressed in the ovary and represses the promoter activity of the steroidogenic acute regulatory gene. Endocrinology 145: 3424–3433.
Schrader KA, Gorbatcheva B, Senz J, Heravi-Moussavi A, Melnyk N, Salamanca C et al. (2009). The specificity of the FOXL2 c.402C>G somatic mutation: a survey of solid tumors. PLoS One 4: e7988.
Schumer ST, Cannistra SA . (2003). Granulosa cell tumor of the ovary. J Clin Oncol 21: 1180–1189.
Shah SP, Kobel M, Senz J, Morin RD, Clarke BA, Wiegand KC et al. (2009). Mutation of FOXL2 in granulosa-cell tumors of the ovary. N Engl J Med 360: 2719–2729.
Uda M, Ottolenghi C, Crisponi L, Garcia JE, Deiana M, Kimber W et al. (2004). Foxl2 disruption causes mouse ovarian failure by pervasive blockage of follicle development. Hum Mol Genet 13: 1171–1181.
Wei MC, Lindsten T, Mootha VK, Weiler S, Gross A, Ashiya M et al. (2000). tBID, a membrane-targeted death ligand, oligomerizes BAK to release cytochrome c. Genes Dev 14: 2060–2071.
Yin XM, Wang K, Gross A, Zhao Y, Zinkel S, Klocke B et al. (1999). Bid-deficient mice are resistant to Fas-induced hepatocellular apoptosis. Nature 400: 886–891.
Zou H, Li Y, Liu X, Wang X . (1999). An APAF-1.cytochrome c multimeric complex is a functional apoptosome that activates procaspase-9. J Biol Chem 274: 11549–11556.
Acknowledgements
This work was supported by the Basic Science Research Program (2009-0066320) and the Priority Research Centers Program (2009-0093821) through the National Research Foundation of Korea (NRF) of the Ministry of Education, Science and Technology and by a grant (A084923) from the Korea Healthcare Technology R&D Project from the Ministry of Health, Welfare and Family Affairs.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kim, JH., Yoon, S., Park, M. et al. Differential apoptotic activities of wild-type FOXL2 and the adult-type granulosa cell tumor-associated mutant FOXL2 (C134W). Oncogene 30, 1653–1663 (2011). https://doi.org/10.1038/onc.2010.541
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.541
Keywords
This article is cited by
-
A cellular model provides insights into the pathogenicity of the oncogenic FOXL2 somatic variant p.Cys134Trp
British Journal of Cancer (2024)
-
Immunohistochemical markers of prognosis in adult granulosa cell tumors of the ovary – a review
Journal of Ovarian Research (2023)
-
Incidental FOXL2 mutated adult granulosa cell tumour of the ovary with thecoma-like foci
Virchows Archiv (2023)
-
Aberrant granulosa cell-fate related to inactivated p53/Rb signaling contributes to granulosa cell tumors and to FOXL2 downregulation in the mouse ovary
Oncogene (2020)
-
New STAT3-FOXL2 pathway and its function in cancer cells
BMC Molecular and Cell Biology (2019)