Abstract
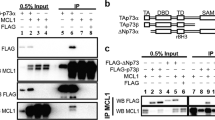
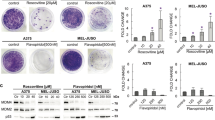
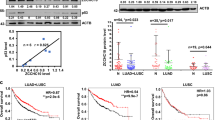
p73 encodes multiple functionally distinct isoforms. Proapoptotic TAp73 isoforms contain a transactivation (TA) domain, and like p53, have tumor suppressor properties and are activated by chemotherapies to induce cell death. In contrast, antiapoptotic ΔNp73 isoforms lack the TA domain and are dominant-negative inhibitors of p53 and TAp73. ΔNp73 proteins are overexpressed in a variety of tumors including neuroblastoma. Thus, identification of drugs that upregulate TAp73 and/or downregulate ΔNp73 represents a potential therapeutic strategy. Here, we report that cyclooxygenase (COX) inhibitors induce apoptosis independent of p53, and differentially modulate endogenous p73 isoforms in neuroblastoma and other tumors. COX inhibitor-mediated apoptosis is associated with the induction of TAp73β and its target genes. COX inhibitors also downregulate the alternative-spliced ΔNp73AS isoforms, Δexon2 and Δexon2/3. Furthermore, forced expression of ΔNp73AS results in diminished apoptosis in response to the selective COX-2 inhibitor celecoxib. Celecoxib-mediated downregulation of ΔNp73AS is associated with decreased E2F1 levels and diminished E2F1 activation of the p73 promoter. These results provide the first evidence that COX inhibitors differentially modulate p73 isoforms leading to enhanced apoptosis, and support the potential use of COX inhibitors as novel regulators of p73 to enhance chemosensitivity in tumors with deregulated E2F1 and in those with wild-type (wt) or mutant p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Amin AR, Paul RK, Thakur VS, Agarwal ML . (2007). A novel role for p73 in the regulation of Akt-Foxo1a-Bim signaling and apoptosis induced by the plant lectin, Concanavalin A. Cancer Res 67: 5617–5621.
Balint E, Phillips AC, Kozlov S, Stewart CL, Vousden KH . (2002). Induction of p57(KIP2) expression by p73beta. Proc Natl Acad Sci USA 99: 3529–3534.
Bock JM, Menon SG, Sinclair LL, Bedford NS, Goswami PC, Domann FE et al. (2007). Celecoxib toxicity is cell cycle phase specific. Cancer Res 67: 3801–3808.
Buhlmann S, Putzer BM . (2008). DNp73 a matter of cancer: mechanisms and clinical implications. Biochim Biophys Acta 1785: 207–216.
Casciano I, Mazzocco K, Boni L, Pagnan G, Banelli B, Allemanni G et al. (2002). Expression of DeltaNp73 is a molecular marker for adverse outcome in neuroblastoma patients. Cell Death Differ 9: 246–251.
Concin N, Hofstetter G, Berger A, Gehmacher A, Reimer D, Watrowski R et al. (2005). Clinical relevance of dominant-negative p73 isoforms for responsiveness to chemotherapy and survival in ovarian cancer: evidence for a crucial p53-p73 cross-talk in vivo. Clin Cancer Res 11: 8372–8383.
Dominguez G, Garcia JM, Pena C, Silva J, Garcia V, Martinez L et al. (2006). DeltaTAp73 upregulation correlates with poor prognosis in human tumors: putative in vivo network involving p73 isoforms, p53, and E2F-1. J Clin Oncol 24: 805–815.
Fillippovich I, Sorokina N, Gatei M, Haupt Y, Hobson K, Moallem E et al. (2001). Transactivation-deficient p73alpha (p73Deltaexon2) inhibits apoptosis and competes with p53. Oncogene 20: 514–522.
Flores ER, Sengupta S, Miller JB, Newman JJ, Bronson R, Crowley D et al. (2005). Tumor predisposition in mice mutant for p63 and p73: evidence for broader tumor suppressor functions for the p53 family. Cancer Cell 7: 363–373.
Gonzalez S, Perez-Perez MM, Hernando E, Serrano M, Cordon-Cardo C . (2005). p73beta-mediated apoptosis requires p57kip2 induction and IEX-1 inhibition. Cancer Res 65: 2186–2192.
Grosch S, Maier TJ, Schiffmann S, Geisslinger G . (2006). Cyclooxygenase-2 (COX-2)-independent anticarcinogenic effects of selective COX-2 inhibitors. J Natl Cancer Inst 98: 736–747.
Hansford LM, McKee AE, Zhang L, George RE, Gerstle JT, Thorner PS et al. (2007). Neuroblastoma cells isolated from bone marrow metastases contain a naturally enriched tumor-initiating cell. Cancer Res 67: 11234–11243.
Irwin M, Marin MC, Phillips AC, Seelan RS, Smith DI, Liu W et al. (2000). Role for the p53 homologue p73 in E2F-1-induced apoptosis. Nature 407: 645–648.
Irwin MS . (2004). Family feud in chemosensitvity: p73 and mutant p53. Cell Cycle 3: 319–323.
Irwin MS, Kondo K, Marin MC, Cheng LS, Hahn WC, Kaelin Jr WG . (2003). Chemosensitivity linked to p73 function. Cancer Cell 3: 403–410.
Johnsen JI, Lindskog M, Ponthan F, Pettersen I, Elfman L, Orrego A et al. (2004). Cyclooxygenase-2 is expressed in neuroblastoma, and nonsteroidal anti-inflammatory drugs induce apoptosis and inhibit tumor growth in vivo. Cancer Res 64: 7210–7215.
Johnson AJ, Smith LL, Zhu J, Heerema NA, Jefferson S, Mone A et al. (2005). A novel celecoxib derivative, OSU03012, induces cytotoxicity in primary CLL cells and transformed B-cell lymphoma cell line via a caspase- and Bcl-2-independent mechanism. Blood 105: 2504–2509.
Kashfi K, Rigas B . (2005). Non-COX-2 targets and cancer: expanding the molecular target repertoire of chemoprevention. Biochem Pharmacol 70: 969–986.
Lau L, Hansford LM, Cheng LS, Hang M, Baruchel S, Kaplan DR et al. (2007). Cyclooxygenase inhibitors modulate the p53>HDM2 pathway and enhance chemotherapy-induced apoptosis in neuroblastoma. Oncogene 26: 1920–1931.
Lau LM, Nugent JK, Zhao X, Irwin MS . (2008). HDM2 antagonist Nutlin-3 disrupts p73-HDM2 binding and enhances p73 function. Oncogene 27: 997–1003.
Maris JM, Hogarty MD, Bagatell R, Cohn SL . (2007). Neuroblastoma. Lancet 369: 2106–2120.
Muller M, Schilling T, Sayan AE, Kairat A, Lorenz K, Schulze-Bergkamen H et al. (2005). TAp73>Delta Np73 influences apoptotic response, chemosensitivity and prognosis in hepatocellular carcinoma. Cell Death Differ 12: 1564–1577.
Ponthan F, Wickstrom M, Gleissman H, Fuskevag OM, Segerstrom L, Sveinbjornsson B et al. (2007). Celecoxib prevents neuroblastoma tumor development and potentiates the effect of chemotherapeutic drugs in vitro and in vivo. Clin Cancer Res 13: 1036–1044.
Pozniak CD, Radinovic S, Yang A, McKeon F, Kaplan DR, Miller FD . (2000). An anti-apoptotic role for the p53 family member, p73, during developmental neuron death. Science 289: 304–306.
Putzer BM . (2007). E2F1 death pathways as targets for cancer therapy. J Cell Mol Med 11: 239–251.
Putzer BM, Tuve S, Tannapfel A, Stiewe T . (2003). Increased DeltaN-p73 expression in tumors by upregulation of the E2F1-regulated, TA-promoter-derived DeltaN′-p73 transcript. Cell Death Differ 10: 612–614.
Sayan AE, Paradisi A, Vojtesek B, Knight RA, Melino G, Candi E . (2005). New antibodies recognizing p73: comparison with commercial antibodies. Biochem Biophys Res Commun 330: 186–193.
Stempak D, Gammon J, Klein J, Koren G, Baruchel S . (2002). Single-dose and steady-state pharmacokinetics of celecoxib in children. Clin Pharmacol Ther 72: 490–497.
Stiewe T, Putzer BM . (2000). Role of the p53-homologue p73 in E2F1-induced apoptosis. Nat Genet 26: 464–469.
Stiewe T, Theseling CC, Putzer BM . (2002). Transactivation-deficient Delta TA-p73 inhibits p53 by direct competition for DNA binding: implications for tumorigenesis. J Biol Chem 277: 14177–14185.
Thun MJ, Henley SJ, Patrono C . (2002). Nonsteroidal anti-inflammatory drugs as anticancer agents: mechanistic, pharmacologic, and clinical issues. J Natl Cancer Inst 94: 252–266.
Xue C, Haber M, Flemming C, Marshall GM, Lock RB, MacKenzie KL et al. (2007). p53 determines multidrug sensitivity of childhood neuroblastoma. Cancer Res 67: 10351–10360.
Zaika AI, Slade N, Erster SH, Sansome C, Joseph TW, Pearl M et al. (2002). DeltaNp73, a dominant-negative inhibitor of wild-type p53 and TAp73, is up-regulated in human tumors. J Exp Med 196: 765–780.
Zhu J, Huang JW, Tseng PH, Yang YT, Fowble J, Shiau CW et al. (2004). From the cyclooxygenase-2 inhibitor celecoxib to a novel class of 3-phosphoinositide-dependent protein kinase-1 inhibitors. Cancer Res 64: 4309–4318.
Acknowledgements
This study was supported by the Canadian Cancer Society and the National Cancer Institute of Canada (CCS Grant no. 019274, MS Irwin), the James Fund for Neuroblastoma Research at SickKids Foundation and Lloyd Carr-Harris Foundation. MS Irwin is a Canada Research Chair in Cancer Biology. L Lau was supported by the Terry Fox Foundation Clinical Research Fellowship (National Cancer Institute of Canada).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Lau, L., Wolter, J., Lau, J. et al. Cyclooxygenase inhibitors differentially modulate p73 isoforms in neuroblastoma. Oncogene 28, 2024–2033 (2009). https://doi.org/10.1038/onc.2009.59
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.59
Keywords
This article is cited by
-
Apoptosis induction by combination of drugs or a conjugated molecule associating non-steroidal anti-inflammatory and nitric oxide donor effects in medullary thyroid cancer models: implication of the tumor suppressor p73
Thyroid Research (2015)
-
A balancing act: orchestrating amino-truncated and full-length p73 variants as decisive factors in cancer progression
Oncogene (2015)
-
Attenuation of inflammatory-mediated neurotoxicity by Saururus chinensis extract in LPS-induced BV-2 microglia cells via regulation of NF-κB signaling and anti-oxidant properties
BMC Complementary and Alternative Medicine (2014)
-
Functions, divergence and clinical value of TAp73 isoforms in cancer
Cancer and Metastasis Reviews (2013)
-
Analysis of the intracellular localization of p73 N-terminal protein isoforms TAp73 and ∆Np73 in medulloblastoma cell lines
Journal of Molecular Histology (2010)