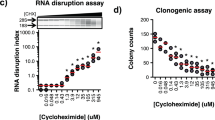
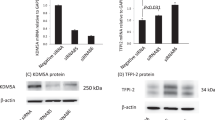
Abstract
The PPM1D gene is aberrantly amplified in a range of common cancers and encodes a protein phosphatase that is a potential therapeutic target. However, the issue of whether inhibition of PPM1D in human tumour cells that overexpress this protein compromises their viability has not yet been fully addressed. We show here, using an RNA interference (RNAi) approach, that inhibition of PPM1D can indeed reduce the viability of human tumour cells and that this effect is selective; tumour cell lines that overexpress PPM1D are sensitive to PPM1D inhibition whereas cell lines with normal levels are not. Loss of viability associated with PPM1D RNAi in human tumour cells occurs via the activation of the kinase P38. To identify chemical inhibitors of PPM1D, a high-throughput screening of a library of small molecules was performed. This strategy successfully identified a compound that selectively reduces viability of human tumour cell lines that overexpress PPM1D. As expected of a specific inhibitor, the toxicity to PPM1D overexpressing cell lines after inhibitor treatment is P38 dependent. These results further validate PPM1D as a therapeutic target and identify a proof-of-principle small molecule inhibitor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Archer S, Miller KJ, Rej R, Periana C, Fricker L . (1982). Ring-hydroxylated analogues of lucanthone as antitumor agents. J Med Chem 25: 220–227.
Belova GI, Demidov ON, Fornace Jr AJ, Bulavin DV . (2005). Chemical inhibition of Wip1 phosphatase contributes to suppression of tumorigenesis. Cancer Biol Ther 4: 1154–1158.
Brody LC . (2005). Treating cancer by targeting a weakness. N Engl J Med 353: 949–950.
Brummelkamp TR, Bernards R, Agami R . (2002). A system for stable expression of short interfering RNAs in mammalian cells. Science 296: 550–553.
Bulavin DV, Demidov ON, Saito S, Kauraniemi P, Phillips C, Amundson SA et al. (2002). Amplification of PPM1D in human tumors abrogates p53 tumor-suppressor activity. Nat Genet 31: 210–215.
Bulavin DV, Phillips C, Nannenga B, Timofeev O, Donehower LA, Anderson CW et al. (2004). Inactivation of the Wip1 phosphatase inhibits mammary tumorigenesis through p38 MAPK-mediated activation of the p16(Ink4a)-p19(Arf) pathway. Nat Genet 36: 343–350.
Burns S, Travers J, Collins I, Rowlands MG, Newbatt Y, Thompson N et al. (2006). Identification of small-molecule inhibitors of protein kinase B (PKB/AKT) in an AlphaScreenTM high-throughput screen. J Biomol Screen 11: 822–827.
Cheng JC, Fang JG, Chen WF, Zhou B, Yang L, Liu ZL . (2006). Structure-activity relationship studies of resveratrol and its analogues by the reaction kinetics of low density lipoprotein peroxidation. Bioorg Chem 34: 142–157.
Choi J, Nannenga B, Demidov ON, Bulavin DV, Cooney A, Brayton C et al. (2002). Mice deficient for the wild-type p53-induced phosphatase gene (Wip1) exhibit defects in reproductive organs, immune function, and cell cycle control. Mol Cell Biol 22: 1094–1105.
Collins I, Workman P . (2006). New approaches to molecular cancer therapeutics. Nat Chem Biol 2: 689–700.
Demidov ON, Kek C, Shreeram S, Timofeev O, Fornace AJ, Appella E et al. (2007). The role of the MKK6/p38 MAPK pathway in Wip1-dependent regulation of ErbB2-driven mammary gland tumorigenesis. Oncogene 26: 2502–2506.
Fiscella M, Zhang H, Fan S, Sakaguchi K, Shen S, Mercer WE et al. (1997). Wip1, a novel human protein phosphatase that is induced in response to ionizing radiation in a p53-dependent manner. Proc Natl Acad Sci USA 94: 6048–6053.
Fitzgerald CE, Patel SB, Becker JW, Cameron PM, Zaller D, Pikounis VB et al. (2003). Structural basis for p38alpha MAP kinase quinazolinone and pyridol-pyrimidine inhibitor specificity. Nat Struct Biol 10: 764–769.
Fujimoto H, Onishi N, Kato N, Takekawa M, Xu XZ, Kosugi A et al. (2006). Regulation of the antioncogenic Chk2 kinase by the oncogenic Wip1 phosphatase. Cell Death Differ 13: 1170–1180.
Gallego M, Virshup DM . (2005). Protein serine/threonine phosphatases: life, death, and sleeping. Curr Opin Cell Biol 17: 197–202.
Harrison M, Li J, Degenhardt Y, Hoey T, Powers S . (2004). Wip1-deficient mice are resistant to common cancer genes. Trends Mol Med 10: 359–361.
Hirasawa A, Saito-Ohara F, Inoue J, Aoki D, Susumu N, Yokoyama T et al. (2003). Association of 17q21–q24 gain in ovarian clear cell adenocarcinomas with poor prognosis and identification of PPM1D and APPBP2 as likely amplification targets. Clin Cancer Res 9: 1995–2004.
Li J, Yang Y, Peng Y, Austin RJ, van Eyndhoven WG, Nguyen KC et al. (2002). Oncogenic properties of PPM1D located within a breast cancer amplification epicenter at 17q23. Nat Genet 31: 133–134.
Lu X, Bocangel D, Nannenga B, Yamaguchi H, Appella E, Donehower LA . (2004). The p53-induced oncogenic phosphatase PPM1D interacts with uracil DNA glycosylase and suppresses base excision repair. Mol Cell 15: 621–634.
Lu X, Nannenga B, Donehower LA . (2005). PPM1D dephosphorylates Chk1 and p53 and abrogates cell cycle checkpoints. Genes Dev 19: 1162–1174.
Manning BD, Tee AR, Logsdon MN, Blenis J, Cantley LC . (2002). Identification of the tuberous sclerosis complex-2 tumor suppressor gene product tuberin as a target of the phosphoinositide 3-kinase/akt pathway. Mol Cell 10: 151–162.
McGovern SL, Caselli E, Grigorieff N, Shoichet BK . (2002). A common mechanism underlying promiscuous inhibitors from virtual and high-throughput screening. J Med Chem 45: 1712–1722.
McGovern SL, Helfand BT, Feng B, Shoichet BK . (2003). A specific mechanism of nonspecific inhibition. J Med Chem 46: 4265–4272.
Mendrzyk F, Radlwimmer B, Joos S, Kokocinski F, Benner A, Stange DE et al. (2005). Genomic and protein expression profiling identifies CDK6 as novel independent prognostic marker in medulloblastoma. J Clin Oncol 23: 8853–8862.
Nannenga B, Lu X, Dumble M, Van Maanen M, Nguyen TA, Sutton R et al. (2006). Augmented cancer resistance and DNA damage response phenotypes in PPM1D null mice. Mol Carcinog 45: 594–604.
Oliva-Trastoy M, Berthonaud V, Chevalier A, Ducrot C, Marsolier-Kergoat MC, Mann C et al. (2006). The Wip1 phosphatase (PPM1D) antagonizes activation of the Chk2 tumour suppressor kinase. Oncogene 26: 1449–1458.
Proia DA, Nannenga BW, Donehower LA, Weigel NL . (2006). Dual roles for the phosphatase PPM1D in regulating progesterone receptor function. J Biol Chem 281: 7089–7101.
Ryan AJ, Gray NM, Lowe PN, Chung CW . (2003). Effect of detergent on ‘promiscuous’ inhibitors. J Med Chem 46: 3448–3451.
Saito-Ohara F, Imoto I, Inoue J, Hosoi H, Nakagawara A, Sugimoto T et al. (2003). PPM1D is a potential target for 17q gain in neuroblastoma. Cancer Res 63: 1876–1883.
Scanlon W . (1973). Patent. Ger Offen DE 2245518/19730426.
Schmid AC, Woscholski R . (2004). Phosphatases as small-molecule targets: inhibiting the endogenous inhibitors of kinases. Biochem Soc Trans 32: 348–349.
Shreeram S, Demidov ON, Hee WK, Yamaguchi H, Onishi N, Kek C et al. (2006). Wip1 phosphatase modulates ATM-dependent signaling pathways. Mol Cell 23: 757–764.
Takekawa M, Adachi M, Nakahata A, Nakayama I, Itoh F, Tsukuda H et al. (2000). p53-inducible wip1 phosphatase mediates a negative feedback regulation of p38 MAPK-p53 signaling in response to UV radiation. EMBO J 19: 6517–6526.
Von Leoprechting A, Kumpf R, Menzel S, Reulle D, Griebel R, Valler MJ et al. (2004). Miniaturization and validation of a high-throughput serine kinase assay using the AlphaScreen platform. J Biomol Screen 9: 719–725.
Warner G, Illy C, Pedro L, Roby P, Bosse R . (2004). AlphaScreen kinase HTS platforms. Curr Med Chem 11: 721–730.
Yamaguchi H, Durell SR, Feng H, Bai Y, Anderson CW, Appella E . (2006). Development of a substrate-based cyclic phosphopeptide inhibitor of protein phosphatase 2Cdelta, Wip1. Biochemistry 45: 13193–13202.
Yoda A, Xu XZ, Onishi N, Toyoshima K, Fujimoto H, Kato N et al. (2006). Intrinsic kinase activity and SQ/TQ domain of Chk2 kinase as well as N-terminal domain of Wip1 phosphatase are required for regulation of Chk2 by Wip1. J Biol Chem 281: 24847–24862.
Yu E, Ahn YS, Jang SJ, Kim MJ, Yoon HS, Gong G et al. (2006). Overexpression of the wip1 gene abrogates the p38 MAPK/p53/Wip1 pathway and silences p16 expression in human breast cancers. Breast Cancer Res Treat 101: 269–278.
Zhang JH, Chung TD, Oldenburg KR . (1999). A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J Biomol Screen 4: 67–73.
Acknowledgements
We thank CJ Marshall and J Metcalfe (The Institute of Cancer Research, UK) for materials. This work was supported by Cancer Research UK (grant numbers C309/A2187 and C309/A8274) and Breakthrough Breast Cancer. PW is a Cancer Research UK Life Fellow.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Rights and permissions
About this article
Cite this article
Rayter, S., Elliott, R., Travers, J. et al. A chemical inhibitor of PPM1D that selectively kills cells overexpressing PPM1D. Oncogene 27, 1036–1044 (2008). https://doi.org/10.1038/sj.onc.1210729
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210729
Keywords
This article is cited by
-
Allosteric inhibition of PPM1D serine/threonine phosphatase via an altered conformational state
Nature Communications (2022)
-
PPM1D mutations are oncogenic drivers of de novo diffuse midline glioma formation
Nature Communications (2022)
-
WIP1 phosphatase as pharmacological target in cancer therapy
Journal of Molecular Medicine (2017)
-
WIP1 modulates responsiveness to Sonic Hedgehog signaling in neuronal precursor cells and medulloblastoma
Oncogene (2016)
-
PPM1D phosphatase, a target of p53 and RBM38 RNA-binding protein, inhibits p53 mRNA translation via dephosphorylation of RBM38
Oncogene (2015)