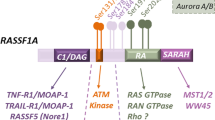
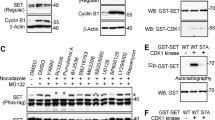
Abstract
The RAS association domain family 1A (RASSF1A) gene is located at chromosome 3p21.3 within a specific area of common heterozygous and homozygous deletions. RASSF1A frequently undergoes promoter methylation-associated inactivation in human cancers. Rassf1a−/− mice are prone to both spontaneous and carcinogen-induced tumorigenesis, supporting the notion that RASSF1A is a tumor suppressor. However, it is not fully understood how RASSF1A is involved in tumor suppression pathways. Here we show that overexpression of RASSF1A inhibits centrosome separation. RASSF1A interacts with Aurora-A, a mitotic kinase. Surprisingly, knockdown of RASSF1A by siRNA led to reduced activation of Aurora-A, whereas overexpression of RASSF1A resulted in increased activation of Aurora-A, suggesting that RASSF1A is involved in Aurora-A activation. Like other Aurora-A activators, RASSF1A was also a substrate of Aurora-A in vitro. The failure of recombinant RASSF1A to activate recombinant Aurora-A indicates that RASSF1A may not activate Aurora-A directly and suggests that RASSF1A may function as a scaffold to bring together Aurora-A and its activator(s). Inhibition of centrosome separation by RASSF1A overexpression is most likely a consequence of hyperstabilization of microtubules by this protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- APC:
-
anaphase-promoting complex
- DAPI:
-
4,6-diamidino-2-phenylindole
- MBP:
-
maltose-binding protein
- MEF:
-
mouse embryonic fibroblast
- MTOC:
-
microtubule-organizing center
- NORE:
-
novel Ras effector
- RASSF1A:
-
RAS association domain family 1A
References
Abbondanzo SJ, Gadi I, Stewart CL . (1993). Derivation of embryonic stem cell lines. Methods Enzymol 225: 803–823.
Agathanggelou A, Cooper WN, Latif F . (2005). Role of the Ras-association domain family 1 tumor suppressor gene in human cancers. Cancer Res 65: 3497–3508.
Anand S, Penrhyn-Lowe S, Venkitaraman AR . (2003). AURORA-A amplification overrides the mitotic spindle assembly checkpoint, inducing resistance to Taxol. Cancer Cell 3: 51–62.
Andrews PD, Knatko E, Moore WJ, Swedlow JR . (2003). Mitotic mechanics: the auroras come into view. Curr Opin Cell Biol 15: 672–683.
Bischoff JR, Anderson L, Zhu Y, Mossie K, Ng L, Souza B et al. (1998). A homologue of Drosophila aurora kinase is oncogenic and amplified in human colorectal cancers. EMBO J 17: 3052–3065.
Blangy A, Lane HA, d’Herin P, Harper M, Kress M, Nigg EA . (1995). Phosphorylation by p34cdc2 regulates spindle association of human Eg5, a kinesin-related motor essential for bipolar spindle formation in vivo. Cell 83: 1159–1169.
Boveri T . (1888). Zellenstudien II. Die Befruchtung und Teilung des Eies von Ascaris megalocephala. Jena Zeit Naturw 22: 685–882.
Carmena M, Earnshaw WC . (2003). The cellular geography of aurora kinases. Nat Rev Mol Cell Biol 4: 842–854.
Castro A, Arlot-Bonnemains Y, Vigneron S, Labbe JC, Prigent C, Lorca T . (2002). APC/Fizzy-related targets Aurora-A kinase for proteolysis. EMBO Rep 3: 457–462.
Crane R, Gadea B, Littlepage L, Wu H, Ruderman JV . (2004). Aurora A, meiosis and mitosis. Biol Cell 96: 215–229.
Dallol A, Agathanggelou A, Fenton SL, Ahmed-Choudhury J, Hesson L, Vos MD et al. (2004). RASSF1A interacts with microtubule-associated proteins and modulates microtubule dynamics. Cancer Res 64: 4112–4116.
Dammann R, Li C, Yoon JH, Chin PL, Bates S, Pfeifer GP . (2000). Epigenetic inactivation of a RAS association domain family protein from the lung tumour suppressor locus 3p21.3. Nat Genet 25: 315–319.
Dammann R, Schagdarsurengin U, Seidel C, Strunnikova M, Rastetter M, Baier K et al. (2005). The tumor suppressor RASSF1A in human carcinogenesis: an update. Histol Histopathol 20: 645–663.
Doxsey S . (2001a). Re-evaluating centrosome function. Nat Rev Mol Cell Biol 2: 688–698.
Doxsey S, Zimmerman W, Mikule K . (2005). Centrosome control of the cell cycle. Trends Cell Biol 15: 303–311.
Doxsey SJ . (2001b). Centrosomes as command centres for cellular control. Nat Cell Biol 3: E105–E108.
Ewart-Toland A, Briassouli P, de Koning JP, Mao JH, Yuan J, Chan F et al. (2003). Identification of Stk6/STK15 as a candidate low-penetrance tumor-susceptibility gene in mouse and human. Nat Genet 34: 403–412.
Ewart-Toland A, Dai Q, Gao YT, Nagase H, Dunlop MG, Farrington SM et al. (2005). Aurora-A/STK15 T+91A is a general low penetrance cancer susceptibility gene: a meta-analysis of multiple cancer types. Carcinogenesis 26: 1368–1373.
Gaglio T, Saredi A, Bingham JB, Hasbani MJ, Gill SR, Schroer TA et al. (1996). Opposing motor activities are required for the organization of the mammalian mitotic spindle pole. J Cell Biol 135: 399–414.
Giet R, Glover DM . (2001). Drosophila aurora B kinase is required for histone H3 phosphorylation and condensin recruitment during chromosome condensation and to organize the central spindle during cytokinesis. J Cell Biol 152: 669–682.
Giet R, Petretti C, Prigent C . (2005). Aurora kinases, aneuploidy and cancer, a coincidence or a real link? Trends Cell Biol 15: 241–250.
Glover DM, Leibowitz MH, McLean DA, Parry H . (1995). Mutations in aurora prevent centrosome separation leading to the formation of monopolar spindles. Cell 81: 95–105.
Guo C, Tommasi S, Liu L, Yee JK, Dammann R, Pfeifer GP . (2007). RASSF1A is part of a complex similar to the Drosophila Hippo/Salvador/Lats tumor-suppressor Network. Curr Biol 17: 700–705.
Guse A, Mishima M, Glotzer M . (2005). Phosphorylation of ZEN-4/MKLP1 by aurora B regulates completion of cytokinesis. Curr Biol 15: 778–786.
Hachet V, Canard C, Gonczy P . (2007). Centrosomes promote timely mitotic entry in C. elegans embryos. Dev Cell 12: 531–541.
Hinchcliffe EH, Miller FJ, Cham M, Khodjakov A, Sluder G . (2001). Requirement of a centrosomal activity for cell cycle progression through G1 into S phase. Science 291: 1547–1550.
Hirota T, Kunitoku N, Sasayama T, Marumoto T, Zhang D, Nitta M et al. (2003). Aurora-A and an interacting activator, the LIM protein Ajuba, are required for mitotic commitment in human cells. Cell 114: 585–598.
Honda K, Mihara H, Kato Y, Yamaguchi A, Tanaka H, Yasuda H et al. (2000). Degradation of human Aurora2 protein kinase by the anaphase-promoting complex-ubiquitin-proteasome pathway. Oncogene 19: 2812–2819.
Hutterer A, Berdnik D, Wirtz-Peitz F, Zigman M, Schleiffer A, Knoblich JA . (2006). Mitotic activation of the kinase Aurora-A requires its binding partner Bora. Dev Cell 11: 147–157.
Keen N, Taylor S . (2004). Aurora-kinase inhibitors as anticancer agents. Nat Rev Cancer 4: 927–936.
Khokhlatchev A, Rabizadeh S, Xavier R, Nedwidek M, Chen T, Zhang XF et al. (2002). Identification of a novel Ras-regulated proapoptotic pathway. Curr Biol 12: 253–265.
Kufer TA, Sillje HH, Korner R, Gruss OJ, Meraldi P, Nigg EA . (2002). Human TPX2 is required for targeting Aurora-A kinase to the spindle. J Cell Biol 158: 617–623.
Lerman MI, Minna JD . (2000). The 630-kb lung cancer homozygous deletion region on human chromosome 3p21.3: identification and evaluation of the resident candidate tumor suppressor genes. The International Lung Cancer Chromosome 3p21.3 Tumor Suppressor Gene Consortium. Cancer Res 60: 6116–6133.
Littlepage LE, Wu H, Andresson T, Deanehan JK, Amundadottir LT, Ruderman JV . (2002). Identification of phosphorylated residues that affect the activity of the mitotic kinase Aurora-A. Proc Natl Acad Sci USA 99: 15440–15445.
Liu L, Baier K, Dammann R, Pfeifer GP . (2007). The tumor suppressor RASSF1A does not interact with Cdc20, an activator of the anaphase-promoting complex. Cell Cycle 6: 1663–1665.
Liu L, Tommasi S, Lee DH, Dammann R, Pfeifer GP . (2003). Control of microtubule stability by the RASSF1A tumor suppressor. Oncogene 22: 8125–8136.
Liu L, Vo A, Liu G, McKeehan WL . (2005a). Distinct structural domains within C19ORF5 support association with stabilized microtubules and mitochondrial aggregation and genome destruction. Cancer Res 65: 4191–4201.
Liu L, Vo A, Liu G, McKeehan WL . (2005b). Putative tumor suppressor RASSF1 interactive protein and cell death inducer C19ORF5 is a DNA binding protein. Biochem Biophys Res Commun 332: 670–676.
Liu L, Vo A, McKeehan WL . (2005c). Specificity of the methylation-suppressed A isoform of candidate tumor suppressor RASSF1 for microtubule hyperstabilization is determined by cell death inducer C19ORF5. Cancer Res 65: 1830–1838.
Marumoto T, Hirota T, Morisaki T, Kunitoku N, Zhang D, Ichikawa Y et al. (2002). Roles of aurora-A kinase in mitotic entry and G2 checkpoint in mammalian cells. Genes Cells 7: 1173–1182.
Marumoto T, Honda S, Hara T, Nitta M, Hirota T, Kohmura E et al. (2003). Aurora-A kinase maintains the fidelity of early and late mitotic events in HeLa cells. J Biol Chem 278: 51786–51795.
Marumoto T, Zhang D, Saya H . (2005). Aurora-A—a guardian of poles. Nat Rev Cancer 5: 42–50.
Meraldi P, Honda R, Nigg EA . (2002). Aurora-A overexpression reveals tetraploidization as a major route to centrosome amplification in p53−/− cells. EMBO J 21: 483–492.
Meraldi P, Honda R, Nigg EA . (2004). Aurora kinases link chromosome segregation and cell division to cancer susceptibility. Curr Opin Genet Dev 14: 29–36.
Mortlock A, Keen NJ, Jung FH, Heron NM, Foote KM, Wilkinson R et al. (2005). Progress in the development of selective inhibitors of Aurora kinases. Curr Top Med Chem 5: 199–213.
Naruganahalli KS, Lakshmanan M, Dastidar SG, Ray A . (2006). Therapeutic potential of Aurora kinase inhibitors in cancer. Curr Opin Investig Drugs 7: 1044–1051.
Ortiz-Vega S, Khokhlatchev A, Nedwidek M, Zhang XF, Dammann R, Pfeifer GP et al. (2002). The putative tumor suppressor RASSF1A homodimerizes and heterodimerizes with the Ras-GTP binding protein Nore1. Oncogene 21: 1381–1390.
Piel M, Nordberg J, Euteneuer U, Bornens M . (2001). Centrosome-dependent exit of cytokinesis in animal cells. Science 291: 1550–1553.
Portier N, Audhya A, Maddox PS, Green RA, Dammermann A, Desai A et al. (2007). A microtubule-independent role for centrosomes and aurora a in nuclear envelope breakdown. Dev Cell 12: 515–529.
Pugacheva EN, Golemis EA . (2005). The focal adhesion scaffolding protein HEF1 regulates activation of the Aurora-A and Nek2 kinases at the centrosome. Nat Cell Biol 7: 937–946.
Pugacheva EN, Jablonski SA, Hartman TR, Henske EP, Golemis EA . (2007). HEF1-dependent Aurora A activation induces disassembly of the primary cilium. Cell 129: 1351–1363.
Rieder CL, Faruki S, Khodjakov A . (2001). The centrosome in vertebrates: more than a microtubule-organizing center. Trends Cell Biol 11: 413–419.
Rong R, Jiang LY, Sheikh MS, Huang Y . (2007). Mitotic kinase Aurora-A phosphorylates RASSF1A and modulates RASSF1A-mediated microtubule interaction and M-phase cell cycle regulation. Oncogene 26: 7700–7708.
Rong R, Jin W, Zhang J, Sheikh MS, Huang Y . (2004). Tumor suppressor RASSF1A is a microtubule-binding protein that stabilizes microtubules and induces G2/M arrest. Oncogene 23: 8216–8230.
Satinover DL, Leach CA, Stukenberg PT, Brautigan DL . (2004). Activation of Aurora-A kinase by protein phosphatase inhibitor-2, a bifunctional signaling protein. Proc Natl Acad Sci USA 101: 8625–8630.
Shivakumar L, Minna J, Sakamaki T, Pestell R, White MA . (2002). The RASSF1A tumor suppressor blocks cell cycle progression and inhibits cyclin D1 accumulation. Mol Cell Biol 22: 4309–4318.
Song MS, Song SJ, Ayad NG, Chang JS, Lee JH, Hong HK et al. (2004). The tumour suppressor RASSF1A regulates mitosis by inhibiting the APC-Cdc20 complex. Nat Cell Biol 6: 129–137.
Taguchi S, Honda K, Sugiura K, Yamaguchi A, Furukawa K, Urano T . (2002). Degradation of human Aurora-A protein kinase is mediated by hCdh1. FEBS Lett 519: 59–65.
Toji S, Yabuta N, Hosomi T, Nishihara S, Kobayashi T, Suzuki S et al. (2004). The centrosomal protein Lats2 is a phosphorylation target of Aurora-A kinase. Genes Cells 9: 383–397.
Tommasi S, Dammann R, Zhang Z, Wang Y, Liu L, Tsark WM et al. (2005). Tumor susceptibility of Rassf1a knockout mice. Cancer Res 65: 92–98.
Trieselmann N, Armstrong S, Rauw J, Wilde A . (2003). Ran modulates spindle assembly by regulating a subset of TPX2 and Kid activities including Aurora A activation. J Cell Sci 116: 4791–4798.
Tsai MY, Wiese C, Cao K, Martin O, Donovan P, Ruderman J et al. (2003). A Ran signalling pathway mediated by the mitotic kinase Aurora A in spindle assembly. Nat Cell Biol 5: 242–248.
Uzbekov R, Kireyev I, Prigent C . (2002). Centrosome separation: respective role of microtubules and actin filaments. Biol Cell 94: 275–288.
van der Weyden L, Tachibana KK, Gonzalez MA, Adams DJ, Ng BL, Petty R et al. (2005). The RASSF1A isoform of RASSF1 promotes microtubule stability and suppresses tumorigenesis. Mol Cell Biol 25: 8356–8367.
Vos MD, Martinez A, Elam C, Dallol A, Taylor BJ, Latif F et al. (2004). A role for the RASSF1A tumor suppressor in the regulation of tubulin polymerization and genomic stability. Cancer Res 64: 4244–4250.
Walter AO, Seghezzi W, Korver W, Sheung J, Lees E . (2000). The mitotic serine/threonine kinase Aurora2/AIK is regulated by phosphorylation and degradation. Oncogene 19: 4906–4916.
Wang X, Zhou YX, Qiao W, Tominaga Y, Ouchi M, Ouchi T et al. (2006). Overexpression of aurora kinase A in mouse mammary epithelium induces genetic instability preceding mammary tumor formation. Oncogene 25: 7148–7158.
Waters JC, Cole RW, Rieder CL . (1993). The force-producing mechanism for centrosome separation during spindle formation in vertebrates is intrinsic to each aster. J Cell Biol 122: 361–372.
Weaver BA, Cleveland DW . (2006). Does aneuploidy cause cancer? Curr Opin Cell Biol 18: 658–667.
Zhang D, Hirota T, Marumoto T, Shimizu M, Kunitoku N, Sasayama T et al. (2004). Cre-loxP-controlled periodic Aurora-A overexpression induces mitotic abnormalities and hyperplasia in mammary glands of mouse models. Oncogene 23: 8720–8730.
Zhao ZS, Lim JP, Ng YW, Lim L, Manser E . (2005). The GIT-associated kinase PAK targets to the centrosome and regulates Aurora-A. Mol Cell 20: 237–249.
Zhou H, Kuang J, Zhong L, Kuo WL, Gray JW, Sahin A et al. (1998). Tumour amplified kinase STK15/BTAK induces centrosome amplification, aneuploidy and transformation. Nat Genet 20: 189–193.
Acknowledgements
We thank E Nigg and H Nojima for the Aurora-A and LATS2 plasmid constructs.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, L., Guo, C., Dammann, R. et al. RASSF1A interacts with and activates the mitotic kinase Aurora-A. Oncogene 27, 6175–6186 (2008). https://doi.org/10.1038/onc.2008.220
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.220
Keywords
This article is cited by
-
Targeting AURKA in Cancer: molecular mechanisms and opportunities for Cancer therapy
Molecular Cancer (2021)
-
NDR2 kinase contributes to cell invasion and cytokinesis defects induced by the inactivation of RASSF1A tumor-suppressor gene in lung cancer cells
Journal of Experimental & Clinical Cancer Research (2019)
-
RASSF1A, puppeteer of cellular homeostasis, fights tumorigenesis, and metastasis—an updated review
Cell Death & Disease (2019)
-
New type of interaction between the SARAH domain of the tumour suppressor RASSF1A and its mitotic kinase Aurora A
Scientific Reports (2019)
-
RASSF1 tumor suppressor gene in pancreatic ductal adenocarcinoma: correlation of expression, chromosomal status and epigenetic changes
BMC Cancer (2016)