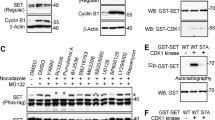
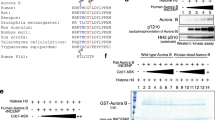
Abstract
P21-activated kinases (Paks), a family of serine/threonine kinases, are effectors of the Rho GTPases Cdc42 and Rac1. Mammalian Pak1 and Pak homologs in simple eukaryotes are implicated in controlling G2/M transition and/or mitosis. Another serine/threonine kinase, polo-like kinase 1 (Plk1), is an important regulator of mitotic events, such as centrosome maturation, mitotic entry, spindle formation, sister chromatid cohesion and cytokinesis. Plk1 phosphorylation is thought to be one of the critical regulatory events leading to these Plk1-mediated functions. We show here that Pak1 is required for cell proliferation, mitotic progression and Plk1 activity in HeLa cells. Gain or loss of Pak function directly impacted phosphorylation and activity of Plk1. Phosphorylation of Plk1 on Ser 49 is important for metaphase-associated events. Inhibition of Pak activity leads to delay in G2/M progression and abnormal spindle formation, mirroring some attributes of Plk1 deregulation. Our results reveal a role for Pak in regulating Plk1 activity and mitotic progression, and connect Pak to the complex protein interaction network enabling cell division.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Balasenthil S, Sahin AA, Barnes CJ, Wang RA, Pestell RG, Vadlamudi RK et al. (2004). p21-activated kinase-1 signaling mediates cyclin D1 expression in mammary epithelial and cancer cells. J Biol Chem 279: 1422–1428.
Banerjee M, Worth D, Prowse DM, Nikolic M . (2002). Pak1 phosphorylation on t212 affects microtubules in cells undergoing mitosis. Curr Biol 12: 1233–1239.
Barr FA, Sillje HH, Nigg EA . (2004). Polo-like kinases and the orchestration of cell division. Nat Rev Mol Cell Biol 5: 429–440.
Bode AM, Dong Z . (2005). Inducible covalent posttranslational modification of histone H3. Sci STKE 2005: re4.
Bokoch GM . (2003). Biology of the p21-activated kinases. Annu Rev Biochem 72: 743–781.
De Luca M, Lavia P, Guarguaglini G . (2006). A functional interplay between Aurora-A, Plk1 and TPX2 at spindle poles: Plk1 controls centrosomal localization of Aurora-A and TPX2 spindle association. Cell Cycle 5: 296–303.
DerMardirossian C, Schnelzer A, Bokoch GM . (2004). Phosphorylation of RhoGDI by Pak1 mediates dissociation of Rac GTPase. Mol Cell 15: 117–127.
Ellinger-Ziegelbauer H, Karasuyama H, Yamada E, Tsujikawa K, Todokoro K, Nishida E . (2000). Ste20-like kinase (SLK), a regulatory kinase for polo-like kinase (Plk) during the G2/M transition in somatic cells. Genes Cells 5: 491–498.
Glover DM . (2005). Polo kinase and progression through M phase in Drosophila: a perspective from the spindle poles. Oncogene 24: 230–237.
Hofmann C, Shepelev M, Chernoff J . (2004). The genetics of Pak. J Cell Sci 117: 4343–4354.
Holm C, Rayala S, Jirstrom K, Stal O, Kumar R, Landberg G . (2006). Association between Pak1 expression and subcellular localization and tamoxifen resistance in breast cancer patients. J Natl Cancer Inst 98: 671–680.
Jang Y-J, Ma S, Terada Y, Erikson RL . (2002). Phosphorylation of threonine 210 and the role of serine 137 in the regulation of mammalian polo-like kinase. J Biol Chem 277: 44115–44120.
Knaus UG, Morris S, Dong HJ, Chernoff J, Bokoch GM . (1995). Regulation of human leukocyte p21-activated kinases through G protein-coupled receptors. Science 269: 221–223.
Kumar R, Gururaj AE, Barnes CJ . (2006). p21-activated kinases in cancer. Nat Rev Cancer 6: 459–471.
Kumar R, Vadlamudi RK . (2002). Emerging functions of p21-activated kinases in human cancer cells. J Cell Physiol 193: 133–144.
Lei M, Lu W, Meng W, Parrini MC, Eck MJ, Mayer BJ et al. (2000). Structure of PAK1 in an autoinhibited conformation reveals a multistage activation switch. Cell 102: 387–397.
Lenart P, Petronczki M, Steegmaier M, Di Fiore B, Lipp JJ, Hoffmann M et al. (2007). The small-molecule inhibitor BI 2536 reveals novel insights into mitotic roles of polo-like kinase 1. Curr Biol 17: 304–315.
Li F, Adam L, Vadlamudi RK, Zhou H, Sen S, Chernoff J et al. (2002). p21-activated kinase 1 interacts with and phosphorylates histone H3 in breast cancer cells. EMBO Rep 3: 767–773.
Lowery DM, Lim D, Yaffe MB . (2005). Structure and function of polo-like kinases. Oncogene 24: 248–259.
Manser E, Leung T, Salihuddin H, Zhao ZS, Lim L . (1994). A brain serine/threonine protein kinase activated by Cdc42 and Rac1. Nature 367: 40–46.
Mira J-P, Benard V, Groffen J, Sanders LC, Knaus UG . (2000). Endogenous, hyperactive Rac3 controls proliferation of breast cancer cells by a p21-activated kinase-dependent pathway. Proc Natl Acad Sci USA 97: 185–189.
Nheu T, He H, Hirokawa Y, Walker F, Wood J, Maruta H . (2004). PAK is essential for RAS-induced upregulation of cyclin D1 during the G1 to S transition. Cell Cycle 3: 71–74.
Nowak SJ, Corces VG . (2004). Phosphorylation of histone H3: a balancing act between chromosome condensation and transcriptional activation. Trends Genet 20: 214–220.
Seong YS, Kamijo K, Lee JS, Fernandez E, Kuriyama R, Miki T et al. (2002). A spindle checkpoint arrest and a cytokinesis failure by the dominant-negative polo-box domain of Plk1 in U-2 OS cells. J Biol Chem 277: 32282–32293.
Shalom-Barak T, Knaus UG . (2002). A p21-activated kinase-controlled metabolic switch up-regulates phagocyte NADPH oxidase. J Biol Chem 277: 40659–40665.
Sumara I, Gimenez-Abian JF, Gerlich D, Hirota T, Kraft C, De la Torre C et al. (2004). Roles of polo-like kinase 1 in the assembly of functional mitotic spindles. Curr Biol 14: 1712–1722.
Thiel DA, Reeder MK, Pfaff A, Coleman TR, Sells MA, Chernoff J . (2002). Cell cycle-regulated phosphorylation of p21-activated kinase 1. Curr Biol 12: 1227–1232.
Thullberg M, Gad A, Beeser A, Chernoff J, Stromblad S . (2007). The kinase-inhibitory domain of p21-activated kinase 1 (PAK1) inhibits cell cycle progression independent of PAK1 kinase activity. Oncogene 26: 1820–1828.
Vadlamudi RK, Barnes CJ, Rayala S, Li F, Balasenthil S, Marcus S et al. (2005). p21-activated kinase 1 regulates microtubule dynamics by phosphorylating tubulin cofactor B. Mol Cell Biol 25: 3726–3736.
van de Weerdt B, Medema R . (2006). Polo-like kinases: a team in control of the division. Cell Cycle 5: 853–864.
van de Weerdt BC, van Vugt MA, Lindon C, Kauw JJ, Rozendaal MJ, Klompmaker R et al. (2005). Uncoupling anaphase-promoting complex/cyclosome activity from spindle assembly checkpoint control by deregulating polo-like kinase 1. Mol Cell Biol 25: 2031–2044.
van Vugt MA, Medema RH . (2005). Getting in and out of mitosis with polo-like kinase-1. Oncogene 24: 2844–2859.
van Vugt MA, van de Weerdt BC, Vader G, Janssen H, Calafat J, Klompmaker R et al. (2004). Polo-like kinase-1 is required for bipolar spindle formation but is dispensable for anaphase promoting complex/Cdc20 activation and initiation of cytokinesis. J Biol Chem 279: 36841–36854.
Xie S, Xie B, Lee MY, Dai W . (2005). Regulation of cell cycle checkpoints by polo-like kinases. Oncogene 24: 277–286.
Xu B, Kim ST, Lim DS, Kastan MB . (2002). Two molecularly distinct G(2)/M checkpoints are induced by ionizing irradiation. Mol Cell Biol 22: 1049–1059.
Zhao ZS, Lim JP, Ng YW, Lim L, Manser E . (2005). The GIT-associated kinase PAK targets to the centrosome and regulates Aurora-A. Mol Cell 20: 237–249.
Acknowledgements
We thank E Nishida for Plk wild-type constructs, the laboratories of G Bokoch and S Reed for sharing antibodies, J Birkenfeld for advice and K Schreiber for illustrations and administrative assistance. This work was supported by NIH (AI35947, AI24838 to UGK), by funds from the California Breast Cancer Research Program of the University of California 8FB-0026 (BM), NIH training grant T32 AI07244-22 (MY) and a German Academic Exchange (DAAD) fellowship (AS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Rights and permissions
About this article
Cite this article
Maroto, B., Ye, M., von Lohneysen, K. et al. P21-activated kinase is required for mitotic progression and regulates Plk1. Oncogene 27, 4900–4908 (2008). https://doi.org/10.1038/onc.2008.131
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.131
Keywords
This article is cited by
-
Unique vulnerability of RAC1-mutant melanoma to combined inhibition of CDK9 and immune checkpoints
Oncogene (2024)
-
PAK2 is essential for chromosome alignment in metaphase I oocytes
Cell Death & Disease (2023)
-
Expression of Marker PAK1 in Sinonasal Polyposis
Indian Journal of Otolaryngology and Head & Neck Surgery (2022)
-
Inhibition of polo-like kinase 1 suppresses microtubule dynamics in MCF-7 cells
Molecular and Cellular Biochemistry (2020)
-
The p21-activated kinase (PAK1) is involved in diet-induced beta cell mass expansion and survival in mice and human islets
Diabetologia (2016)