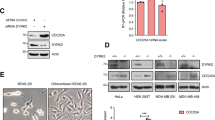
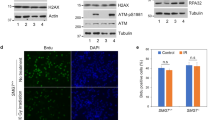
Abstract
Although heat-shock factor (HSF) 1 is a known transcriptional factor of heat-shock proteins, other pathways like production of aneuploidy and increased protein stability of cyclin B1 have been proposed. In the present study, the regulatory domain of HSF1 (amino-acid sequence 212–380) was found to interact directly with the amino-acid sequence 106–171 of Cdc20. The association between HSF1 and Cdc20 inhibited the interaction between Cdc27 and Cdc20, the phosphorylation of Cdc27 and the ubiquitination activity of anaphase-promoting complex (APC). The overexpression of HSF1 inhibited mitotic exit and the degradations of cyclin B1 and securin, which resulted in production of aneuploidy and multinucleated cells, but regulatory domain-deficient HSF1 did not. Moreover, HSF1-overexpressing cells showed elevated levels of micronuclei and genomic alteration. The depletion of HSF1 from cells highly expressing HSF1 reduced nocodazole-mediated aneuploidy in cells. These findings suggest a novel function of HSF1 frequently overexpressed in cancer cells, to inhibit APC/C activity by interacting with Cdc20, and to result in aneuploidy development and genomic instability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cahill DP, Lengauer C, Yu J, Riggins GJ, Willson JKV, Markowitz SD et al. (1998). Mutations of mitotic checkpoint genes in human cancers. Nature 392: 300–303.
Cen H, Zheng S, Fang YM, Tang XP, Dong Q . (2004). Induction of HSF1 expression is associated with sporadic colorectal cancer. World J Gastroenterol 10: 3122–3126.
Cross FR . (2003). Two redundant oscillatory mechanisms in the yeast cell cycle. Dev Cell 4: 741–752.
Hahn GM, van Kersen I . (1998). Isolation and initial characterization of thermoresistant RIF tumor cell strains. Cancer Res 48: 1803–1807.
Hida K, Klagsbrun M . (2005). A new perspective on tumor endothelial cells: unexpected chromosome and centrosome abnormalities. Cancer Res 65: 2507–2510.
Hoang AT, Huang J, Rudra-Ganguly N, Zheng J, Powell WC, Rabindran SK et al. (2000). A novel association between the human heat-shock transcription factor 1 (HSF1) and prostate adenocarcinoma. Am J Pathol 156: 857–864.
Lengauer C, Kinzler KW, Vogelstein B . (1997). Genetic instability in colorectal cancers. Nature 386: 623–627.
Li GC, Werb Z . (1982). Correlation between synthesis of heat-shock proteins and development of thermotolerance in Chinese hamster fibroblasts. Proc Nat Acad Sci 79: 3218–3222.
Min JN, Huang L, Zimonjiic DB, Moskophidis D, Mivechi NF . (2007). Selective suppression of lymphomas by functional loss of Hsf1 in p53-deficeint mouse model for spontaneous tumors. Oncogene 26: 5086–5097.
Pirkkala L, Nykanen P, Sistonen L . (2001). Roles of the heat-shock transcription factors in regulation of the heat-shock response and beyond. FASEB J 15: 1118–1131.
Ricaniadis N, Kataki A, Agnantis N, Androulakis G, Karakousis CP . (2001). Long-term prognostic significance of HSP70, c-myc and HLA-DR expression in patients with malignant melanoma. Eur J Surg Oncol 27: 88–93.
Seo HR, Chung DY, Lee YJ, Lee DH, Kim JI, Bae S et al. (2006). Heat shock protein 25 or inducible heat shock protein 70 activates heat shock factor 1: dephosphorylation on serine 307 through inhibition of ERK1/2 phosphorylation. J Biol Chem 281: 17220–17227.
Shin CR, Suh EJ, Lee KH, Kim TY, Kim SH . (2003). p55CDC/hCDC20 mutant induces mitotic catastrophe by inhibiting the MAD2-dependent spindle checkpoint activity in tumor cells. Cancer Lett 201: 203–210.
Soh JW, Lee EH, Prywes R, Weinstein IB . (1999). Novel roles of specific isoforms of protein kinase C in activation of the c-fos serum response element. Mol Cell Biol 19: 1313–1324.
Song MS, Song SJ, Ayad NG, Chang JS, Lee JH, Hong HK et al. (2004). The tumor suppressor RASSF1A regulates mitosis by inhibiting the APC-Cdc20 complex. Nat Cell Biol 6: 129–137.
Su TT, Sprenger F, DiGregorio PJ, Campbell SD, O'Farrell PH . (1998). Exit from mitosis in Drosophila syncytial embryos requires proteolysis and cyclin degradation, and is associated with localized dephosphorylation. Genes Dev 12: 1495–1503.
Wang Y, Theriault JR, He H, Gong J, Calderwood SK . (2004). Expression of a dominant negative heat-shock factor-1 construct inhibits aneuploidy in prostate carcinoma cells. J Biol Chem 279: 32651–32659.
Acknowledgements
This work was supported by the Korean Science and Engineering Foundation (KOSEF) and by the Korean Ministry of Science and Technology (MOST), through the National Nuclear Technology Program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Lee, Y., Lee, H., Lee, J. et al. A novel function for HSF1-induced mitotic exit failure and genomic instability through direct interaction between HSF1 and Cdc20. Oncogene 27, 2999–3009 (2008). https://doi.org/10.1038/sj.onc.1210966
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210966
Keywords
This article is cited by
-
Heat shock factor 1 suppression induces spindle abnormalities and sensitizes cells to antimitotic drugs
Cell Division (2021)
-
The importance of CDC27 in cancer: molecular pathology and clinical aspects
Cancer Cell International (2021)
-
Interplay between HSF1 and p53 signaling pathways in cancer initiation and progression: non-oncogene and oncogene addiction
Cellular Oncology (2019)
-
Polymorphisms in human heat shock factor-1 and analysis of potential biological consequences
Cell Stress and Chaperones (2015)
-
Advances in NKG2D ligand recognition and responses by NK cells
Immunology & Cell Biology (2014)