Abstract
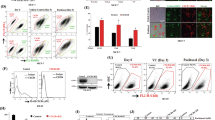
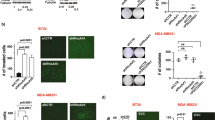
Chemokine receptor CXCR4 and its ligand CXCL12 are suggested to be involved in migration, invasion and metastasis of breast cancer cells. Mutation of the tumor suppressor gene p53 in breast cancer is associated with metastasis and aggressive clinical phenotype. In this report, we demonstrate that wild type but not the dominant-negative mutant (V143A) or cancer-specific mutants (R175H or R280K) of p53 repress CXCR4 expression. Recently described cancer-specific p53 isoform, Δ133p53, also failed to repress CXCR4 promoter activity. Short-interfering RNA-mediated depletion of p53 increased endogenous CXCR4 expression in MCF-7 breast cancer cells that contain wild-type p53. Basal CXCR4 promoter activity in HCT116 colon carcinoma cells deleted of p53 [HCT116(p53KO)] was 10-fold higher compared to that in parental HCT116 cells with functional wild-type p53. Deletion analysis of CXCR4 promoter identified a seven-base pair p53-repressor element homologous to cyclic AMP/AP-1 response (CRE/AP-1) element. Electrophoretic mobility shift and chromatin immunoprecipitation assays revealed binding of ATF-1 and cJun to the CRE/AP-1 element. The p53 rescue drug PRIMA-1 reduced CXCR4 mRNA and cell surface expression in MDA-MB-231 cells, which express R280K mutant p53. CP-31398, another p53 rescue drug, similarly reduced cell surface levels of CXCR4. PRIMA-1-mediated decrease in CXCR4 expression correlated with reduced invasion of MDA-MB-231 cells through matrigel. These results suggest a mechanism for elevated CXCR4 expression and metastasis of breast cancers with p53 mutations or isoform expression. We propose that p53 rescue drugs either alone or in combination with chemotherapeutic drugs may be effective in reducing CXCR4-mediated metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bachelder RE, Wendt MA, Mercurio AM . (2002). Vascular endothelial growth factor promotes breast carcinoma invasion in an autocrine manner by regulating the chemokine receptor CXCR4. Cancer Res 62: 7203–7206.
Balkwill F . (2004). Cancer and the chemokine network. Nat Rev Cancer 4: 540–550.
Bargonetti J, Chicas A, White D, Prives C . (1997). p53 represses Sp1 DNA binding and HIV-LTR directed transcription. Cell Mol Biol 43: 935–949.
Berns EM, Foekens JA, Vossen R, Look MP, Devilee P, Henzen-Logmans SC et al. (2000). Complete sequencing of TP53 predicts poor response to systemic therapy of advanced breast cancer. Cancer Res 60: 2155–2162.
Bourdon JC, Fernandes K, Murray-Zmijewski F, Liu G, Diot A, Xirodimas DP et al. (2005). p53 isoforms can regulate p53 transcriptional activity. Genes Dev 19: 2122–2137.
Bunz F, Fauth C, Speicher MR, Dutriaux A, Sedivy JM, Kinzler KW et al. (2002). Targeted inactivation of p53 in human cells does not result in aneuploidy. Cancer Res 62: 1129–1133.
Bykov VJ, Issaeva N, Selivanova G, Wiman KG . (2002a). Mutant p53-dependent growth suppression distinguishes PRIMA-1 from known anticancer drugs: a statistical analysis of information in the National Cancer Institute database. Carcinogenesis 23: 2011–2018.
Bykov VJ, Issaeva N, Shilov A, Hultcrantz M, Pugacheva E, Chumakov P et al. (2002b). Restoration of the tumor suppressor function to mutant p53 by a low-molecular-weight compound. Nat Med 8: 282–288.
Cabioglu N, Sahin A, Doucet M, Yavuz E, Igci A, E OY et al. (2005). Chemokine receptor CXCR4 expression in breast cancer as a potential predictive marker of isolated tumor cells in bone marrow. Clin Exp Metastasis 22: 39–46.
Caruz A, Samsom M, Alonso JM, Alcami J, Baleux F, Virelizier JL et al. (1998). Genomic organization and promoter characterization of human CXCR4 gene. FEBS Lett 426: 271–278.
Casey G, Lo-Hsueh M, Lopez ME, Vogelstein B, Stanbridge EJ . (1991). Growth suppression of human breast cancer cells by the introduction of a wild-type p53 gene. Oncogene 6: 1791–1797.
Dong Y, Asch HL, Ying A, Asch BB . (2002). Molecular mechanism of transcriptional repression of gelsolin in human breast cancer cells. Exp Cell Res 276: 328–336.
Foster BA, Coffey HA, Morin MJ, Rastinejad F . (1999). Pharmacological rescue of mutant p53 conformation and function. Science 286: 2507–2510.
Frasor J, Danes JM, Komm B, Chang KC, Lyttle CR, Katzenellenbogen BS . (2003). Profiling of estrogen up- and down-regulated gene expression in human breast cancer cells: insights into gene networks and pathways underlying estrogenic control of proliferation and cell phenotype. Endocrinology 144: 4562–4574.
Fridman JS, Lowe SW . (2003). Control of apoptosis by p53. Oncogene 22: 9030–9040.
Gupta GP, Minn AJ, Kang Y, Siegel PM, Serganova I, Cordon-Cardo C et al. (2005). Identifying site-specific metastasis genes and functions. Cold Spring Harb Symp Quant Biol 70: 149–158.
Helbig G, Christopherson II KW, Bhat-Nakshatri P, Kumar S, Kishimoto H, Miller KD et al. (2003). NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem 278: 21631–21638.
Kang Y, Siegel PM, Shu W, Drobnjak M, Kakonen SM, Cordon-Cardo C et al. (2003). A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3: 537–549.
Khan MZ, Shimizu S, Patel JP, Nelson A, Le MT, Mullen-Przeworski A et al. (2005). Regulation of neuronal P53 activity by CXCR 4. Mol Cell Neurosci 30: 58–66.
Kibler KV, Jeang KT . (2001). CREB/ATF-dependent repression of cyclin a by human T-cell leukemia virus type 1 Tax protein. J Virol 75: 2161–2173.
Kim E, Gunther W, Yoshizato K, Meissner H, Zapf S, Nusing RM et al. (2003). Tumor suppressor p53 inhibits transcriptional activation of invasion gene thromboxane synthase mediated by the proto-oncogenic factor ets-1. Oncogene 22: 7716–7727.
Kucia M, Reca R, Miekus K, Wanzeck J, Wojakowski W, Janowska-Wieczorek A et al. (2005). Trafficking of normal stem cells and metastasis of cancer stem cells involve similar mechanisms: pivotal role of the SDF-1-CXCR4 axis. Stem Cells 23: 879–894.
Lee BC, Lee TH, Zagozdzon R, Avraham S, Usheva A, Avraham HK . (2005). Carboxyl-terminal Src kinase homologous kinase negatively regulates the chemokine receptor CXCR4 through YY1 and impairs CXCR4/CXCL12 (SDF-1alpha) mediated breast cancer cell migration. Cancer Res 65: 2840–2845.
Li Y, Mao Y, Brandt-Rauf PW, Williams AC, Fine RL . (2005). Selective induction of apoptosis in mutant p53 premalignant and malignant cancer cells by PRIMA-1 through the c-Jun-NH2-kinase pathway. Mol Cancer Ther 4: 901–909.
Liang Z, Yoon Y, Votaw J, Goodman MM, Williams L, Shim H . (2005). Silencing of CXCR4 blocks breast cancer metastasis. Cancer Res 65: 967–971.
Matteucci E, Locati M, Desiderio MA . (2005). Hepatocyte growth factor enhances CXCR4 expression favoring breast cancer cell invasiveness. Exp Cell Res 310: 176–185.
Mayr B, Montminy M . (2001). Transcriptional regulation by the phosphorylation-dependent factor CREB. Nat Rev Mol Cell Biol 2: 599–609.
Mori T, Kim J, Yamano T, Takeuchi H, Huang S, Umetani N et al. (2005). Epigenetic up-regulation of C-C chemokine receptor 7 and C-X-C chemokine receptor 4 expression in melanoma cells. Cancer Res 65: 1800–1807.
Moriuchi M, Moriuchi H, Margolis DM, Fauci AS . (1999). USF/c-Myc enhances, while Yin-Yang 1 suppresses, the promoter activity of CXCR4, a coreceptor for HIV-1 entry. J Immunol 162: 5986–5992.
Muller A, Homey B, Soto H, Ge N, Catron D, Buchanan ME et al. (2001). Involvement of chemokine receptors in breast cancer metastasis. Nature 410: 50–56.
Salvucci O, Bouchard A, Baccarelli A, Deschenes J, Sauter G, Simon R et al. (2005). The role of CXCR4 receptor expression in breast cancer: a large tissue microarray study. Breast Cancer Res Treat 97: 275–283.
Scian MJ, Stagliano KE, Ellis MA, Hassan S, Bowman M, Miles MF et al. (2004). Modulation of gene expression by tumor-derived p53 mutants. Cancer Res 64: 7447–7454.
Shaulian E, Karin M . (2002). AP-1 as a regulator of cell life and death. Nat Cell Biol 4: E131–E136.
Sheridan C, Sadaria M, Bhat-Nakshatri P, Goulet Jr R, Edenberg HJ, McCarthy BP et al. (2006). Negative regulation of MHC class II gene expression by CXCR4. Exp Hematol 34: 1085–1092.
Smith MC, Luker KE, Garbow JR, Prior JL, Jackson E, Piwnica-Worms D et al. (2004). CXCR4 regulates growth of both primary and metastatic breast cancer. Cancer Res 64: 8604–8612.
Staller P, Sulitkova J, Lisztwan J, Moch H, Oakeley EJ, Krek W . (2003). Chemokine receptor CXCR4 downregulated by von Hippel-Lindau tumour suppressor pVHL. Nature 425: 307–311.
Sun Y, Zeng XR, Wenger L, Firestein GS, Cheung HS . (2004). P53 down-regulates matrix metalloproteinase-1 by targeting the communications between AP-1 and the basal transcription complex. J Cell Biochem 92: 258–269.
Vousden KH, Lu X . (2002). Live or let die: the cell's response to p53. Nat Rev Cancer 2: 594–604.
Acknowledgements
We thank Drs B Vogelstein, P Staller, M Kaplan, S Boswell and M Smith for various reagents. This work is supported by grants R01 CA89153, N01-CN-15133 (to HN), R01 HL67384 and R01 DK53674 (to HB) from the National Institutes of Health, American Institute for Cancer Research (03A069-REN) and Michael Guest Innovative Research Award from Walther Cancer Institute (to HN). HN is Marian J Morrison Investigator in Breast Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Mehta, S., Christopherson, K., Bhat-Nakshatri, P. et al. Negative regulation of chemokine receptor CXCR4 by tumor suppressor p53 in breast cancer cells: implications of p53 mutation or isoform expression on breast cancer cell invasion. Oncogene 26, 3329–3337 (2007). https://doi.org/10.1038/sj.onc.1210120
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210120
Keywords
This article is cited by
-
TCRP1 activated by mutant p53 promotes NSCLC proliferation via inhibiting FOXO3a
Oncogenesis (2022)
-
p53 deficiency promotes bone regeneration by functional regulation of mesenchymal stromal cells and osteoblasts
Journal of Bone and Mineral Metabolism (2022)
-
Distinct expression and function of breast cancer metastasis suppressor 1 in mutant P53 glioblastoma
Cellular Oncology (2022)
-
PRIMA-1MET-induced neuroblastoma cell death is modulated by p53 and mycn through glutathione level
Journal of Experimental & Clinical Cancer Research (2019)
-
FAK-ERK activation in cell/matrix adhesion induced by the loss of apolipoprotein E stimulates the malignant progression of ovarian cancer
Journal of Experimental & Clinical Cancer Research (2018)