Abstract
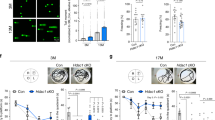
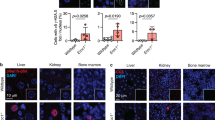
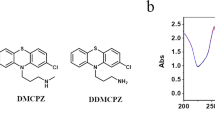
OGG1 (8-oxoguanine DNA glycosylase-1) is one of the main DNA glycosylases present in mammalian cells. The enzyme removes 7,8-dihydro-8-oxoguanine (8-oxoG) lesions, believed to be the most important oxidized lesions due to their relatively high incidence and their miscoding properties. This study shows that in prenatal mice brains the repair capacity for 8-oxoG is 5–10-fold higher than in adult mice brains. Western blot analysis and repair activity in extracts from Ogg1−/− mice revealed that OGG1 was responsible for the efficient 8-oxoG removal from prenatal mice. To investigate how OGG1 protects against oxidative stress-induced mutagenesis, pregnant Big Blue/wild-type and Big Blue/Ogg1−/− mice were exposed to nontoxic doses of gamma radiation. A 2.5-fold increase in the mutation frequency in Ogg1−/− mouse brains was obtained by exposure to 3.5 Gy at day 19 postfertilization. This was largely due to GC to TA transversions, believed to originate from 8-oxoG mispairing with A during replication. Furthermore, rapid cell divisions seemed to be required for fixation of mutations, as a similar dose of radiation did not increase the mutation frequency, or the frequency of GC to TA transversion, in the adult brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Aburatani H, Hippo Y, Ishida T, Takashima R, Matsuba C, Kodama T et al. (1997). Cancer Res 57: 2151–2156.
Achanta G, Huang P . (2004). Cancer Res 64: 6233–6239.
Ames BN . (1989). Mutat Res 214: 41–46.
Andrew SE, McKinnon M, Cheng BS, Francis A, Penney J, Reitmair AH et al. (1998). Proc Natl Acad Sci USA 95: 1126–1130.
Andrew SE, Reitmair AH, Fox J, Hsiao L, Francis A, McKinnon M et al. (1997). Oncogene 15: 123–129.
Arai T, Kelly VP, Komoro K, Minowa O, Noda T, Nishimura S . (2003). Cancer Res 63: 4287–4292.
Audebert M, Chevillard S, Levalois C, Gyapay G, Vieillefond A, Klijanienko J et al. (2000). Cancer Res 60: 4740–4744.
Bjelland S, Seeberg E . (2003). Mutat Res 531: 37–80.
Bjørås M, Luna L, Johnsen B, Hoff E, Haug T, Rognes T et al. (1997). EMBO J 16: 6314–6322.
Bohr VA . (2002). Free Radic Biol Med 32: 804–812.
Breen AP, Murphy JA . (1995). Free Radic Biol Med 18: 1033–1077.
Dianov GL, Souza-Pinto N, Nyaga SG, Thybo T, Stevnsner T, Bohr VA . (2001). Prog Nucleic Acid Res Mol Biol 68: 285–297.
Dou H, Mitra S, Hazra TK . (2003). J Biol Chem 278: 49679–49684.
Elahi A, Zheng Z, Park J, Eyring K, McCaffrey T, Lazarus P . (2002). Carcinogenesis 23: 1229–1234.
Franks LM, Wilson PD, Whelan RD . (1974). Gerontologia 20: 21–26.
Hollstein M, Sidransky D, Vogelstein B, Harris CC . (1991). Science 253: 49–53.
Hyun JW, Choi JY, Zeng HH, Lee YS, Kim HS, Yoon SH et al. (2000). Oncogene 19: 4476–4479.
Ishida T, Takashima R, Fukayama M, Hamada C, Hippo Y, Fujii T et al. (1999). Int J Cancer 80: 18–21.
Karahalil B, Hogue BA, de Souza-Pinto NC, Bohr VA . (2002). FASEB J 16: 1895–1902.
Kasai H, Nishimura S . (1984). Nucleic Acids Res 12: 2137–2145.
Kasai H . (1997). Mutat Res 387: 147–163.
Klungland A, Lindahl T . (1997). EMBO J 16: 3341–3348.
Klungland A, Rosewell I, Hollenbach S, Larsen E, Daly G, Epe B et al. (1999). Proc Natl Acad Sci USA 96: 13300–13305.
Kohler SW, Provost GS, Fieck A, Kretz PL, Bullock WO, Putman DL et al. (1991). Environ Mol Mutagen 18: 316–321.
Korr H . (1980). Adv Anat Embryol Cell Biol 61: 1–72.
Kubota Y, Nash RA, Klungland A, Schar P, Barnes DE, Lindahl T . (1996). EMBO J 15: 6662–6670.
Kucherlapati M, Yang K, Kuraguchi M, Zhao J, Lia M, Heyer J et al. (2002). Proc Natl Acad Sci USA 99: 9924–9929.
Larsen E, Gran C, Saether BE, Seeberg E, Klungland A . (2003). Mol Cell Biol 23: 5346–5353.
Le Marchand L, Donlon T, Lum-Jones A, Seifried A, Wilkens LR . (2002). Cancer Epidemiol Biomarkers Prev 11: 409–412.
Lin LH, Cao S, Yu L, Cui J, Hamilton WJ, Liu PK . (2000). J Neurochem 74: 1098–1105.
Lipton L, Halford SE, Johnson V, Novelli MR, Jones A, Cummings C et al. (2003). Cancer Res 63: 7595–7599.
Lovell MA, Gabbita SP, Markesbery WR . (1999). J Neurochem 72: 771–776.
Lovell MA, Xie C, Markesbery WR . (2000). Brain Res 855: 116–123.
Ludwig DL, MacInnes MA, Takiguchi Y, Purtymun PE, Henrie M, Flannery M et al. (1998). Mutat Res 409: 17–29.
Michaels ML, Cruz C, Grollman AP, Miller JH . (1992). Proc Natl Acad Sci USA 89: 7022–7025.
Michaels ML, Miller JH . (1992). J Bacteriol 174: 6321–6325.
Michaels ML, Pham L, Cruz C, Miller JH . (1991). Nucleic Acids Res 19: 3629–3632.
Minowa O, Arai T, Hirano M, Monden Y, Nakai S, Fukuda M et al. (2000). Proc Natl Acad Sci USA 97: 4156–4161.
Morland I, Rolseth V, Luna L, Rognes T, Bjørås M, Seeberg E . (2002). Nucleic Acids Res 30: 4926–4936.
Nishino H, Buettner VL, Sommer SS . (1996). Mutat Res 372: 97–105.
Prolla TA, Baker SM, Harris AC, Tsao JL, Yao X, Bronner CE et al. (1998). Nat Genet 18: 76–79.
Radicella JP, Dherin C, Desmaze C, Fox MS, Boiteux S . (1997). Proc Natl Acad Sci USA 94: 8010–8015.
Roldan-Arjona T, Wei YF, Carter KC, Klungland A, Anselmino C, Wang RP et al. (1997). Proc Natl Acad Sci USA 94: 8016–8020.
Rosenquist TA, Zaika E, Fernandes AS, Zharkov DO, Miller H, Grollman AP . (2003). DNA Repair 2: 581–591.
Sakumi K, Tominaga Y, Furuichi M, Xu P, Tsuzuki T, Sekiguchi M et al. (2003). Cancer Res 63: 902–905.
Shinmura K, Kohno T, Kasai H, Koda K, Sugimura H, Yokota J . (1998). J Cancer Res 89: 825–828.
Sieber OM, Lipton L, Crabtree M, Heinimann K, Fidalgo P, Phillips RK et al. (2003). N Engl J Med 348: 791–799.
Stuart GR, Oda Y, de Boer JG, Glickman BW . (2000). Genetics 154: 1291–1300.
Sugo N, Aratani Y, Nagashima Y, Kubota Y, Koyama H . (2000). EMBO J 19: 1397–1404.
Tebbs RS, Flannery ML, Meneses JJ, Hartmann A, Tucker JD, Thompson LH et al. (1999). Dev Biol 208: 513–529.
Verjat T, Dhenaut A, Radicella JP, Araneda S . (2000). Mutat Res 25: 127–138.
Wikman H, Risch A, Klimek F, Schmezer P, Spiegelhalder B, Dienemann H et al. (2000). Int J Cancer 88: 932–937.
Xie Y, Yang H, Cunanan C, Okamoto K, Shibata D, Pan J et al. (2004). Cancer Res 64: 3096–3102.
Acknowledgements
This work was supported by the Norwegian Cancer Society and the Research Council of Norway. We are grateful to our group members and to Veslemøy Rolseth and Pål Falnes for critiquing the manuscript. We are also grateful to Einar Rødland for undertaking a statistical analysis of the mutation frequencies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Larsen, E., Reite, K., Nesse, G. et al. Repair and mutagenesis at oxidized DNA lesions in the developing brain of wild-type and Ogg1−/− mice. Oncogene 25, 2425–2432 (2006). https://doi.org/10.1038/sj.onc.1209284
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209284
Keywords
This article is cited by
-
Restoration of Cognitive Performance in Mice Carrying a Deficient Allele of 8-Oxoguanine DNA Glycosylase by X-ray Irradiation
Neurotoxicity Research (2018)
-
No cancer predisposition or increased spontaneous mutation frequencies in NEIL DNA glycosylases-deficient mice
Scientific Reports (2017)
-
Evidence that OGG1 Glycosylase Protects Neurons against Oxidative DNA Damage and Cell Death under Ischemic Conditions
Journal of Cerebral Blood Flow & Metabolism (2011)
-
Laktat und Redoxstatus in malignen Tumoren
Der Anaesthesist (2007)