Abstract
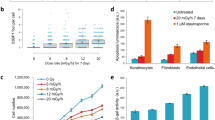
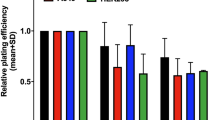
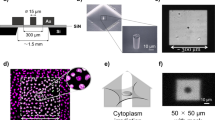
That irradiated cells affect their unirradiated ‘bystander’ neighbors is evidenced by reports of increased clonogenic mortality, genomic instability, and expression of DNA-repair genes in the bystander cell populations. The mechanisms underlying the bystander effect are obscure, but genomic instability suggests DNA double-strand breaks (DSBs) may be involved. Formation of DSBs induces the phosphorylation of the tumor suppressor protein, histone H2AX and this phosphorylated form, named γ-H2AX, forms foci at DSB sites. Here we report that irradiation of target cells induces γ-H2AX focus formation in bystander cell populations. The effect is manifested by increases in the fraction of cells in a population that contains multiple γ-H2AX foci. After 18 h coculture with cells irradiated with 20 α-particles, the fraction of bystander cells with multiple foci increased 3.7-fold. Similar changes occurred in bystander populations mixed and grown with cells irradiated with γ-rays, and in cultures containing media conditioned on γ-irradiated cells. DNA DSB repair proteins accumulated at γ-H2AX foci, indicating that they are sites of DNA DSB repair. Lindane, which blocks gap-junctions, prevented the bystander effect in mixing but not in media transfer protocols, while c-PTIO and aminoguanidine, which lower nitric oxide levels, prevented the bystander effect in both protocols. Thus, multiple mechanisms may be involved in transmitting bystander effects. These studies show that H2AX phosphorylation is an early step in the bystander effect and that the DNA DSBs underlying γ-H2AX focus formation may be responsible for its downstream manifestations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Azzam EI, de Toledo SM and Little JB . (2001). Proc. Natl. Acad. Sci. USA, 98, 473–478.
Azzam EI, de Toledo SM and Little JB . (2003). Cancer Res., 63, 7128–7135.
Azzam EI, de Toledo SM and Little JB . (2004). Curr. Cancer Drug Targets, 4, 53–64.
Banath JP, MacPhail SH and Olive PL . (2004). Cancer Res., 64, 7144–7149.
Bassing CH, Suh H, Ferguson DO, Chua KF, Manis J, Eckersdorff M, Gleason M, Bronson R, Lee C and Alt FW . (2003). Cell, 114, 359–370.
Bonner WM . (2004). Mutat. Res., 568, 33–39.
Brenner DJ, Little JB and Sachs RK . (2001). Radiat. Res., 155, 402–408.
Celeste A, Fernandez-Capetillo O, Kruhlak MJ, Pilch DR, Staudt DW, Lee A, Bonner WM and Nussenzweig A . (2003). Nat. Cell Biol., 5, 675–679.
Celeste A, Petersen S, Romanienko PJ, Fernandez-Capetillo O, Chen HT, Sedelnikova OA, Reina-San-Martin B, Coppola V, Meffre E, Difilippantonio MJ, Redon C, Pilch DR, Olaru A, Eckhaus M, Camerini-Otero RD, Tessarollo L, Livak F, Manova K, Bonner W, Nussenzweig M and Nussenzweig A . (2002). Science, 296, 922–927.
Chen HT, Bhandoola A, Difilippantonio MJ, Zhu J, Brown MJ, Tai X, Rogakou EP, Brotz TM, Bonner WM, Ried T and Nussenzweig A . (2000). Science, 290, 1962–1964.
Fernandez-Capetillo O, Lee A, Nussenzweig M and Nussenzweig A . (2004). DNA Repair, 3, 959–967.
Fernandez-Capetillo O, Mahadevaian SK, Celeste A, Romanienko PJ, Camerini-Otero RD, Bonner WM, Manova K, Burgoyne P and Nussenzweig A . (2003). Dev. Cell, 4, 497–508.
Hall EJ and Hei TK . (2003). Oncogene, 22, 7034–7042.
Highfield DP and Dewey WC . (1975). Methods Cell Biol., 9, 85–101.
Hill MA, Ford JR, Clapham P, Marsden SJ, Stevens DL, Townsend KM and Goodhead DT . (2005). Radiat. Res., 163, 36–44.
Kashino G, Prise KM, Schettino G, Folkard M, Vojnovic B, Michael BD, Suzuki K, Kodama S and Watanabe M . (2004). Mutat. Res., 556, 209–215.
Lyng FM, Seymour CB and Mothersill C . (2002). Radiat. Res., 157, 365–370.
MacPhail SH, Banath JP, Yu Y, Chu E and Olive PL . (2003). Radiat. Res., 159, 759–767.
Mahadevaian SK, Turner JMA, Rogakou EP, Baudat F, Blanko-Rodriguez J, Jasin M, Bonner WM and Burgoyne PS . (2001). Nat. Genet., 27, 271–276.
Marples B, Wouters BG and Joiner MC . (2003). Radiat. Res., 160, 38–45.
Mitchell SA, Marino SA, Brenner DJ and Hall EJ . (2004). Int. J. Radiat. Biol., 80, 465–472.
Mothersill C and Seymour C . (2001). Radiat. Res., 155, 759–767.
Mothersill C and Seymour C . (2004). Nat. Rev. Cancer, 4, 158–164.
Mothersill C, Seymour CB and Joiner MC . (2002). Radiat. Res., 157, 526–532.
Nagasawa H and Little JB . (1992). Cancer Res., 52, 6394–6396.
Paull TT, Rogakou EP, Yamazaki V, Kirchgessner C, Gellert M and Bonner WM . (2000). Curr. Biol., 10, 886–895.
Ponnaiya B, Jenkins-Baker G, Brenner DJ, Hall EJ, Randers-Pehrson G and Geard CR . (2004). Rad. Res., 162, 426–432.
Randers-Pehrson G, Geard CR, Johnson G, Elliston CD and Brenner DJ . (2001). Radiat. Res., 156, 210–214.
Redon C, Pilch DR, Rogakou E, Sedelnikova O, Newrock K and Bonner W . (2002). Curr. Opin. Genet. Dev., 12, 162–169.
Rogakou EP, Boon C, Redon C and Bonner WM . (1999). J. Cell Biol., 146, 905–915.
Rogakou EP, Pilch DR, Orr AH, Ivanova VS and Bonner WM . (1998). J. Biol. Chem., 273, 5858–5868.
Rothkamm K and Lobrich M . (2003). Proc. Natl. Acad. Sci. USA, 100, 5057–5062.
Sawant SG, Randers-Pehrson G, Geard CR, Brenner DJ and Hall EJ . (2001). Radiat. Res., 155, 397–401.
Sedelnikova OA, Horikawa I, Zimonjic DB, Popescu NC, Bonner WM and Barrett JC . (2004a). Nat. Cell Biol., 6, 168–170.
Sedelnikova OA, Panyutin IV, Neumann RD, Bonner WM and Panyutin IG . (2004b). Int. J. Radiat. Biol., 80, 927–931.
Sedelnikova OA, Rogakou EP, Panyutin IG and Bonner WM . (2002). Radiat. Res., 158, 486–492.
Seymour CB and Mothersill C . (2000). Radiat. Res., 153, 508–511.
Shao C, Folkard M, Michael BD and Prise KM . (2004). Proc. Natl. Acad. Sci. USA, 101, 13495–13500.
Shao C, Stewart M, Folkard M, Michael BD and Prise KM . (2003). Cancer Res., 63, 8437–8442.
Suzuki M, Zhou H, Geard CR and Hei TK . (2004). Radiat. Res., 162, 264–269.
Ward JF . (2000). Cold Spring Harb. Symp. Quant. Biol., 65, 377–382.
Wu L-J, Randers-Pehrson G, Xu A, Waldren CA, Geard CR, Yu ZL and Hei TK . (1999). Proc. Natl. Acad. Sci. USA, 96, 4959–4964.
Xie A, Puget N, Shim I, Odate S, Jarzyna I, Bassing CH, Alt FW and Scully R . (2004). Mol. Cell, 16, 1017–1025.
Yang H, Asaad N and Held KD . (2005). Oncogene, 24, 2096–2103.
Ye ZC, Wyeth MS, Baltan-Tekkok S and Ransom BR . (2003). J. Neurosci., 23, 3588–3596.
Zhou H, Randers-Pehrson G, Waldren CA, Vannais D, Hall EJ and Hei TK . (2000). Proc. Natl. Acad. Sci. USA, 97, 2099–2104.
Acknowledgements
We thank RARAF staff, Columbia University, for experimental support, R Neumann, NIH, and L Feinendegen, Heinrich-Heine-University Dusseldorf, for critical reading of the manuscript, W DeGraff, NIH, for help with γ-irradiation of the cells and I Kareva, NIH, for technical assistance. This work was supported by grants from the National Institutes of Health (NIH/NIBIB) (P41-EB002033), Department of Energy (DOE) (DE-FG02-03ER63629), and National Aeronautics and Space Administration (NASA) (NAG9-1519).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sokolov, M., Smilenov, L., Hall, E. et al. Ionizing radiation induces DNA double-strand breaks in bystander primary human fibroblasts. Oncogene 24, 7257–7265 (2005). https://doi.org/10.1038/sj.onc.1208886
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208886
Keywords
This article is cited by
-
The cross-talk between Bax, Bcl2, caspases, and DNA damage in bystander HepG2 cells is regulated by γ-radiation dose and time of conditioned media transfer
Apoptosis (2022)
-
Field size effects on DNA damage and proliferation in normal human cell populations irradiated with X-ray microbeams
Scientific Reports (2021)
-
Evaluation of the role of mitochondria in the non-targeted effects of ionizing radiation using cybrid cellular models
Scientific Reports (2020)
-
Cx43 channels and signaling via IP3/Ca2+, ATP, and ROS/NO propagate radiation-induced DNA damage to non-irradiated brain microvascular endothelial cells
Cell Death & Disease (2020)
-
Radioprotection of targeted and bystander cells by methylproamine
Strahlentherapie und Onkologie (2015)