Abstract
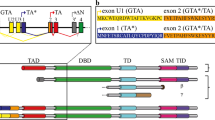
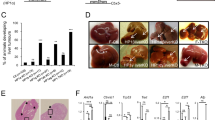
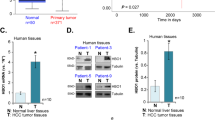
The TP63 gene, a member of the TP53 gene family, encodes several isoforms with (TAp63) or without (ΔNp63) transactivating properties. Whereas the role of p63 in the normal development of squamous epithelia is well established, its function in other cell types remains to be elucidated. Here, we have analysed the expression of TA and ΔNp63 isoforms in liver cells, by using both primary hepatocytes from wild type and p53-null mice and three human hepatocellular carcinoma (HCC) cell lines, according to the transformation state and the TP53 status of the cells. We observed the expression of ΔNp63 isoforms only in a p53-null context. On the other hand, the expression of TAp63 isoforms was restricted to the HCC cell lines, whatever the TP53 status. We then studied the expression of TP63 upon genotoxic treatment. When treated with UVB or H2O2, hepatocytes did not exhibit any change in p63 mRNA level. At the opposite, upon treatment with topoisomerase II inhibitors (doxorubicin or etoposide), the expression of TAp63 isoforms was clearly induced, independently of the TP53 status of cells. The same treatment did not induce any variation in the expression of ΔNp63 isoforms, both at mRNA and protein levels. In HCC cell lines, doxorubicin or etoposide treatment also resulted in an increase of TAp63 transcripts only. This increase was accompanied by an increase in the intracellular level of TAp63 alpha protein. In parallel, we observed an upregulation of some p53-target genes related to cell cycle regulation, such as WAF1/CIP1, PIG3, 14-3-3σ or GADD45, independently of the TP53 status of cells. In conclusion, we report for the first time that TA and ΔNp63 alpha proteins are present in liver cells. Furthermore, our results suggest that p63 may partially substitute for wild-type p53, in counteracting uncontrolled liver cell proliferation in response to certain forms of DNA-damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Benard J, Douc-Rasy S and Ahomadegbe JC . (2003). Hum. Mutat., 21, 182–191.
Bruix J and Llovet JM . (2002). Hepatology, 35, 519–524.
Chou YY, Cheng AL and Hsu HC . (1997). J. Gastroenterol. Hepatol., 12, 569–575.
Costanzo A, Merlo P, Pediconi N, Fulco M, Sartorelli V, Cole PA, Fontemaggi G, Fanciulli M, Schiltz L, Blandino G, Balsano C and Levrero M . (2002). Mol. Cell, 9, 175–186.
Courtois S, Caron de Fromentel C and Hainaut P . (2004). Oncogene, 23, 631–638.
De Laurenzi V, Rossi A, Terrinoni A, Barcaroli D, Levrero M, Costanzo A, Knight RA, Guerrieri P and Melino G . (2000). Biochem. Biophys. Res. Commun., 273, 342–346.
Dohn M, Zhang S and Chen X . (2001). Oncogene, 20, 3193–3205.
Gillet R, Grimber G, Bennoun M, Caron de Fromentel C, Briand P and Joulin V . (2000). Oncogene, 19, 3498–3507.
Gong JG, Costanzo A, Yang HQ, Melino G, Kaelin Jr WG, Levrero M and Wang JY . (1999). Nature, 399, 806–809.
Hainaut P and Hollstein M . (2000). Adv. Cancer Res., 77, 82–137.
Hande KR . (1998). Eur. J. Cancer, 34, 1514–1521.
Hortobagyi GN . (1997). Drugs, 54 (Suppl 4), 1–7.
Irwin MS and Kaelin WG . (2001). Cell Growth Differ., 12, 337–349.
Katoh I, Aisaki KI, Kurata SI, Ikawa S and Ikawa Y . (2000). Oncogene, 19, 3126–3130.
Kartasheva NN, Contente A, Lenz-Stoppler C, Roth J and Dobbelstein M . (2002). Oncogene, 18, 4715–4727.
Lee TK, Lau TC and Ng IO . (2002). Cancer Chemother. Pharmacol., 49, 78–86.
Levrero M, De Laurenzi V, Costanzo A, Gong J, Wang JY and Melino G . (2000). J. Cell Sci., 113 (Part 10), 1661–1670.
Liefer KM, Koster MI, Wang XJ, Yang A, McKeon F and Roop DR . (2000). Cancer Res., 60, 4016–4020.
Montesano R, Hainaut P and Wild CP . (1997). J. Natl. Cancer Inst., 89, 1844–1851.
Mueller H and Eppenberger U . (1996). Anticancer Res., 16, 3845–3848.
Okada Y, Osada M, Kurata S, Sato S, Aisaki K, Kageyama Y, Kihara K, Ikawa Y and Katoh I . (2002). Exp. Cell Res., 276, 194–200.
Parkin DM, Bray F, Ferlay J and Pisani P . (2001). Int. J. Cancer, 94, 153–156.
Pediconi N, Ianari A, Costanzo A, Belloni L, Gallo R, Cimino L, Porcellini A, Screpanti I, Balsano C, Alesse E, Gulino A and Levrero M . (2003). Nat. Cell Biol., 5, 552–558.
Pluquet O and Hainaut P . (2001). Cancer Lett., 174, 1–15.
Sayan AE, Sayan BS, Findikli N and Ozturk M . (2001). Oncogene, 20, 5111–5117.
Shen HM and Ong CN . (1996). Mutat. Res., 366, 23–44.
Staib F, Hussain SP, Hofseth LJ, Wang XW and Harris CC . (2003). Hum. Mutat., 21, 201–216.
Stein B, Rahmsdorf HJ, Steffen A, Litfin M and Herrlich P . (1989). Mol. Cell Biol., 9, 5169–5181.
van Bokhoven H and McKeon F . (2002). Trends Mol. Med., 8, 133–139.
Vogelstein B, Lane D and Levine AJ . (2000). Nature, 408, 307–310.
Vossio S, Palescandolo E, Pediconi N, Moretti F, Balsano C, Levrero M and Costanzo A . (2002). Oncogene, 21, 3796–3803.
Waltermann A, Kartasheva NN and Dobbelstein M . (2003). Oncogene, 22, 5686–5693.
Wu G, Nomoto S, Hoque MO, Dracheva T, Osada M, Lee CC, Dong SM, Guo Z, Benoit N, Cohen Y, Rechthand P, Califano J, Moon CS, Ratovitski E, Jen J, Sidransky D and Trink B . (2003). Cancer Res., 63, 2351–2357.
Yang A, Kaghad M, Caput D and McKeon F . (2002). Trends Genet., 18, 90–95.
Yang A, Schweitzer R, Sun D, Kaghad M, Walker N, Bronson RT, Tabin C, Sharpe A, Caput D, Crum C and McKeon F . (1999). Nature, 398, 714–718.
Yuan ZM, Shioya H, Ishiko T, Sun X, Gu J, Huang YY, Lu H, Kharbanda S, Weichselbaum R and Kufe D . (1999). Nature, 399, 814–817.
Zhu H, Chang BD, Uchiumi T and Roninson IB . (2002). Cell Cycle, 1, 59–66.
Acknowledgements
We thank Drs V Joulin and P Briand for their support to the work. This study was supported by grants from Electricité De France and Association pour la Recherche contre le Cancer, France.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petitjean, A., Cavard, C., Shi, H. et al. The expression of TA and ΔNp63 are regulated by different mechanisms in liver cells. Oncogene 24, 512–519 (2005). https://doi.org/10.1038/sj.onc.1208215
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208215
Keywords
This article is cited by
-
The sodium/iodide symporter NIS is a transcriptional target of the p53-family members in liver cancer cells
Cell Death & Disease (2013)
-
Role of p63 in Development, Tumorigenesis and Cancer Progression
Cancer Microenvironment (2012)
-
TP63 P2 promoter functional analysis identifies β-catenin as a key regulator of ΔNp63 expression
Oncogene (2011)
-
Resveratrol-induced p53-independent apoptosis of human nasopharyngeal carcinoma cells is correlated with the downregulation of ΔNp63
Cancer Gene Therapy (2010)
-
Plk1 regulates liver tumor cell death by phosphorylation of TAp63
Oncogene (2009)