Abstract
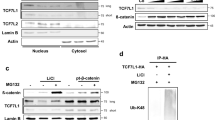
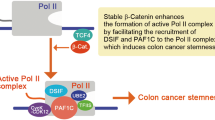
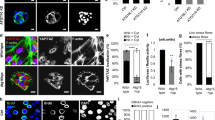
Aberrant activation of the Wnt pathway is observed in numerous cancers, and is particularly important in colon cancer. We demonstrate that Rac1 GTPase can significantly increase the signaling activity of β-catenin in cells with inherent dysregulation of the canonical Wnt signaling pathway. Expression of dominant-negative (N17)Rac1 mutant in colon cancer cells caused a marked inhibition of Wnt signaling, as determined by the TCF/LEF-responsive (TOPFLASH) transcription assay. Expression of a constitutively active (V12)Rac1 mutant caused up to 40-fold induction from the TOPFLASH promoter, and this was dependent on the presence of stabilized β-catenin. This induction was completely blocked by the expression of dominant-negative TCF-4, suggesting that β-catenin and TCF-4 complex formation is required for Rac1-mediated transcription. Furthermore, we show that Cyclin D1, an important biological Wnt target gene, is regulated by Rac1 in a β-catenin/TCF-dependent manner. We observed that Rac1 co-immunoprecipitates with β-catenin and TCF-4 only in its active GTP-bound form. Both cell fractionation studies and fluorescence microscopy indicate that overexpression of V12Rac1 results in increased cytosolic and nuclear expression of β-catenin. Interestingly, mutation of the polybasic region of Rac1, which prevents its nuclear localization, also caused an appreciable decrease in nuclear localization of β-catenin, and effectively abolished its β-catenin-dependent transcription co-activator function. Taken together, our data demonstrate a novel mechanism of Wnt pathway regulation whereby activation of Rac1 amplifies the signaling activity of stabilized/mutated β-catenin by promoting its accumulation in the nucleus, and synergizing with β-catenin to augment TCF/LEF-dependent gene transcription.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Aspenstrom P . (1999). Curr. Opin. Cell Biol., 11, 95–102.
Briggs MW, Li Z and Sacks DB . (2002). J. Biol. Chem., 277, 7453–7465.
Chan TA, Wang Z, Dang LH, Vogelstein B and Kinzler KW . (2002). Proc. Natl. Acad. Sci. USA, 99, 8265–8270.
Coso OA, Chiariello M, Yu JC, Teramoto H, Crespo P, Xu N, Miki T and Gutkind JS . (1995). Cell, 81, 1137–1146.
Fritz G, Just I and Kaina B . (1999). Int. J. Cancer, 81, 682–687.
Frost JA, Xu S, Hutchison MR, Marcus S and Cobb MH . (1996). Mol. Cell Biol., 16, 3707–3713.
Fukata M, Kuroda S, Nakagawa M, Kawajiri A, Itoh N, Shoji I, Matsuura Y, Yonehara S, Fujisawa H, Kikuchi A and Kaibuchi K . (1999). J. Biol. Chem., 274, 26044–26050.
Habets GG, Scholtes EH, Zuydgeest D, van der Kammen RA, Stam JC, Berns A and Collard JG . (1994). Cell, 77, 537–549.
Hall A . (1998). Science, 279, 509–514.
Huelsken J and Birchmeier W . (2001). Curr. Opin. Genet. Dev., 11, 547–553.
Ishitani T, Ninomiya-Tsuji J, Nagai S, Nishita M, Meneghini M, Barker N, Waterman M, Bowerman B, Clevers H, Shibuya H and Matsumoto K . (1999). Nature, 399, 798–802.
Jordan P, Brazao R, Boavida MG, Gespach C and Chastre E . (1999). Oncogene, 18, 6835–6839.
Joyce D, Bouzahzah B, Fu M, Albanese C, D'Amico M, Steer J, Klein JU, Lee RJ, Segall JE, Westwick JK, Der CJ and Pestell RG . (1999). J. Biol. Chem., 274, 25245–25249.
Kishida S, Yamamoto H, Ikeda S, Kishida M, Sakamoto I, Koyama S and Kikuchi A . (1998). J. Biol. Chem., 273, 10823–10826.
Korinek V, Barker N, Morin PJ, van Wichen D, de Weger R, Kinzler KW, Vogelstein B and Clevers H . (1997). Science, 275, 1784–1787.
Kuroda S, Fukata M, Nakagawa M, Fujii K, Nakamura T, Ookubo T, Izawa I, Nagase T, Nomura N, Tani H, Shoji I, Matsuura Y, Yonehara S and Kaibuchi K . (1998). Science, 281, 832–835.
Lanning CC, Ruiz-Velasco R and Williams CL . (2003). J. Biol. Chem., 278, 12495–12506.
Liu J, Stevens J, Rote CA, Yost HJ, Hu Y, Neufeld KL, White RL and Matsunami N . (2001). Mol. Cell, 7, 927–936.
Malliri A and Collard JG . (2003). Curr. Opin. Cell Biol., 15, 583–589.
Meigs TE, Fields TA, McKee DD and Casey PJ . (2001). Proc. Natl. Acad. Sci. USA, 98, 519–524.
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B and Kinzler KW . (1997). Science, 275, 1787–1790.
Munemitsu S, Albert I, Souza B, Rubinfeld B and Polakis P . (1995). Proc. Natl. Acad. Sci. USA, 92, 3046–3050.
Murga C, Zohar M, Teramoto H and Gutkind JS . (2002). Oncogene, 21, 207–216.
Nishita M, Hashimoto MK, Ogata S, Laurent MN, Ueno N, Shibuya H and Cho KW . (2000). Nature, 403, 781–785.
O'Connor KL and Mercurio AM . (2001). J. Biol. Chem., 276, 47895–47900.
Perona R, Montaner S, Saniger L, Sanchez-Perez I, Bravo R and Lacal JC . (1997). Genes Dev., 11, 463–475.
Polakis P . (2000). Genes Dev., 14, 1837–1851.
Roose J and Clevers H . (1999). Biochim. Biophys. Acta, 1424, M23–37.
Sahai E and Marshall CJ . (2002). Nat. Rev. Cancer, 2, 133–142.
Salgia R, Li JL, Lo SH, Brunkhorst B, Kansas GS, Sobhany ES, Sun Y, Pisick E, Hallek M, Ernst T, Tantravahi R, Chen LB and Griffin JD . (1995). J. Biol. Chem., 270, 5039–5047.
Sampson EM, Haque ZK, Ku MC, Tevosian SG, Albanese C, Pestell RG, Paulson KE and Yee AS . (2001). EMBO J., 20, 4500–4511.
Schmitz AA, Govek EE, Bottner B and Van Aelst L . (2000). Exp. Cell Res., 261, 1–12.
Schnelzer A, Prechtel D, Knaus U, Dehne K, Gerhard M, Graeff H, Harbeck N, Schmitt M and Lengyel E . (2000). Oncogene, 19, 3013–3020.
Seeling JM, Miller JR, Gil R, Moon RT, White R and Virshup DM . (1999). Science, 283, 2089–2091.
Seidensticker MJ and Behrens J . (2000). Biochim. Biophys. Acta, 1495, 168–182.
Sharpe C, Lawrence N and Martinez Arias A . (2001). Bioessays, 23, 311–318.
Tetsu O and McCormick F . (1999). Nature, 398, 422–426.
Wang JC . (1985). Annu. Rev. Biochem., 54, 665–697.
Zorn AM, Barish GD, Williams BO, Lavender P, Klymkowsky MW and Varmus HE . (1999). Mol. Cell, 4, 487–498.
Acknowledgements
We thank S Kulkarni and L Dixon for their expert technical assistance, and our colleagues mentioned in Materials and methods for kindly providing reagents. This work was supported by the National Cancer Institute of Canada, Grant No. 010277 (BB), Canadian Institute For Health Research, Grant No. MOP_64225 (BB), Ontario Graduate Scholarship (SE), and the Ontario Student Opportunity Trust Funds (SE).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Esufali, S., Bapat, B. Cross-talk between Rac1 GTPase and dysregulated Wnt signaling pathway leads to cellular redistribution of β-catenin and TCF/LEF-mediated transcriptional activation. Oncogene 23, 8260–8271 (2004). https://doi.org/10.1038/sj.onc.1208007
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208007
Keywords
This article is cited by
-
Muscarinic receptor agonist-induced βPix binding to β-catenin promotes colon neoplasia
Scientific Reports (2023)
-
Connecting DCX, COMT and FMR1 in social behavior and cognitive impairment
Behavioral and Brain Functions (2022)
-
Phosphatidylserine recognition and Rac1 activation are required for Müller glia proliferation, gliosis and phagocytosis after retinal injury
Scientific Reports (2020)
-
Blockade of ARHGAP11A reverses malignant progress via inactivating Rac1B in hepatocellular carcinoma
Cell Communication and Signaling (2018)
-
PAK2–c-Myc–PKM2 axis plays an essential role in head and neck oncogenesis via regulating Warburg effect
Cell Death & Disease (2018)