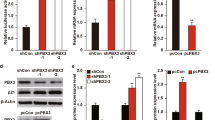
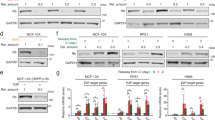
Abstract
p21 is a potent inhibitor of cyclin-dependent kinases capable of arresting cell cycle progression. p21 is primarily regulated at the transcriptional level by several transcription factors, including p53. Previously, we reported that certain members of the E2F family of transcription factors may activate p21 transcription via a p53-independent mechanism. To further elucidate the consequences of E2F-1-regulated induction of p21, we developed cell lines with a tamoxifen-dependent form of E2F-1. We confirmed direct interaction of E2F-1 with the proximal region of the p21 promoter. Interestingly, elevated E2F-1 activity was sufficient to arrest a substantial subset of cells in S phase and this effect was correlated to and dependent on the induction of p21 protein. Since E2F proteins control genes required for cell cycle progression and are activated by various oncogenic events, we believe that the p21-dependent arrest described in this report represents an additional mechanism that guards against unrestricted cell proliferation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Brugarolas J, Chadrasekaran C, Gordon J, Beach D, Jacks T and Hannon G . (1995). Nature, 377, 552–557.
Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP, Sedivy JM, Kinzler KW and Vogelstein B . (1998). Science, 282, 1497–1501.
Deng C, Zhang P, Harper JW, Elledge SJ and Leder P . (1995). Cell, 82, 675–684.
Dolbeare F, Gratzner H, Pallavicini MG and Gray JW . (1983). Proc. Natl. Acad. Sci. USA, 80, 5573–5577.
El-Deiry W, Tokino T, Velculescu V, Levy D, Parsons R, Trent J, Lin D, Mercer WE, Kinzler K and Vogelstein B . (1993). Cell, 75, 817–825.
Farhana L, Dawson M, Rishi AK, Zhang Y, Van Buren E, Trivedi C, Reichert U, Fang G, Kirschner MW and Fontana JA . (2002). Cancer Res., 62, 3842–3849.
Gartel AL, Goufman E, Tevosian SG, Shih H, Yee AS and Tyner AL . (1998). Oncogene, 17, 3463–3469.
Gartel AL, Serfas MS and Tyner AL . (1996). Proc. Soc. Exp. Biol. Med., 213, 138–149.
Gartel AL and Tyner AL . (1998). Prog. Mol. Subcell. Biol., 20, 43–71.
Gille H and Downward J . (1999). J Biol. Chem., 274, 22033–22040.
Harper JW, Adami G, Wei N, Keyomarsi K and Elledge S . (1993). Cell, 75, 805–816.
Hiyama H, Iavarone A and Reeves SA . (1998). Oncogene, 16, 1513–1523.
Johnson DG, Schwarz JK, Cress WD and Nevins JR . (1993). Nature, 365, 349–352.
Krek W, Xu G and Livingston DM . (1995). Cell, 83, 1149–1158.
Logan TJ, Evans DL, Mercer WE, Bjornsti MA and Hall DJ . (1995). Cancer Res., 55, 2883–2891.
Ma Y, Croxton R, Moorer Jr RL and Cress WD . (2002). Arch. Biochem. Biophys., 399, 212–224.
Muller H, Bracken AP, Vernell R, Moroni MC, Christians F, Grassilli E, Prosperini E, Vigo E, Oliner JD and Helin K . (2001). Genes Dev., 15, 267–285.
Nevins JR . (2001). Hum. Mol. Genet., 10, 699–703.
Ogryzko VV, Wong P and Howard BH . (1997). Mol. Cell. Biol., 17, 4877–4882.
Ohtani K, DeGregori J and Nevins JR . (1995). Proc. Natl. Acad. Sci. USA, 92, 12146–12150.
Shan B and Lee WH . (1994). Mol. Cell. Biol., 14, 8166–8173.
Spruck CH, Won KA and Reed SI . (1999). Nature, 401, 297–300.
Takahashi Y, Rayman JB and Dynlacht BD . (2000). Genes Dev., 14, 804–816.
Tsai KY, Hu Y, Macleod KF, Crowley D, Yamasaki L and Jacks T . (1998). Mol. Cell, 2, 293–304.
Vigo E, Muller H, Prosperini E, Hateboer G, Cartwright P, Moroni MC and Helin K . (1999). Mol. Cell. Biol., 19, 6379–6395.
Waldman T, Kinzler KW and Vogelstein B . (1995). Cancer Res., 55, 5187–5190.
Yamasaki L, Bronson R, Williams BO, Dyson NJ, Harlow E and Jacks T . (1998). Nat. Genet., 18, 360–364.
Zhang Y, Rishi AK, Dawson MI, Tschang R, Farhana L, Boyanapalli M, Reichert U, Shroot B, Van Buren EC and Fontana JA . (2000). Cancer Res., 60, 2025–2032.
Acknowledgements
This work was supported by IDPH Grant (ALG), NIH Grant CA91146-01A1 (ALG) and NIH Grant DK48836 (ALT).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radhakrishnan, S., Feliciano, C., Najmabadi, F. et al. Constitutive expression of E2F-1 leads to p21-dependent cell cycle arrest in S phase of the cell cycle. Oncogene 23, 4173–4176 (2004). https://doi.org/10.1038/sj.onc.1207571
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207571
Keywords
This article is cited by
-
Stomatin-like protein 2 promotes cell proliferation and survival under 5-Fluorouracil stress in hepatocellular carcinoma
Molecular Biology Reports (2024)
-
Ghrelin enhances cisplatin sensitivity in HO-8910 PM human ovarian cancer cells
Journal of Ovarian Research (2021)
-
Exposure of the cytoplasm to low-dose X-rays modifies ataxia telangiectasia mutated-mediated DNA damage responses
Scientific Reports (2021)
-
Multiple functions of p21 in cancer radiotherapy
Journal of Cancer Research and Clinical Oncology (2021)
-
Efficient gene editing of human long-term hematopoietic stem cells validated by clonal tracking
Nature Biotechnology (2020)