Abstract
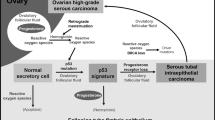
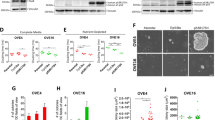
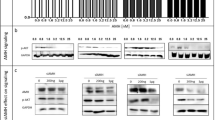
Progesterone (P4) has been implicated as a protective factor for epithelial ovarian cancers, yet little is known about its mechanism of action. We previously reported that pregnancy-equivalent doses of P4 inhibited the growth of normal and malignant human ovarian surface epithelial (HOSE) cells. Here, we investigated how P4-induced cell death in two immortalized normal (HOSE 642, HOSE 12-12) and two malignant (OVCA 429, OVCA 432) HOSE cell lines. The exposure of HOSE or OVCA cell cultures to 10−6 M P4 induced time-dependent increases in early and late apoptotic cells and activation of caspase-8 and -3, but not that of caspase-9. A general caspase inhibitor Z-VAD effectively blocked the P4-induced cell death in a dose-dependent manner. Comparable levels of Fas mRNA and protein were expressed in HOSE and OVCA cell lines, and these levels were unaffected by P4. In contrast, levels of FasL mRNA and protein were higher in OVCA cells than in HOSE cells. Interestingly, the hormone enhanced levels of FasL mRNA and protein in HOSE cells, but lowered their levels in OVCA cells. The exposure of HOSE or OVCA cells to an activating anti-Fas antibody induced cell loss, whereas treatment of cells with a blocking anti-FasL antibody reduced the P4-induced cell loss. Cotreatment of cells with the activating anti-Fas antibody and P4 produced additive effects on cell loss. These results reveal for the first time that P4 induces apoptosis in HOSE and OVCA cells via activation of a caspase-8-initiated Fas/FasL signaling pathway. They also demonstrate differential P4-regulation of FasL expression between HOSE and OVCA cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Adami HO, Hsieh CC, Lambe M, Trichopoulos D, Leon D, Persson I, Ekbom A and Janson PO . (1994). Lancet, 344, 1250–1254.
Adami HO, Lambe M, Persson I, Ekbom A, Hsieh CC and Trichopoulos D . (1995). Lancet, 345, 789.
Akhmedkhanov A, Zeleniuch-Jacquotte A and Toniolo P . (2001). Ann. N.Y. Acad. Sci., 943, 296–315.
Alnemri ES, Livingston DJ, Nicholson DW, Salvesen G, Thornberry NA, Wong WW and Yuan J . (1996). Cell, 87, 171.
Ashkenazi A and Dixit VM . (1998). Science, 281, 1305–1308.
Bast Jr RC, Feeney M, Lazarus H, Nadler LM, Colvin RB and Knapp RC . (1981). J. Clin. Invest., 68, 1331–1337.
Bennett MW, O'Connell J, Houston A, Kelly J, O'Sullivan GC, Collins JK and Shanahan F . (2001). J. Clin. Pathol., 54, 598–604.
Bennett MW, O'Connell J, O'Sullivan GC, Brady C, Roche D, Collins JK and Shanahan F . (1998). J. Immunol., 160, 5669–5675.
Bu SZ, Yin DL, Ren XH, Jiang LZ, Wu ZJ, Gao QR and Pei G . (1997). Cancer, 79, 1944–1950.
Cramer DW, Hutchison B, Welch WR, Scully RE and Ryan KJ . (1983). J. Natl. Can. Inst., 71, 711–716.
Cramer DW and Welch WR . (1993). J. Natl. Cancer Inst., 71, 717–721.
Das H, Koizumi T, Sugimoto T, Chakraborty S, Ichimura T, Hasegawa K and Nishimura R . (2000). Br. J. Cancer, 82, 1682–1688.
Earnshaw WC, Martins LM and Kaufmann SH . (1999). Annu. Rev. Biochem., 68, 383–424.
Ghahremani M, Foghi A and Dorrington JH . (1998). Gynecol. Oncol., 70, 275–281.
Gratas C, Tohma Y, Barnas C, Taniere P, Hainaut P and Ohgaki H . (1998). Cancer Res., 58, 2057–2062.
Griffith TS and Ferguson TA . (1997). Immunol. Today, 18, 240–244.
Hayakawa A, Wu J, Kawamoto Y, Zhou YW, Tanuma S, Nakashima I and Suzuki H . (2002). Apoptosis, 7, 107–113.
Hu Z and Deng X . (2000). Zhonghua Fu Chan Ke Za Zhi, 35, 423–426.
Keane MM, Ettenberg SA, Lowrey GA, Russell EK and Lipkowitz S . (1996). Cancer Res., 56, 4791–4798.
Lambe M, Wuu J, Rossing MA and Hsieh CC . (1999). Lancet, 353, 1941.
Lepple-Wienhues A, Belka C, Laun T, Jekle A, Walter B, Wieland U, Welz M, Heil L, Kun J, Busch G, Weller M, Bamberg M, Gulbins E and Lang F . (1999). Proc. Natl. Acad. Sci. USA, 96, 13795–13800.
Milner AE, Palmer DH, Hodgkin EA, Eliopoulos AG, Knox PG, Poole CJ, Kerr DJ and Young LS . (2002). Cell Death Differ., 9, 287–300.
Moller P, Koretz K, Leithauser F, Bruderlein S, Henne C, Quentmeier A and Krammer PH . (1994). Int. J. Cancer, 57, 371–377.
Munakata S, Enomoto T, Tsujimoto M, Otsuki Y, Miwa H, Kanno H and Aozasa K . (2000). Br. J. Cancer, 84, 1446–1452.
Murdoch WJ and McDonnel AC . (2002). Reproduction, 123, 743–750.
Nagata S . (1994). Philos. Trans. R. Soc. London B. Biol. Sci., 345, 281–287.
O'Connell J, Houston A, Bennett MW, O'Sullivan GC and Shanahan F . (2001). Nat. Med., 7, 271–274.
O'Connell J, O'Sullivan GC, Collins JK and Shanahan F . (1996). J. Exp. Med., 184, 1075–1082.
Pinkoski MJ and Green DR . (2000). Curr. Opin. Genet. Dev., 10, 114–119.
Reimer T, Herrnring C, Koczan D, Richter D, Gerber B, Kabelitz D, Friese K and Thiesen HJ . (2000). Cancer Res., 60, 822–828.
Riman T, Persson I and Nilsson S . (1998). Clin. Endocrinol. (Oxf.), 49, 695–707.
Rodriguez GC, Nagarsheth NP, Lee KL, Bentley RC, Walmer DK, Cline M, Whitaker RS, Isner P, Berchuck A, Dodge RK and Hughes CL . (2002). J. Natl. Cancer Inst., 94, 50–60.
Sampalo A, Navas G, Medina F, Segundo C, Camara C and Brieva JA . (2000). Blood, 96, 3168–3174.
Sapi E, Brown WD, Aschkenazi S, Lim C, Munoz A, Kacinski BM, Rutherford T and Mor G . (2002). J. Soc. Gynecol. Invest., 9, 243–250.
Selam B, Kayisli UA, Mulayim N and Arici A . (2001). Biol. Reprod., 65, 979–985.
Song K, Chen Y, Goke R, Wilmen A, Seidel C, Goke A and Hilliard B . (2000). J. Exp. Med., 191, 1095–1104.
Song J, Rutherford T, Naftolin F, Brown S and Mor G . (2002). Mol. Hum. Reprod., 8, 447–455.
Strand S, Hofmann WJ, Hug H, Muller M, Otto G, Strand D, Mariani SM, Stremmel W, Krammer PH and Galle PR . (1996). Nat. Med., 2, 1361–1366.
Suda T, Takahashi T, Golstein P and Nagata S . (1993). Cell, 75, 1169–1178.
Syed V, Ulinski G, Mok SC, Yiu GK and Ho SM . (2001). Cancer Res., 61, 6768–6776.
Syed V, Ulinski G, Mok SC and Ho SM . (2002). J. Natl. Cancer Inst., 94, 617–629.
Tsao SW, Mok SC, Fey E G, Fletcher J A, Muto MG, Knapp RC and Berkowitz RS . (1995). Exp. Cell Res., 218, 499–507.
Ungefroren H, Voss M, Jansen M, Roeder C, Henne-Bruns D, Kremer B and Kalthoff H . (1998). Cancer Res., 58, 1741–1749.
van Haaften-Day C, Russell P, Davies S, King NJ and Tattersall MH . (2003). Hum. Pathol., 34, 74–79.
Wang S, Pudney J, Song J, Mor G, Schwartz PE and Zheng W . (2003). Gynecol. Oncol., 88, 108–117.
Wajant H . (2002). Science, 296, 1635–1636.
Yonehara S, Ishii A and Yonehara M . (1989). J. Exp. Med., 169, 1747–1756.
Yu S, Lee M, Shin S and Park J . (2001). J. Cell Biochem., 82, 445–451.
Zimmermann KC, Bonzon C and Green DR . (2001). Pharmacol. Ther., 92, 57–70.
Zou H, Li Y, Liu X and Wang X . (1999). J. Biol. Chem., 274, 11549–11556.
Acknowledgements
This study was supported by an Army Ovarian Cancer Research Program Grant DAMD17-99-1-9563 (to S-M Ho) and NIH Grants CA091250 (to V Syed) and CA94221 (to S-M Ho). This publication was made possible by Grant Number 5 P30 DK32520 from the National Institute of Diabetes and Digestive and Kidney Diseases'. We thank Dr Samuel C Mok of Brigham and Women's Hospital, Boston, Massachusetts, for his generous gifts of HOSE and OVCA cell lines and editorial staff of Brigham and Women's Hospital, Boston, for editorial help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Syed, V., Ho, SM. Progesterone-induced apoptosis in immortalized normal and malignant human ovarian surface epithelial cells involves enhanced expression of FasL. Oncogene 22, 6883–6890 (2003). https://doi.org/10.1038/sj.onc.1206828
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206828
Keywords
This article is cited by
-
Progesterone reduces cell survival in primary cultures of endometrioid ovarian cancer
Journal of Ovarian Research (2019)
-
In vitro antitumor activity of progesterone in human adrenocortical carcinoma
Endocrine (2019)
-
Expression of Membrane Progesterone Receptors (mPR/PAQR) in Ovarian Cancer Cells: Implications for Progesterone-Induced Signaling Events
Hormones and Cancer (2010)
-
Präventionsstrategien beim Ovarialkarzinom
Der Gynäkologe (2008)
-
Birth spacing and maternal risk of invasive epithelial ovarian cancer in a Swedish nationwide cohort
Cancer Causes & Control (2008)