Abstract
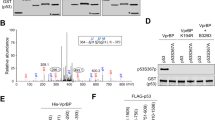
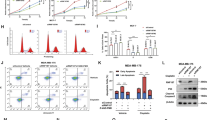
The mechanisms by which p53 prevents development of cancer are much more complicated than previously thought. Under normal conditions, p53 is involved in cell-cycle arrest, Q1apoptosis, DNA repair, and inhibition of angiogenesis; it also promotes degradation of proteins through transcriptional regulation of certain target genes. Here we report the isolation of a novel transcriptional target of p53, designated p53RFP (p53-inducible RING-finger protein), whose product has E3 ubiquitin ligase activity. Its expression was negatively correlated to that of p21WAF1 protein; p53RFP is likely to play a role in the regulation of this protein, probably through interaction with, and ubiquitination of, p21WAF1. p53RFP appears to represent the second known example, the first being MDM2, of an E3 ubiquitin ligase as a p53 target. Our results further suggest that p53 might regulate the stability of p21WAF1 through transcriptional regulation of p53RFP, and this feature may represent a novel mechanism for a p53-dependent cell-cycle checkpoint.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bordallo J, Plemper RK, Finger A and Wolf DH . (1998). Mol. Biol. Cell., 9, 209–222.
Blagosklonny MV, Wu GS, Omura S and el-Deiry WS . (1996). Biochem. Biophys. Res. Commun., 227, 564–569.
Cayrol C and Ducommun B . (1998). Oncogene, 17, 2437–2444
el-Deiry WS, Kern SE, Pietenpol JA, Kinzler KW and Vogelstein B . (1992). Nat. Genet., 1, 45–49
Fang S, Jensen JP, Ludwig RL, Vousden KH and Weissman AM . (2000). J. Biol. Chem., 275, 8945–8951
Fukuchi K, Maruyama H, Takagi Y and Gomi K . (1999). Biochim. Biophys. Acta, 1451, 206–210
Harper JW, Adami GR, Wei N, Keyomarsi K and Elledge SJ . (1993). Cell, 75, 805–816
Hollstein M, Rice K, Greenblatt MS, Soussi T, Fuchs R, Sorlie T, Hovig E, Smith-Sorensen B, Montesano R and Harris CC . (1994). Nucleic Acids Res., 22, 3551–3555
Hupp TR, Lane DP and Ball KL . (2000). Biochem. J., 352(Part 1), 1–17
Jackson PK, Eldridge AG, Freed E, Furstenthal L, Hsu JY, Kaiser BK and Reimann JD . (2000). Trends Cell. Biol., 10, 429–439
Joazeiro CA and Weissman AM . (2000). Cell, 102, 549–552
Marin I and Ferrus A . (2002). Mol. Biol. Evol., 19, 2039–2050
Matsuda N, Suzuki T, Tanaka K and Nakano A . (2001). J. Cell. Sci., 114, 1949–1957
Mimnaugh EG, Bonvini P and Neckers L . (1999). Electrophoresis, 20, 418–428
Oda K, Arakawa H, Tanaka T, Matsuda K, Tanikawa C, Mori T, Nishimori H, Tamai K, Tokino T, Nakamura Y and Taya Y . (2000). Cell, 102, 849–862
Okamura S, Arakawa H, Tanaka T, Nakanishi H, Ng CC, Taya Y, Monden M and Nakamura Y . (2001). Mol. Cell, 8, 85–94
Pickart CM . (2001). Mol. Cell, 8, 499–504
Rousseau D, Cannella D, Boulaire J, Fitzgerald P, Fotedar A and Fotedar R . (1999). Oncogene, 18, 4313–4325
Sasaki Y, Ishida S, Morimoto I, Yamashita T, Kojima T, Kihara C, Tanaka T, Imai K, Nakamura Y and Tokino T . (2002). J. Biol. Chem., 277, 719–724
Sheaff RJ, Singer JD, Swanger J, Smitherman M, Roberts JM and Clurman BE . (2000). Mol. Cell, 5, 403–410
Tokino T and Nakamura Y . (2000). Crit. Rev. Oncol. Hematol., 33, 1–6
Touitou R, Richardson J, Bose S, Nakanishi M, Rivett J and Allday MJ . (2001). EMBO J., 20, 2367–2375
Vogelstein B, Lane D and Levine AJ . (2000). Nature, 408, 307–310
Yu ZK, Gervais JL and Zhang H . (1998). Proc. Natl. Acad. Sci. USA., 95, 11324–11329
Yu J, Zhang L, Hwang PM, Rago C, Kinzler KW and Vogelstein B . (1999). Proc. Natl. Acad. Sci. USA, 96, 14517–14522
Zhang Y, Gao J, Chung KK, Huang H, Dawson VL and Dawson TM . (2000). Proc. Natl. Acad. Sci. USA., 97, 13354–13359
Zhao R, Gish K, Murphy M, Yin Y, Notterman D, Hoffman WH, Tom E, Mack DH and Levine AJ . (2000). Genes Dev., 14, 981–993
Acknowledgements
We thank Dr Bert Vogelstein for HCT116 (p53+/+) and HCT116 (p53−/−) cell line and Dr K Miyazono for the HA-ubiquitin plasmid. We also thank K Matsui and S Onoue for their excellent technical assistance. This work was supported in part by Research for the Future Program Grant 00 L01402 from the Japan Society for the Promotion of Science (to YN), and in part by Grant 13216031 from the Ministry of Education, Culture, Sports, Science and Technology (to HA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ng, CC., Arakawa, H., Fukuda, S. et al. p53RFP, a p53-inducible RING-finger protein, regulates the stability of p21WAF1. Oncogene 22, 4449–4458 (2003). https://doi.org/10.1038/sj.onc.1206586
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206586
Keywords
This article is cited by
-
RIG-I promotes cell proliferation in esophageal squamous cell carcinoma by facilitating p21 degradation
Medical Oncology (2023)
-
Mapping the p53 transcriptome universe using p53 natural polymorphs
Cell Death & Differentiation (2014)
-
Stabilization of p21 (Cip1/WAF1) following Tip60-dependent acetylation is required for p21-mediated DNA damage response
Cell Death & Differentiation (2013)
-
PIR2/Rnf144B regulates epithelial homeostasis by mediating degradation of p21WAF1 and p63
Oncogene (2013)
-
IBRDC2, an IBR-type E3 ubiquitin ligase, is a regulatory factor for Bax and apoptosis activation
The EMBO Journal (2010)