Abstract
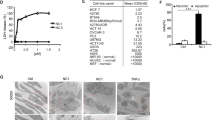
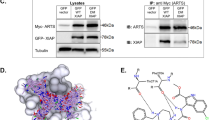
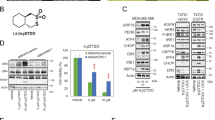
p53 is the most frequently altered gene in human cancer and therefore represents an ideal target for cancer therapy. Several amino terminal p53-derived synthetic peptides were tested for their antiproliferative effects on breast cancer cell lines MDA-MB-468 (mutant p53), MCF-7 (overexpressed wild-type p53), and MDA-MB-157 (null p53). p53(15)Ant peptide representing the majority of the mouse double minute clone 2 binding site on p53 (amino acids 12–26) fused to the Drosophila carrier protein Antennapedia was the most effective. p53(15)Ant peptide induced rapid, nonapoptotic cell death resembling necrosis in all breast cancer cells; however, minimal cytotoxicity was observed in the nonmalignant breast epithelial cells MCF-10–2A and MCF-10F. Bioinformatic/biophysical analysis utilizing hydrophobic moment and secondary structure predictions as well as circular dichroism spectroscopy revealed an α-helical hydrophobic peptide structure with membrane disruptive potential. Based on these findings, p53(15)Ant peptide may be a novel peptide cancer therapeutic because it induces necrotic cell death and not apoptosis, which is uncommon in traditional cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Abbreviations
- CFU-GEMM:
-
Colony forming units for granulocytes, erythryocytes, monocytes, and macrophages
- DMSO:
-
Dimethylsulfoxide
- MTT:
-
3-[4,5-dimethylthiazol-2yl]-2,5-diphenyl tetrazolium bromide
- PI:
-
Propidium iodide
- CPP32:
-
32 kDa cysteine protease
- LDH:
-
Lactate dehydrogenase
- SDS–PAGE:
-
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
- MDM2:
-
Mouse double min clone 2
- PARP:
-
Poly (ADP-ribose) polymerase
- TEM:
-
Transmission electron microscopy
- PBS:
-
Phosphate-buffered saline
- CD:
-
Circular dichroism spectroscopy
- BAF:
-
BOC-Asp(OMe)-FMK caspase inhibitor
References
Berlose JP, Convert O, Derossi D, Brunissen A, and Chassaing G . (1996). Eur. J. Biochem., 242, 372–386.
Bottger A, Bottger V, Sparks A, Liu WL, Howard SF, and Lane DP . (1997). Curr. Biol., 7, 860–869.
Brugidou J, Legrand C, Mery J, and Rabie A . (1995). Biochem. Biophys. Res. Commun., 214, 685–693.
Couet J, Li S, Okamoto T, Ikezu T, and Lisanti MP . (1997). J. Biol. Chem., 272, 6525–6533.
De Kroon AIPM, De Gier J, and De Kruijff B . (1993). Lipid–Peptide Interactions in Model Systems-Membrane Insertion and Translocation of Peptides: Protein–Lipid Interactions. Elsevier: Amsterdam.
Delaconstantinos G . (1987). Anticancer Res., 7, 1011–1021.
Derossi D, Chassaing G, and Prochiantz A . (1998). Trends Cell Biol., 8, 84–87.
Derossi D, Calvet S, Trembleau A, Brunissen A, Chassaing G, and Prochiantz A . (1996). J. Biol. Chem., 271, 18188–18193.
Eisenberg D, Weiss RM, and Terwilliger TC . (1984). Proc. Natl. Acad. Sci., 81, 140–144.
Fahraeus R, Fischer P, Krausz E, and Lane DP . (1999). J. Pathol., 187, 138–146.
Fine RL, Chambers TC, and Sachs CW . (1996). Oncologist, 1, 261–268.
Foster BA, Coffey HA, Morin MJ, and Rastinejad F . (1999). Science, 286, 2507–2510.
Garnier J,G. J-F, and Robson B . (1996). Methods in Enzymology. Doolittle, R (ed). Academic Press: New York, pp. 540–553.
Harris CC . (1996). J. Natl. Cancer Inst., 88, 1442–1455.
Kanovsky M, Raffo A, Drew L, Rosal R, Do T, Friedman FK, Rubinstein P, Visser J, Brandt-Rauf PW, Fine R, Michl J, and Pincus MR . (2001). Proc. Natl. Acad. Sci., 98, 12438–12443.
Kim AL, Raffo AJ, Brandt-Rauf PW, Pincus MP, Monaco R, Abarzua P, and Fine RL . (1999). J. Biol. Chem., 274, 34924–34931.
Kussie PH, Gorina S, Marechal V, Elenbaas B, Moreau J, Levine AJ, and Pavletich NP . (1996). Science, 274, 948–953.
Levine AJ, Perry ME, Chang A, Silver A, Dihmer D, Wu M, and Welsh D . (1994). Br. J. Cancer, 69, 409–416.
Levine AJ . (1997). Cell, 88, 323–331.
Lofts FJ, Hurst HC, Sternberg MJE, and Gullick WJ . (1993). Oncogene, 8, 2813–2820.
Midgley CA, Desterro JM, Saville MK, Howard S, Sparks A, Hay RT, and Lane DP . (2000). Oncogene, 19, 2312–2323.
Momand J, Zambetti GP, Olson DC, George D, and Levine AJ . (1992). Cell, 69, 1237–1245.
Ozbun MA and Butel JS . (1995). Adv. Cancer Res., 66, 71–141.
Perczel A, Park K, and Fasman GD . (1991). Anal Biochem, 4, 669–679
Prives C and Hall PA . (1999). J. Pathol., 187, 112–126.
Prochiantz A . (1999). Ann. N. Y. Acad. Sci., 886, 172–179.
Rousselle C, Clair P, Lefauconnier JM, Kaczorek M, Scherrmann JM, and Temsamani J . (2000). Mol. Pharmacol., 57, 679–686
Schwarze SR and Dowdy SF . (2000). Trends Pharmacol. Sci., 21, 45–48.
Schwarze SR, Hruska KA, and Dowdy SF . (2000). Trends Cell Biol., 10, 290–295.
Segrest JP, De Loof H, Dohlman JG, Brouillette CG, and Anantharamaiah GM . (1990). Proteins, 8, 103–117.
Selivanova G, Ryabchenko L, Jansson E, Iotsova V, and Wiman KG . (1999). Mol. Cell Biol., 19, 3395–3402.
Selivanova G, Iotsova V, Okan I, Fritsche M, Strom M, Groner B, Grafstrom RC, and Wiman KG . (1997). Nat. Med., 3, 632–638.
Sherbert GV . (1989). Exp. Cell Biol., 57, 198–205.
Soussi T . (2000a). Bull. Cancer, 87, 689–690.
Soussi T . (2000b). Ann. N. Y. Acad. Sci., 910, 121–137.
Soussi T, Caron de Fromentel C, and May P . (1990). Oncogene, 5, 945–952.
Spector AA and Burns CD . (1987). Cancer Res., 47, 4529–4537.
Sreerama N and Woody RW (1993). Anal. Biochem., 209, 32–44.
Velculescu VE and El-Deiry WS . (1996). Clin. Chem., 42, 858–868.
Wang JL, Zhang ZJ, Choksi S, Shan S, Lu Z, Croce CM, Alnemri ES, Korngold R, and Huang Z . (2000). Cancer Res., 60, 1498–1502.
Wasylyk C, Salvi R, Argentini M, Dureuil C, Delumeau I, Abecassis J, Debussche L, and Wasylyk B . (1999). Oncogene., 18, 1921–1934.
Yeung TK, Germond C, Chen X, and Wang Z . (1999). Biochem. Biophys. Res. Commun., 263, 398–404.
Zhang R and Wang H . (2000). Curr. Pharmaceut. Des., 6, 393–416.
Acknowledgements
This work was supported in part by the National Institutes of Health RO1 Grant OH 04192 and Environmental Protection Agency Grant R826685 (to PWB-R); NIH RO1 Grant CA 82528 and a Herbert Irving Scholar Award (to RLF); and NIH RO1 Grant CA 42500, a Veteran's Affairs Merit Review Grant, and a grant from the Lustgarten Foundation for Pancreatic Cancer Research (to MRP). We thank Dr Yin Li for helpful discussion.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Do, T., Rosal, R., Drew, L. et al. Preferential induction of necrosis in human breast cancer cells by a p53 peptide derived from the MDM2 binding site. Oncogene 22, 1431–1444 (2003). https://doi.org/10.1038/sj.onc.1206258
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206258
Keywords
This article is cited by
-
A combination of a cell penetrating peptide and a protein translation inhibitor kills metastatic breast cancer cells
Cell Death Discovery (2023)
-
The dual interaction of antimicrobial peptides on bacteria and cancer cells; mechanism of action and therapeutic strategies of nanostructures
Microbial Cell Factories (2022)
-
Antitumorigenic effect of insect-derived peptide poecilocorisin-1 in human skin cancer cells through regulation of Sp1 transcription factor
Scientific Reports (2021)
-
Evaluation of the use of therapeutic peptides for cancer treatment
Journal of Biomedical Science (2017)
-
Production of Cell-Penetrating Peptides in Escherichia coli Using an Intein-Mediated System
Applied Biochemistry and Biotechnology (2015)