Abstract
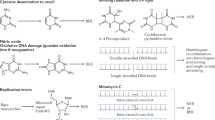
Nucleotide excision repair provides an important cellular defense against a large variety of structurally unrelated DNA alterations. Most of these alterations, if unrepaired, may contribute to mutagenesis, oncogenesis, and developmental abnormalities, as well as cellular lethality. There are two subpathways of nucleotide excision repair; global genomic repair (GGR) and transcription coupled repair (TCR), that is selective for the transcribed DNA strand in expressed genes. Some of the proteins involved in the recognition of DNA damage (including RNA polymerase) are also responsive to natural variations in the secondary structural features of DNA. Gratuitous repair events in undamaged DNA might then contribute to genomic instability. However, damage recognition enzymes for GGR are normally maintained at very low levels unless the cells are genomically stressed. GGR is controlled through the SOS stress response in E. coli and through the activated p53 tumor suppressor in human cells. These inducible responses in human cells are important, as they have been shown to operate upon chemical carcinogen DNA damage at levels to which humans are environmentally exposed. Interestingly, most rodent tissues are deficient in the p53-dependent GGR pathway. Since rodents are used as surrogates for environmental cancer risk assessment, it is essential that we understand how they differ from humans with respect to DNA repair and oncogenic responses to environmental genotoxins. In the case of terminally differentiated mammalian cells, a new paradigm has appeared in which GGR is attenuated but both strands of expressed genes are repaired efficiently.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Adimoolam S, Ford JM . 2002 Proc. Natl. Acad. Sci. USA 99: 12985–12990
Batty D, Rapic'-Otrin V, Levine AS, Wood RD . 2000 J. Mol. Biol. 300: 275–290
Beach AC, Gupta RC . 1992 Carcinogenesis 13: 1053–1074
Ben-Ishai R, Peleg L . 1975 Molecular Mechanisms for Repair of DNA Hanawalt P and Setlow R (eds) New York: Plenum Press pp. 607–610
Birrell GW, Brown JA, Wu HI, Giaever G, Chu AM, Davis RW, Brown JM . 2002 Proc. Natl. Acad. Sci. USA 99: 8778–8783
Bohr VA, Smith CA, Okumoto DS, Hanawalt PC . 1985 Cell 40: 359–369
Bowman K, Sicard D, Ford J, Hanawalt PC . 2000 Mol. Carcinogenesis 29: 17–24
Branum ME, Reardon JT, Sancar A . 2001 J. Biol. Chem. 276: 25421–25426
Couch JL, Hanawalt PC . 1967 Biochem. Biophys. Res. Commun. 26: 779–784
Courcelle J, Khodursky A, Peter B, Brown PO, Hanawalt PC . 2001 Genetics 158: 41–64
Crowley DJ, Hanawalt PC . 1998 J. Bacteriol. 180: 3345–3352
Crowley DJ, Hanawalt PC . 2001 Mutat. Res. 485: 319–329
Dumaz N, van Kranen H, de Vries A, Berg R, Wester P, van Kreijl C, Sarasin A, Daya-Grosjean L, de Gruijl F . 1997 Carcinogenesis 18: 897–904
Dunkern TR, Kaina B . 2002 Mol. Biol. Cell 13: 348–361
Eisen J, Hanawalt PC . 1999 Mut. Res. DNA Repair 435: 171–213
Ford JM, Hanawalt PC . 1995 Proc. Natl. Acad. Sci. USA 92: 8876–8880
Ford JM, Hanawalt PC . 1997 J. Biol. Chem. 272: 28073–28080
Ford JM, Baron EL, Hanawalt PC . 1998 Cancer Res. 58: 599–603
Ganesan AK, Spivak G, Hanawalt PC . 1983 Manipulation and Expression of Genes in Eukaryotes Nagley P, Linnane AW, Peacock WJ, Pateman JA (eds) Australia: Academic Press pp. 45–54
Grivell AR, Grivell MB, Hanawalt PC . 1975 J. Mol. Biol. 98: 219–233
Hanawalt PC, Crowley DJ, Ford JM, Ganesan AK, Lloyd DR, Nouspikel T, Smith CA, Spivak G, Tornaletti S . 2001 Cold Spring Harbor Symp. Quant. Biol. 65: 183–191
Hanawalt PC, Pettijohn DE, Pauling EC, Brunk CF, Smith DW, Kanner LC, Couch JL . 1968 Cold Spring Harb. Symp. Quant. Biol. 33: 187–194
Hanawalt PC, Bohr VA, Leadon SA, Mansbridge JN, Reush MKH . 1987 Processes in Cutaneous Epidermal Differentiation Bernstein I and Hirone T (eds) New York: Praeger Scientific pp. 217–231
Hanawalt PC . 1994 Science 266: 1957–1958
Hanawalt PC . 2001 Environ. Mol. Mut. 38: 89–96
Hwang BJ, Ford JM, Hanawalt PC, Chu G . 1999 Proc. Natl. Acad. Sci. USA 96: 424–428
Hwang BJ, Toering S, Franoeke U, Chu G . 1998 Mol. Cell Biol. 18: 4391–4399
Klimek K . 1965 Neoplasma 12: 459–460
Leadon SA, Cooper PK . 1993 Proc. Natl. Acad. Sci. USA 90: 10499–10503
Lindahl T, Wood RD . 1999 Science 286: 1897–1905
Ljungman M, Zhang F . 1996 Oncogene 13: 823–831
Lloyd D, Hanawalt PC . 2000 Cancer Res. 60: 517–521
Lohman PHM, Paterson MC, Zelle B, Reynolds RJ . 1976 Mut. Res. 46: 138–139
McKay BC, Ljungman M . 1999 Neoplasia 1: 276–284
Mellon I, Hanawalt PC . 1989 Nature 342: 95–98
Mellon I, Spivak G, Hanawalt PC . 1987 Cell 51: 241–249
Moolenaar GF, van Rossum-Fikkert S, van Kestern M, Goosen N . 2002 Proc. Natl. Acad. Sci. USA 99: 1467–1472
Mullaart E, Lohman PHM, Vijg J . 1988 J. Invest. Dermatol. 90: 346–349
Nouspikel T, Hanawalt PC . 2000 Mol. Cell Biol. 20: 1562–1570
Nouspikel T, Hanawalt PC . 2002 DNA Repair 1: 59–75
Nouspikel T, Hanawalt PC . 2003 Bioessays (in press)
Painter RB, Cleaver JS . 1969 Radiat. Res. 37: 451–466
Pauling C, Hanawalt P . 1965 Proc. Natl. Acad. Sci. USA 54: 1728–1735
Peleg L, Raz E, Ben-Ishai R . 1976 Exp. Cell Res. 104: 301–307
Petit C, Sancar A . 1999 Biochime. 81: 15–25
Pettijohn D, Hanawalt PC . 1964 J. Mol. Biol. 9: 395–410
Plosky B, Samson L, Engelward BP, Gold B, Schlaen B, Millas T, Magnotti M, Schor J, Scicchitano DA . 2002 DNA Repair 1: 683–696
Prost S, Ford JM, Taylor C, Doig J, Harrison DJ . 1998 J. Biol. Chem. 273: 33327–33332
Sancar A . 1996 Annu. Rev. Biochem. 65: 43–81
Selby CP, Sancar A . 1993 Science 260: 53–58
Smith ML, Ford JM, Hollander MC, Bortnick RA, Amundson SA, Seo YR, Deng C-X, Hanawalt PC, Fornace AJ . 2000 Mol. Cell. Biol. 20: 3705–3714
Spivak G, Itoh T, Matsunaga T, Nikaido O, Hanawalt P, Yamaizumi M . 2002 DNA Repair 50: 1–15
Sugasawa K, Ng JMY, Masutani C, Iwai S, van der Spek PJ, Eker APM, Hanaoka F, Bootsma D, Hoeijmakers JHJ . 1998 Mol. Cell 2: 223–232
Tang J, Chu G . 2002 DNA Repair 1: 601–616
Tang J, Huang BJ, Ford J, Hanawalt PC, Chu G . 2000 Mol. Cell 5: 737–744
Therrien JP, Drouin R, Baril C, Drobetsky EA . 1999 Proc. Natl. Acad. Sci. USA 96: 15030–15043
Trosko JE, Chu EHY, Carrier WL . 1965 Radiat. Res. 24: 667–672
van Houten B, Eisen JA, Hanawalt PC . 2002 Proc. Natl. Acad. Sci. USA 99: 2581–2583
van Zeeland AA, Smith CA, Hanawalt PC . 1981 Mutat. Res. 82: 173–189
Vijg J, Mullaart E, van der Schans GP, Lohman PHM, Knook DL . 1984 Mutat. Res. 132: 129–138
Wagasugi M, Kawashima A, Morioka H, Linn S, Sancar A, Mori T, Nikaido O, Matsunaga T . 2002 J. Biol. Chem. 277: 1637–1640
Wani MA, Zhu Q, El-Mahdy M, Venkatachalam S, Wani AA . 2000 Cancer Res. 60: 2273–2280
Zou Y, van Houten B . 1999 EMBO J. 18: 4889–4901
Zou Y, Luo C, Geacintov E . 2001 Biochemistry 40: 2923–2931
Acknowledgements
The research in my laboratory has been supported by an Outstanding Investigator Grant (CA 44349) and more recently by grants (CA77712, CA91456 and CA90915) from the National Cancer Institute. I am indebted to my students and colleagues, many of whom are cited in the references, for their remarkable contributions to the ideas as well as the experimental results presented in this review. I wish to thank Denise Flowers for her essential help with the manuscript preparation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hanawalt, P. Subpathways of nucleotide excision repair and their regulation. Oncogene 21, 8949–8956 (2002). https://doi.org/10.1038/sj.onc.1206096
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206096
Keywords
This article is cited by
-
Genetic aberrations in DNA repair pathways: a cornerstone of precision oncology in prostate cancer
British Journal of Cancer (2021)
-
Single-molecule imaging reveals molecular coupling between transcription and DNA repair machinery in live cells
Nature Communications (2020)
-
Tissue-infiltrating macrophages mediate an exosome-based metabolic reprogramming upon DNA damage
Nature Communications (2020)
-
Lineage-specific control of TFIIH by MITF determines transcriptional homeostasis and DNA repair
Oncogene (2019)
-
The R156R ERCC2 polymorphism as a risk factor of endometrial cancer
Tumor Biology (2016)