Abstract
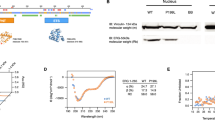
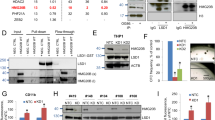
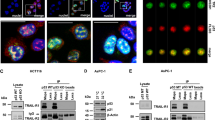
The promyelocytic leukemia protein PML is a tumor and growth suppressor and plays an important role in a multiple pathways of apoptosis and regulation of cell cycle progression. Our previous studies and others also documented a role of PML in transcriptional regulation through its association with transcription coactivator CBP and transcription corepressor HDAC. Here, we showed that PML is a potent transcriptional repressor of Nur77, an orphan receptor and a member of the steroid receptor superfamily of proteins. We found that PML represses Nur77-mediated transactivation through a physical and functional interaction between the two proteins. PML interacts with Nur-77 in vitro in a GST-pull down assay and in vivo by coimmunoprecipitation assay. PML/Nur77 colocalized in vivo in a double immunofluorescent staining and confocal microscopic analysis. Our study further showed that the coiled–coil domain of PML interacts with the DNA-binding domain of Nur77 (amino acids 267–332). Electrophoretic mobility shift assay demonstrated that PML interferes with Nur77 DNA binding in a dose-dependent manner. This study indicates that PML interacts with the DNA-binding domain of Nur77 and represses transcription by preventing it from binding to the target promoter. This study supports a role of PML/Nur77 interaction in regulating cell growth and apoptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Amsen D, Calvo CR, Osborne BA, Kruisbeek AM . 1999 Proc. Natl. Acad. Sci. USA 96: 622–627
Boisvert F-M, Hendzel MJ, Bazett-Jone DP . 2000 J. Cell Biol. 148: 282–292
Boisvert F-M, Kruhlak MJ, Box AK, Hendzel MJ, Bazett-Jones DP . 2001 J. Cell Biol. 152: 1099–1106
Calnan BJ, Szychowski S, Chan KM, Cado D, Winoto A . 1995 Immunity 3: 273–282
Doucas V, Evans RM . 1999 Proc. Natl. Acad. Sci. USA 96: 2633–2638
Doucas V, Tini M, Egan DA, Evans RM . 1999 Proc. Natl. Acad. Sci. USA 96: 2627–2632
Duprez E, Saurin AJ, Desterro JM, Lallemand-Breitenbach V, Howe K, Boddy MN, Solomon E, de The H, Hay RT, Freemont PS . 1999 J. Cell Sci. 112: 381–393
Enmark E, Gustafssan JA . 1996 Mol. Endocrinol. 10: 1293–1307
Everett RD . 2001 Oncogene 20: 7266–7273
Ferbeyre G, Stanchina E, Querido E, Baptiste N, Prives C, Lowe SW . 2000 Genes Dev. 14: 2015–2027
Fogal V, Gostissa M, Sandy P, Zacchi P, Sternsdorf T, Jensen K, Pandolfi PP, Will H, Schneider C, DelSal G . 2000 EMBO J. 22: 6186–6195
Guo A, Salomoni P, Luo J, Shih A, Zhong S, Gu W, Pandolfi PP . 2000 Nat. Cell Biol. 2: 730–736
Hazel TG, Nathans D, Lau LF . 1988 Proc. Natl. Acad. Sci. USA 85: 8444–8449
Herschman HR . 1991 Ann. Rev. Biochem. 60: 281–319
Ishov AM, Sotnikov AG, Negorev D, Vladimirova OV, Neff N, Kamitani T, Yeh ET, Strauss II JF, Maul GG . 1999 J. Cell Biol. 147: 221–234
Kastner P, Perez A, Lutz Y, Rochette E, Gaube MP, Durand B, Lanotte M, Berger R, Chambon P . 1992 EMBO J. 11: 629–642
Kamitani T, Kito K, Nyuyen HP, Wada H, Fukuda-Kamitani T, Yeh ET . 1998 J. Biol. Chem. 273: 26675–26682
Khan MM, Nomura T, Kim H, Kaul SC, Wadhwa R, Shinagawa T, Ichikawa-Iwata E, Zhong S, Pandolfi PP, Ishii S . 2001 Mol. Cell 7: 1233–1243
LaMorte VJ, Dyck JA, Ochs RL, Evans RM . 1998 Proc. Natl. Acad. Sci. USA 95: 4991–4996
Le XF, Yang P, Chang KS . 1996 J. Biol. Chem. 271: 130–135
Le XF, Vallian S, Mu ZM, Hung MC, Chang KS . 1998 Oncogene 16: 1839–1849
Li H, Kolluri KS, Gu J, Dawson MI, Cao X, Hobbs PD, Lin B, Chen G, Lu J-S, Lin F, Xie Z, Fontana JA, Reed JC, Zhang X-K . 2000a Science 289: 1159–1164
Li H, Leo C, Zhu J, Wu XY, O'Neil J, Park EJ, Chen JD . 2000b Mol. Cell. Biol. 20: 1784–1796
Lin RJ, Sternsdorf T, Tini M, Evans RM . 2001 Oncogene 20: 7204–7215
Liu JH, Mu ZM, Chang KS . 1995 J. Exp. Med. 181: 1965–1973
Liu ZG, Smith SW, McLaughlin KA, Schwartz LM, Osborne BA . 1994 Nature 367: 281–284
Maul GG . 1998 Bioessays 20: 660–667
Melnick A, Licht JD . 1999 Blood 93: 3167–3215
Milbrandt J . 1988 Neuron 1: 183–188
Mirzoeva OK, Petrini JHJ . 2001 Mol. Cell. Biol. 21: 281–288
Mu ZM, Chin KV, Liu JH, Lozano G, Chang KS . 1994 Mol. Cell. Biol. 14: 6858–6867
Mu ZM, Le XF, Vallian S, Glassman AB, Chang KS . 1997 Carcinogenesis 18: 2063–2069
Nakai A, Kartha S, Sakurai A, Toback FG, DeGroot LJ . 1990 Mol. Endocrinol. 4: 1438–1443
Pearson M, Carbone R, Sebastiani C, Cioce M, Fagioli M, Saito S, Higashimoto Y, Appella E, Minucci S, Pandolfi PP, Pelicci PG . 2000 Nature 406: 207–210
Quignon F, De Bels F, Koken M, Feunteun J, Ameisen JC, de The H . 1998 Nature Genet. 20: 259–265
Seeler J-S, Dejean A . 2001 Oncogene 20: 7243–7249
Tsuzuki S, Towatari M, Saito H, Enver T . 2000 Mol. Cell. Biol. 20: 6276–6286
Uemura H, Chang C . 1998 Endocrinology 139: 2329–2334
Vallian S, Gaken AJ, Gingold EB, Kouzarides T, Chang KS, Farzaneh F . 1998a Oncogene 90: 3265–3269
Vallian S, Gaken JA, Trayner ID, Gingold EB, Kourizarides T, Chang KS, Farzaneh F . 1997 Exp. Cell. Res. 237: 371–382
Vallian S, Chin KV, Chang KS . 1998b Mol. Cell. Biol. 18: 7147–5156
Wang ZG, Delva L, Gaboli M, Rivi R, Giorgio M, Gordon-Cardo C, Grosweld F, Pandolfi PP . 1998 Science 279: 1547–1551
Weih F, Ryseck RP, Chen L, Brovo R . 1996 Proc. Natl. Acad. Sci. USA 93: 5533–5538
Wilson TE, Mouw AR, Weaver CA, Milbrandt J, Parker KL . 1993 Mol. Cell. Biol. 13: 861–868
Woronicz JD, Cainan B, Ngo V, Winoto A . 1994 Nature 367: 227–281
Wu WS, Vallian S, Seto E, Yang WM, Edmondson D, Roth S, Chang KS . 2001 Mol. Cell. Biol. 21: 2259–2268
Zhong S, Delva L, Tachez C, Cenciarelli C, Gandini D, Zhang H, Kalantry S, Freedman LP, Pandolfi PP . 1999 Nat. Genet. 23: 287–295
Zhong S, Muller S, Ronchetti S, Freemont PS, Dejean A, Pandolfi PP . 2000 Blood 95: 2748–2752
Acknowledgements
DNA sequencing facility and Confocal microscopic analysis were supported by a research grant CA-16627 from the National Cancer Institute. This study was supported by a grant from the National Institute of Health CA-55577 to KS Chang.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, WS., Xu, ZX., Ran, R. et al. Promyelocytic leukemia protein PML inhibits Nur77-mediated transcription through specific functional interactions. Oncogene 21, 3925–3933 (2002). https://doi.org/10.1038/sj.onc.1205491
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205491
Keywords
This article is cited by
-
The function, regulation and therapeutic implications of the tumor suppressor protein, PML
Cell & Bioscience (2015)
-
Role of the nucleus in apoptosis: signaling and execution
Cellular and Molecular Life Sciences (2015)
-
PML-mediated signaling and its role in cancer stem cells
Oncogene (2014)
-
Selective activation of NFAT by promyelocytic leukemia protein
Oncogene (2008)
-
p53 and Nur77/TR3 – transcription factors that directly target mitochondria for cell death induction
Oncogene (2006)