Abstract
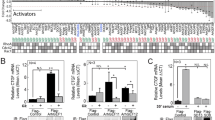
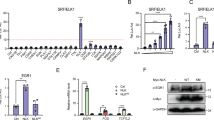
Serum Response Factor (SRF) plays a central role in the transcriptional response of mammalian cells to a variety of extracellular signals. It is a key regulator of many cellular early response genes which are believed to be involved in cell growth, differentiation, and development. The mechanism by which SRF activates transcription in response to mitogenic agents has been extensively studied, however, less is known about regulation of the SRF gene itself. Previously, we identified distinct regulatory elements in the SRF promoter that play a role in activation, including an ETS domain binding site, an overlapping Sp1/Egr-1 binding site, and two SRF binding sites. We further showed that serum induces the SRF gene by a mechanism that requires an intact SRF binding site, also termed a CArG box. In the present study we demonstrate that in response to stimulation by cells by lysophosphatidic acid (LPA) or whole serum, the SRF promoter is upregulated by a bipartite pathway that requires both an Sp1 factor binding site and the CArG motifs for maximal stimulation. The CArG box-dependent component of this pathway is targeted by Rho mediated signals, and the Sp1 binding site dependent component is targeted by Ras mediated signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Arsenian S, Weinhold B, Oelgeschlager M, Ruther U and Nordheim A. . 1998 EMBO J. 17: 6289–6299.
Avantaggiati ML, Natoli G, Balsano C, Chirillo P, Artini M, DeMarzio E, Collepardo D and Levrero M. . 1993 Oncogene 8: 1567–1574.
Bading H, Ginty DD and Greenberg ME. . 1993 Science 260: 181–186.
Belaguli NS, Schildmeyer LA and Schwartz RJ. . 1997 J. Biol. Chem. 272: 18222–18231.
Changelian PS, Feng P, King TC and Milbrandt J. . 1989 Proc. Natl. Acad. Sci. USA 86: 377–381.
Chavrier P, Janssen-Timmen U, Mattei M-G, Zerial M, Bravo R and Charnay P. . 1989 Mol. Cell. Biol. 9: 787–797.
Chen BK, Liu YW, Yamamoto S and Chang WC. . 1997 Biochim. Biophys. Acta 1344: 270–277.
Christy B and Nathans D. . 1989 Mol. Cell. Biol. 9: 4889–4895.
Coso OA, Chiariello M, Yu J-C, Teramoto H, Crespo P, Xu N, Miki T and Gutkind JS. . 1995 Cell 81: 1137–1146.
Croissant JD, Kim JH, Eichele G, Goering L, Lough J, Prywes R and Schwartz RJ. . 1996 Dev. Biol. 177: 250–264.
Fromm C, Coso OA, Montaner S, Xu N and Gutkind JS. . 1997 Proc. Natl. Acad. Sci. USA 94: 10098–10103.
Fujii M, Tsuchiya H, Chuhjo T, Akizawa T and Seiki M. . 1992 Genes Dev. 6: 2066–2067.
Fujii M, Tsuchiya H, Chujo T, Minamino T, Miyamoto K and Seiki M. . 1994 J. Virol. 68: 7275–7283.
Gauthier-Rouviere C, Cavadore J-C, Blanchard J-M, Lamb NJC and Fernandez A. . 1991 Cell Reg. 2: 575–588.
Hanlon M and Sealy L. . 1999 J. Biol. Chem. 274: 14224–14228.
Hill CS, Wynne J and Treisman R. . 1994 EMBO J. 13: 5421–5432.
Hill CS, Wynne J and Treisman R. . 1995 Cell 81: 1159–1170.
Hipskind RA, Buscher D, Nordheim A and Baccarini M. . 1994 Genes Dev. 8: 1803–1816.
Janknecht R, Ernst W, Pingoud V and Nordheim A. . 1993 EMBO J. 12: 5097–5104.
Khachigian LM, Williams AJ and Collins T. . 1995 J. Biol. Chem. 270: 27679–27686.
Latinkic B, O'Brien T and Lau L. . 1991 Nucleic Acids Res. 19: 3261–3267.
Latinkic BV and Lau LF. . 1994 J. Biol. Chem. 269: 23163–23170.
McDonough PM, Hanford DS, Sprenkle AB, Mellon NR and Glembotski CC. . 1997 J. Biol. Chem. 272: 24046–24053.
Misra R, Bonni A, Miranti CK, Rivera VM, Sheng M and Greenberg ME. . 1994 J. Biol. Chem. 269: 25483–25493.
Misra RP, Rivera VM, Wang JM, Fan P-D and Greenberg ME. . 1991 Mol. Cell Biol. 11: 4545–4554.
Moolenar WH. . 1995 J. Biochem. 270: 12949–12952.
Moolenar WH, van der Bend RL, van Corven EJ, Jalink K, Eichholtz T and van Blitterswijk WJ. . 1992 Cold Spring Harbor Symposia on Quantitative Biology 57: 163–167.
Norman C, Runswick M, Pollock R and Treisman R. . 1988 Cell 55: 989–1003.
Ridley AJ and Hall A. . 1992 Cell 70: 389–399.
Rivera VM, Miranti CK, Misra RP, Ginty DD, Chen R-H, Blenis J and Greenberg ME. . 1993 Mol. Cell. Biol. 13: 6260–6273.
Sheng M, Dougan ST, McFadden G and Greenberg ME. . 1988 Mol. Cell Biol. 8: 2787–2796.
Spencer JA, Major ML and Misra RP. . 1999 Mol. Cell Biol. 19: 3977–3988.
Spencer JA and Misra RP. . 1996 J. Biol. Chem. 271: 16535–16543.
Treisman R. . 1985 Cell 42: 889–902.
Treisman R. . 1990 Cancer Biol. 1: 47–58.
Treisman R and Ammerer G. . 1992 Curr. Opin. Genet. Dev. 2: 221–226.
Tsai-Morris C-H, Cao X and Sukhatme VP. . 1988 Nucleic Acids Res. 16: 8835–8846.
Vandromme M, Gauthier-Rouviere C, Gilles C, Lamb N and Fernandez A. . 1992 J. Cell Biol. 118: 1489–1500.
Visvader J, Sassone-Corsi P and Verma I. . 1988 Proc. Natl. Acad. Sci. USA 85: 9474–9478.
Acknowledgements
We thank Vaughn Jackson for critical reading of the manuscript. We thank J Silvio Gutkind for his kind gift of Rho-family inhibitory plasmids, and Robert Tjian for the Gal4 – Sp1 expression plasmid. This work was supported in part by the following to RP Misra: an American Cancer Society Institutional Seed Grant Award from the Medical College of Wisconsin Cancer Center; a Grant-in-Aid from the American Heart Association, Wisconsin Division; a Shannon Award (R55 GM/OD51856) and a FIRST Award (R29 NS36256) from the National Institutes of Health.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Spencer, J., Misra, R. Expression of the SRF gene occurs through a Ras/Sp/SRF-mediated-mechanism in response to serum growth signals. Oncogene 18, 7319–7327 (1999). https://doi.org/10.1038/sj.onc.1203121
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1203121
Keywords
This article is cited by
-
Actomyosin and the MRTF-SRF pathway downregulate FGFR1 in mesenchymal stromal cells
Communications Biology (2020)
-
HOPX functions as a tumour suppressor in head and neck cancer
Scientific Reports (2016)
-
MKL1/2 and ELK4 co-regulate distinct serum response factor (SRF) transcription programs in macrophages
BMC Genomics (2014)
-
GnRH pulse frequency differentially regulates steroidogenic factor 1 (SF1), dosage-sensitive sex reversal-AHC critical region on the X chromosome gene 1 (DAX1), and serum response factor (SRF): potential mechanism for GnRH pulse frequency regulation of LH beta transcription in the rat
Endocrine (2011)
-
Comparative promoter region analysis powered by CORG
BMC Genomics (2005)