Abstract
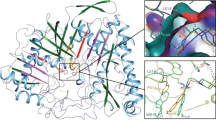
A lead compound obtained from a high volume human non-pancreatic secretory phospholipase A2 (hnps-PLA2) screen has been developed into a potent inhibitor using detailed structural knowledge of inhibitor binding to the enzyme active site. Four crystal structures of hnps-PLA2 complexed with a series of increasingly potent indole inhibitors were determined and used as the structural basis for both understanding this binding and providing valuable insights for further development. The application of structure-based drug design has made possible improvements in the binding of this screening lead to the enzyme by nearly three orders of magnitude. Furthermore, the optimized structure (LY311727) displayed 1,500-fold selectivity when assayed against porcine pancreatic s-PLA2.
Similar content being viewed by others
Article PDF
References
Green, J.-L. et al. Circulating phospholipase A2 activity associated with sepsis and septic shock is indistinguishable from that associated with rheumatoid arthritis. Inflammation 15, 355–367 (1991).
Kramer, R.M. et al. Structure & properties of a human non-pancreatic phospholipase A2 . J.biol. Chem 264, 5768–5775 (1989).
Dennis, E.A., Rhee, S.G., Billah, M.M. & Hannun, Y.A. Role of phospholipases in generating lipid second messengers in signal transduction. FASEB J. 5, 2068–2077 (1991).
Pruzanski, W., Vadas, P. & Browing, J. Secretory non-pancreatic group II phospholipase A2: role in physiologic and inflammatory processes. J. lipid Med. 8, 161–167 (1993).
Santos, A.A. et al. Are events after endotoxemia related to circulating phospholipase A2? Ann. Surg. 219, 183–192 (1994).
Vadas, P. & Pruzanski, W. Induction of group II phospholipase A2 expression and pathogenesis of the sepsis syndrome. Circ. Shock 39, 160–167 (1993).
Baldwin, J.J. et al. Thynopyran-2-sulfonamides: novel topically active carbonic anhydrase inhibitors for the treatment of glaucoma. J. med. Chem. 32, 2510–2513 (1989).
Shoichet, B.K., Stroud, R.M., Santi, D.V., Kuntz, I.D. & Perry, K.M. Structure-based discovery of inhibitors of thymidylate synthase. Science 259, 1445–1450 (1993).
Erickson, J. et al. Design, activity, and 2.8 Å crystal structure of a C2 symmetric inhibitor complexed to HIV-1 protease. Science 249, 527–533 (1990).
Lam, P.Y.S. et al. Rational design of potent, bioavailable, nonpeptide cyclic ureas as HIV protease inhibitors. Science 263, 380–384 (1994).
Ealick, S.E. et al. Application of crystallographic and modelling methods in the design of purine nucleoside phosphorylase inhibitors. Proc. Natn. Acad. Sci U.S.A. 88, 11540–111544 (1991).
von Itzstein, M. et al. Rational design of potent sialidase-based inhibitors of influenza virus replication. Nature 363, 418–423 (1993).
Warner, P., Green, R.C., Gomes, B. & Strimpler, A.M. Non-peptide inhibitors of human leukocyte elastase. 1. The design and synthesis of pyridone-containing inhibitors. J. med. Chem. 37, 3090–3099 (1994).
Beaton, H.G. et al. Discovery of new non-phospholipid inhibitors of the secretory phospholipases A2. J. med. Chem. 37, 557–559 (1994).
Jain, M.K. et al. Fatty acid amides: scooting mode-based discovery of tight-binding competitive inhibitors of secreted phospholipases A2. J. med. Chem. 35, 3584–3586 (1992).
Pisabarro, M.T. et al. Rational modification of human synovial fluid phospholipase A2 inhibitors. J med. Chem. 37, 337–341 (1994).
Wery, J.-P. et al. Structure of recombinant human rheumatoid arthritic synovial fluid phopholipase A2 at 2.2 Å resolution. Nature 352, 79–82 (1991).
Scott, D.L. et al. Structures of free and inhibited human secretory phospholipase A2 from inflammatory exudate. Science 254, 1007–1010 (1991).
White, S.P., Scott, D.L., Otwinski, Z., Gelb, M.H., & Sigler, P.B., Crystal structure of cobra-venom phospholipase 'A2 in a complex with a transition-state analogue. Science 250, 1560–1563 (1990).
Scott, D.L., Otwinowski, Z., Gelb, M.H., & Sigler, P.B. Crystal structure of bee venom phospholpase A2 in a complex with a transition-state analogue. Science 250, 1563–1566 (1990).
Thunnissen, M.M.G.M. et al. X-ray structure of phospholipase A2 complexed with a substrate-derived inhibitor. Nature 347, 689–691 (1990).
Kaplan, L., Weiss, J., & Elsback, P. Low concentrations ofindomethacin inhibit phospholipase A2 of rabbit polymorphonuclear leukocytes. Proc. natn. Acad. Sci. U.S.A. 75, 2955–2958 (1978).
Lobo, I.B., & Hoult, J.R.S. Groups I,II and III extracellular phospholipases A2: Selective inhibition of group II enzymes by indomethacin but not other NSAIDs. Agents Actions 41, 111–113 (1994).
Reynolds, L.J., Hughes, L.L., & Dennis, E.A. Analysis of human synovial fluid phospholipase A2 on short chain phosphatidylcholine-mixed micelles: development of a spectrophotomertic assay suitable for a microtiterplate reader. Analyt. Biochem. 204, 190–197 (1992).
Snyder, D.W., Sommers, C.D., Bobbitt, J.L. & Mihelich, E.D. Characterization of the contractile effects of human recombinant non-pancreatic secretory phopholipase A2 and other PLA2s on guinea pig lung pleural strips. J. Pharmac. exp. Ther. 266, 1147–1155 (1993).
Yu, L. & Dennis, E.A. Critical role of a hydrogen bond in the interaction of phospholipase A2 with transition-state and substrate analogues. Proc. natn. Acad. Sci. U.S.A. 88, 9325–9329 (1991).
Dennis, E.A. Diversity of group types, regulation, and function of phospholipase A2 . J. biol. Chem. 269, 13057–13060 (1994).
Shibata, A. Diffraction data collection with R-AXIS II, an X-ray detecting system using imaging plate. Rigaku J. 7, 28–32 (1990).
Brunger, A. X-PLOR version 3.1 A system for crystallography and NMR. (Yale Univ. Press, New Haven and London; 1992).
Hendrickson, W.A. & Konnert, J.H. Stereochemically restrained least-squares refinement. in Biomolecular Structure, Function, Conformation and Evolution. (ed. Srinivasan, R.) 43–57 (Pergamon, Oxford; 1981).
Jones, T.A. A graphics model building and refinement system for macromolecules. J. appl. Crystallogr. 11, 268–272 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schevitz, R., Bach, N., Carlson, D. et al. Structure-based design of the first potent and selective inhibitor of human non-pancreatic secretory phospholipase A2. Nat Struct Mol Biol 2, 458–465 (1995). https://doi.org/10.1038/nsb0695-458
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0695-458
This article is cited by
-
Interplay between C1-inhibitor and group IIA secreted phospholipase A2 impairs their respective function
Immunologic Research (2023)
-
Group IIA secreted phospholipase A2 inhibition by elemolic acid as a function of anti-inflammatory activity
Scientific Reports (2022)
-
Molecular modeling of Gly80 and Ser80 variants of human group IID phospholipase A2 and their receptor complexes: potential basis for weight loss in chronic obstructive pulmonary disease
Journal of Molecular Modeling (2016)
-
Interactions of selected indole derivatives with phospholipase A2: in silico and in vitro analysis
Journal of Molecular Modeling (2013)
-
Secreted phospholipase A2, lipoprotein hydrolysis, and atherosclerosis: integration with lipidomics
Analytical and Bioanalytical Chemistry (2011)