Abstract
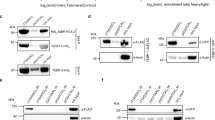
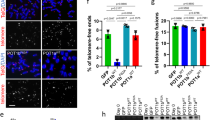
In mammalian cells, the telomeric repeat binding factor (TRF) homology (TRFH) domain–containing telomeric proteins TRF1 and TRF2 associate with a collection of molecules necessary for telomere maintenance and cell-cycle progression. However, the specificity and the mechanisms by which TRF2 communicates with different signaling pathways remain largely unknown. Using oriented peptide libraries, we demonstrate that the TRFH domain of human TRF2 recognizes [Y/F]XL peptides with the consensus motif YYHKYRLSPL. Disrupting the interactions between the TRF2 TRFH domain and its targets resulted in telomeric DNA-damage responses. Furthermore, our genome-wide target analysis revealed phosphatase nuclear targeting subunit (PNUTS) and microcephalin 1 (MCPH1) as previously unreported telomere-associated proteins that directly interact with TRF2 via the [Y/F]XL motif. PNUTS and MCPH1 can regulate telomere length and the telomeric DNA-damage response, respectively. Our findings indicate that an array of TRF2 molecules functions as a protein hub and regulates telomeres by recruiting different signaling molecules via a linear sequence code.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Blackburn, E.H. Switching and signaling at the telomere. Cell 106, 661–673 (2001).
Hackett, J.A. & Greider, C.W. Balancing instability: dual roles for telomerase and telomere dysfunction in tumorigenesis. Oncogene 21, 619–626 (2002).
Granger, M.P., Wright, W.E. & Shay, J.W. Telomerase in cancer and aging. Crit. Rev. Oncol. Hematol. 41, 29–40 (2002).
Harrington, L. & Robinson, M.O. Telomere dysfunction: multiple paths to the same end. Oncogene 21, 592–597 (2002).
Collins, K. & Mitchell, J.R. Telomerase in the human organism. Oncogene 21, 564–579 (2002).
Maser, R.S. & DePinho, R.A. Connecting chromosomes, crisis, and cancer. Science 297, 565–569 (2002).
Hahn, W.C. & Weinberg, R.A. Modelling the molecular circuitry of cancer. Nat. Rev. Cancer 2, 331–341 (2002).
Blasco, M.A. Mammalian telomeres and telomerase: why they matter for cancer and aging. Eur. J. Cell Biol. 82, 441–446 (2003).
Cech, T.R. Beginning to understand the end of the chromosome. Cell 116, 273–279 (2004).
Henderson, E., Hardin, C.C., Walk, S.K., Tinoco Jr, I. & Blackburn, E.H. Telomeric DNA oligonucleotides form novel intramolecular structures containing guanine-guanine base pairs. Cell 51, 899–908 (1987).
de Lange, T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 19, 2100–2110 (2005).
Songyang, Z. & Liu, D. Inside the mammalian telomere interactome: regulation and regulatory activities of telomeres. Crit. Rev. Eukaryot. Gene Expr. 16, 103–118 (2006).
Broccoli, D., Smogorzewska, A., Chong, L. & de Lange, T. Human telomeres contain two distinct Myb-related proteins, TRF1 and TRF2. Nat. Genet. 17, 231–235 (1997).
Bilaud, T. et al. Telomeric localization of TRF2, a novel human telobox protein. Nat. Genet. 17, 236–239 (1997).
Verdun, R.E. & Karlseder, J. Replication and protection of telomeres. Nature 447, 924–931 (2007).
Iwano, T., Tachibana, M., Reth, M. & Shinkai, Y. Importance of TRF1 for functional telomere structure. J. Biol. Chem. 279, 1442–1448 (2004).
Celli, G.B. & de Lange, T. DNA processing is not required for ATM-mediated telomere damage response after TRF2 deletion. Nat. Cell Biol. 7, 712–718 (2005).
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003).
Denchi, E.L. & de Lange, T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT1. Nature 448, 1068–1071 (2007).
Wang, R.C., Smogorzewska, A. & de Lange, T. Homologous recombination generates T-loop-sized deletions at human telomeres. Cell 119, 355–368 (2004).
van Steensel, B., Smogorzewska, A. & de Lange, T. TRF2 protects human telomeres from end-to-end fusions. Cell 92, 401–413 (1998).
van Overbeek, M. & de Lange, T. Apollo, an Artemis-related nuclease, interacts with TRF2 and protects human telomeres in S phase. Curr. Biol. 16, 1295–1302 (2006).
Lenain, C. et al. The Apollo 5′ exonuclease functions together with TRF2 to protect telomeres from DNA repair. Curr. Biol. 16, 1303–1310 (2006).
Chen, Y. et al. A shared docking motif in TRF1 and TRF2 used for differential recruitment of telomeric proteins. Science 319, 1092–1096 (2008).
Songyang, Z. et al. SH2 domains recognize specific phosphopeptide sequences. Cell 72, 767–778 (1993).
Songyang, Z. Recognition and regulation of primary-sequence motifs by signaling modular domains. Prog. Biophys. Mol. Biol. 71, 359–372 (1999).
Yaffe, M.B. et al. A motif-based profile scanning approach for genome-wide prediction of signaling pathways. Nat. Biotechnol. 19, 348–353 (2001).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Karlseder, J., Broccoli, D., Dai, Y., Hardy, S. & de Lange, T. p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 283, 1321–1325 (1999).
Liu, D., O'Connor, M.S., Qin, J. & Songyang, Z. Telosome, a mammalian telomere-associated complex formed by multiple telomeric proteins. J. Biol. Chem. 279, 51338–51342 (2004).
O'Connor, M.S., Safari, A., Liu, D., Qin, J. & Songyang, Z. The human Rap1 protein complex and modulation of telomere length. J. Biol. Chem. 279, 28585–28591 (2004).
Hu, C.D. & Kerppola, T.K. Simultaneous visualization of multiple protein interactions in living cells using multicolor fluorescence complementation analysis. Nat. Biotechnol. 21, 539–545 (2003).
Chen, L.Y., Liu, D. & Songyang, Z. Telomere maintenance through spatial control of telomeric proteins. Mol. Cell. Biol. 27, 5898–5909 (2007).
Allen, P.B., Kwon, Y.G., Nairn, A.C. & Greengard, P. Isolation and characterization of PNUTS, a putative protein phosphatase 1 nuclear targeting subunit. J. Biol. Chem. 273, 4089–4095 (1998).
Lin, S.Y., Rai, R., Li, K., Xu, Z.X. & Elledge, S.J. BRIT1/MCPH1 is a DNA damage responsive protein that regulates the Brca1-Chk1 pathway, implicating checkpoint dysfunction in microcephaly. Proc. Natl. Acad. Sci. USA 102, 15105–15109 (2005).
Rai, R. et al. BRIT1 regulates early DNA damage response, chromosomal integrity, and cancer. Cancer Cell 10, 145–157 (2006).
Alderton, G.K. et al. Regulation of mitotic entry by microcephalin and its overlap with ATR signalling. Nat. Cell Biol. 8, 725–733 (2006).
Wood, J.L., Singh, N., Mer, G. & Chen, J. MCPH1 functions in an H2AX-dependent but MDC1-independent pathway in response to DNA damage. J. Biol. Chem. 282, 35416–35423 (2007).
O'Connor, M.S., Safari, A., Xin, H., Liu, D. & Songyang, Z. A critical role for TPP1 and TIN2 interaction in high-order telomeric complex assembly. Proc. Natl. Acad. Sci. USA 103, 11874–11879 (2006).
Xin, H. et al. TPP1 is a homologue of ciliate TEBP-β and interacts with POT1 to recruit telomerase. Nature 445, 559–562 (2007).
Bae, N.S. & Baumann, P.A. RAP1/TRF2 complex inhibits nonhomologous end-joining at human telomeric DNA ends. Mol. Cell 26, 323–334 (2007).
Price, C.M. wRAPing up the end to prevent telomere fusions. Mol. Cell 26, 463–464 (2007).
Kim, H., Chen, J. & Yu, X. Ubiquitin-binding protein RAP80 mediates BRCA1-dependent DNA damage response. Science 316, 1202–1205 (2007).
Songyang, Z. & Cantley, L.C. The use of peptide library for the determination of kinase peptide substrates. Methods Mol. Biol. 87, 87–98 (1998).
Liu, D. et al. PTOP interacts with POT1 and regulates its localization to telomeres. Nat. Cell Biol. 6, 673–680 (2004).
Mendez, J. & Stillman, B. Chromatin association of human origin recognition complex, Cdc6, and minichromosome maintenance proteins during the cell cycle: assembly of prereplication complexes in late mitosis. Mol. Cell. Biol. 20, 8602–8612 (2000).
Acknowledgements
We thank S.Y. Jung and Q. He for technical help and M. Lei (University of Michigan) for the GST-TRF2TRFH fusion proteins. We thank J. Pennington and T. Palzkill for peptide synthesis. Work in the laboratories of Z.S. and D.L. is supported by awards from the US National Institutes of Health, the US Department of Defense, American Heart Association, the Welch foundation and the American Cancer Society. Z.S. is funded by the Leukemia and Lymphoma Society.
Author information
Authors and Affiliations
Contributions
H.K., O.-H.L., H.X. and L.-Y.C. designed and performed most of the experiments; D.L. and J.Q. did the telomere length and MS experiment, respectively; A.S. and H.K.C. provided technical support. S.-Y.L. provided the MCHP1 reagents; D.L. and Z.S. wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 1320 kb)
Rights and permissions
About this article
Cite this article
Kim, H., Lee, OH., Xin, H. et al. TRF2 functions as a protein hub and regulates telomere maintenance by recognizing specific peptide motifs. Nat Struct Mol Biol 16, 372–379 (2009). https://doi.org/10.1038/nsmb.1575
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1575
This article is cited by
-
Microcephalin 1/BRIT1-TRF2 interaction promotes telomere replication and repair, linking telomere dysfunction to primary microcephaly
Nature Communications (2020)
-
The role of telomere-binding modulators in pluripotent stem cells
Protein & Cell (2020)
-
Serum Extracellular Vesicles Retard H9C2 Cell Senescence by Suppressing miR-34a Expression
Journal of Cardiovascular Translational Research (2019)
-
Structural and functional analyses of the mammalian TIN2-TPP1-TRF2 telomeric complex
Cell Research (2017)
-
Systematic analysis of human telomeric dysfunction using inducible telosome/shelterin CRISPR/Cas9 knockout cells
Cell Discovery (2017)