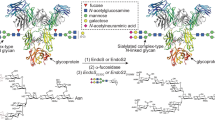
Abstract
In eukaryotes, glycosylated proteins are ubiquitous components of extracellular matrices and cellular surfaces. Their oligosaccharide moieties are implicated in a wide range of cell?cell and cell?matrix recognition events that are required for biological processes ranging from immune recognition to cancer development. Glycosylation was previously considered to be restricted to eukaryotes; however, through advances in analytical methods and genome sequencing, there have been increasing reports of both O-linked and N-linked protein glycosylation pathways in bacteria, particularly amongst mucosal-associated pathogens. Studying glycosylation in relatively less-complicated bacterial systems provides the opportunity to elucidate and exploit glycoprotein biosynthetic pathways. We will review the genetic organization, glycan structures and function of glycosylation systems in mucosal bacterial pathogens, and speculate on how this knowledge may help us to understand glycosylation processes in more complex eukaryotic systems and how it can be used for glycoengineering.
Key Points
-
It is now apparent that organisms from all three domains of life are capable of modifying their proteins through glycosylation.
-
Similar processes of glycoconjugate biosynthesis are conserved among all three domains by segregating key steps of the pathway with membranes, by the use of nucleotide-activated and/or lipid-linked intermediates, and by transfer of sugars to the same amino acid sequons.
-
Among Bacteria, mucosal pathogens have a particular propensity to glycosylate surface structures.
-
Mucosal pathogens frequently share similar sugar biosynthetic genes, resulting in similar glycans such as bacillosamine- and pseudaminic acid-like structures.
-
Campylobacter jejuni is unique among Bacteria as it has well-characterized O- and N-linked glycosylation systems.
-
The recent demonstration of the transfer of the Campylobacter N-linked glycosylation pathway into Escherichia coli opens up the possibility of producing recombinant glycoproteins. Together with the detailed characterization of several bacterial glycosylation pathways, the opportunity to engineer countless permutations of novel glycoproteins in a simple E. coli host is now possible.
-
Further characterization of the Campylobacter O- and N-linked glycosylation systems and other bacterial systems will provide useful models to study more complex eukaryotic protein glycosylation pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wacker, M. et al. N-linked glycosylation in Campylobacter jejuni and its functional transfer into E. coli. Science 298, 1790?1793 (2002). Demonstration of the functional transfer of a glycosylation system into E. coli.
Young, N. M. et al. Structure of the N-linked glycan present on multiple glycoproteins in the Gram-negative bacterium, Campylobacter jejuni. J. Biol. Chem. 277, 42530?42539 (2002). First determination of the structure of the Campylobacter N -linked glycan and confirmation of the glycosylation of multiple proteins by the N -linked general glycosylation pathway in Campylobacter.
Thibault, P. et al. Identification of the carbohydrate moieties and glycosylation motifs in Campylobacter jejuni flagellin. J. Biol. Chem. 276, 34862?34870 (2001). Determination of the structure of the Campylobacter O -linked glycan.
Logan, S. M., Kelly, J. F., Thibault, P., Ewing, C. P. & Guerry, P. Structural heterogeneity of carbohydrate modifications affects serospecificity of Campylobacter flagellins. Mol. Microbiol. 46, 587?597 (2002). Detailed demonstration of the complexity of O -linked sugar modification on the surface-exposed flagella of Campylobacter.
Goon, S., Kelly, J. F., Logan, S. M., Ewing, C. P. & Guerry, P. Pseudaminic acid, the major modification on Campylobacter flagellin, is synthesized via the Cj1293 gene. Mol. Microbiol. 50, 659?671 (2003).
Power, P. M. & Jennings, M. P. The genetics of glycosylation in Gram-negative bacteria. FEMS Microbiol. Lett. 218, 211?222 (2003).
Benz, I. & Schmidt, M. A. Never say never again: protein glycosylation in pathogenic bacteria. Mol. Microbiol. 45, 267?276 (2002).
Schmidt, M. A., Riley, L. W. & Benz, I. Sweet new world: glycoproteins in bacterial pathogens. Trends Microbiol. 11, 554?561 (2003).
Messner, P. & Schäffer, C. in Progress in the Chemistry of Organic Natural Products (eds Herz, W., Falk, H. & Kirby, G. W.) 51?124 (Springer?Verlag, Wien, 2003).
Szymanski, C. M., Logan, S. M., Linton, D. & Wren, B. W. Campylobacter ? a tale of two protein glycosylation systems. Trends Microbiol. 11, 233?238 (2003).
Doig, P., Kinsella, N., Guerry, P. & Trust, T. J. Characterization of a post-translational modification of Campylobacter flagellin: identification of a sero-specific glycosyl moiety. Mol. Microbiol. 19, 379?387 (1996).
Parkhill, J. et al. The genome sequence of the food-borne pathogen Campylobacter jejuni reveals hypervariable sequences. Nature 403, 665?668 (2000).
Guerry, P. et al. Identification and characterization of genes required for post-translational modification of Campylobacter coli VC167 flagellin. Mol. Microbiol. 19, 369?378 (1996).
Linton, D. et al. Phase variation of a β-1,3 galactosyltransferase involved in generation of the ganglioside GM1-like lipo-oligosaccharide of Campylobacter jejuni. Mol. Microbiol. 37, 501?514 (2000).
Macnab, R. M. How bacteria assemble flagella. Annu. Rev. Microbiol. (2003).
Karlyshev, A. V., Linton, D., Gregson, N. A. & Wren, B. W. A novel paralogous gene family involved in phase-variable flagella-mediated motility in Campylobacter jejuni. Microbiology 148, 473?480 (2002).
Korolik, V., Fry, B. N., Alderton, M. R., van der Zeijst, B. A. & Coloe, P. J. Expression of Campylobacter hyoilei lipo-oligosaccharide (LOS) antigens in Escherichia coli. Microbiology 143, 3481?3489 (1997).
Fry, B. N. et al. The lipopolysaccharide biosynthesis locus of Campylobacter jejuni 81116. Microbiology 144, 2049?2061 (1998).
Szymanski, C. M., Yao, R., Ewing, C. P., Trust, T. J. & Guerry, P. Evidence for a system of general protein glycosylation in Campylobacter jejuni. Mol. Microbiol. 32, 1022?1030 (1999). First report of the Campylobacter general glycosylation pathway.
Zufferey, R. et al. STT3, a highly conserved protein required for yeast oligosaccharyl transferase activity in vivo. EMBO J. 14, 4949?4960 (1995).
Knauer, R. & Lehle, L. The oligosaccharyltransferase complex from yeast. Biochim. Biophys. Acta 1426, 259?273 (1999).
Silberstein, S. & Gilmore, R. Biochemistry, molecular biology, and genetics of the oligosaccharyltransferase. FASEB J. 10, 849?858 (1996).
Yan, A., Ahmed, E., Yan, Q. & Lennarz, W. J. New findings on interactions among the yeast oligosaccharyl transferase subunits using a chemical cross-linker. J. Biol. Chem. 278, 33078?33087 (2003).
Yan, Q. & Lennarz, W. J. Studies on the function of oligosaccharyl transferase subunits. Stt3p is directly involved in the glycosylation process. J. Biol. Chem. 277, 47692?47700 (2002). First evidence that STT3p is directly involved in glycosylation.
Virji, M. in Glycomicrobiology. (ed. Doyle,R. J.) 31?65 (Kluwer Academic/Plenum, New York, 2000).
Hegge, F. T. et al. Unique modifications with phosphocholine and phosphoethanolamine define alternate antigenic forms of Neisseria gonorrhoeae type IV pili. Proc. Natl Acad. Sci. USA 101, 10798?10803 (2004).
Endo, T. et al. α-1,4-N-acetylgalactosamine transferase gene and process for producing this enzyme and N-acetylgalactosamine-containing complex carbohydrate. Japan patent WO 0177337 (2002).
Suzuki, S., Perry, M., Thibault, P. & Honda, S. in Capillary Electrophoresis of Carbohydrates. (eds Thibault, P. & Honda, S.) 285?305 (Humana Press, New Jersey, 2003).
Castric, P., Cassels, F. J. & Carlson, R. W. Structural characterization of the Pseudomonas aeruginosa 1244 pilin glycan. J. Biol. Chem. 276, 26479?26485 (2001).
Schirm, M. et al. Structural, genetic and functional characterization of the flagellin glycosylation process in Helicobacter pylori. Mol. Microbiol. 48, 1579?1592 (2003).
Soo, E. C. et al. Selective detection and identification of sugar nucleotides by CE-electrospray-MS and its application to bacterial metabolomics. Anal. Chem. 76, 619?626 (2004). Novel method for the analysis of sugar pathway intermediates, leading to a new era of metabolomics.
Kneidinger, B., Graninger, M., Puchberger, M., Kosma, P. & Messner, P. Biosynthesis of nucleotide-activated D-glycero-D-manno-heptose. J. Biol. Chem. 276, 20935?20944 (2001).
Karlyshev, A. V. et al. Analysis of Campylobacter jejuni capsular loci reveals multiple mechanisms for the generation of structural diversity and the ability to form complex heptoses. Mol. Microbiol. 55, 90?103 (2005).
Bernatchez, S. et al. A single bifunctional UDP-GlcNAc/Glc 4-epimerase supports the synthesis of three cell surface glycoconjugates in Campylobacter jejuni. J. Biol. Chem. 27 Oct 2004 (10.1074/jbc.M407767200).
Fry, B. N. et al. The galE gene of Campylobacter jejuni is involved in lipopolysaccharide synthesis and virulence. Infect. Immun. 68, 2594?2601 (2000).
Stimson, E. et al. Meningococcal pilin: a glycoprotein substituted with digalactosyl 2,4-diacetamido-2,4,6-trideoxyhexose. Mol. Microbiol. 17, 1201?1214 (1995). Identification of a bacillosamine derivative on meningococcal pilin.
Fischer, W. & Haas, R. The RecA protein of Helicobacter pylori requires a post-translational modification for full activity. J. Bacteriol. 186, 777?784 (2004).
Grass, S. et al. The Haemophilus influenzae HMW1 adhesin is glycosylated in a process that requires HMW1C and phosphoglucomutase, an enzyme involved in lipooligosaccharide biosynthesis. Mol. Microbiol. 48, 737?751 (2003).
Benz, I. & Schmidt, M. A. Glycosylation with heptose residues mediated by the aah gene product is essential for adherence of the AIDA-I adhesin. Mol. Microbiol. 40, 1403?1413 (2001).
DiGiandomenico, A. et al. Glycosylation of Pseudomonas aeruginosa 1244 pilin: glycan substrate specificity. Mol. Microbiol. 46, 519?530 (2002). Demonstration that the Pseudomonas O -glycan transferase, PilO, can attach foreign O-antigens to pilin, providing new possibilities for glycoengineering.
Kahler, C. M. et al. Polymorphisms in pilin glycosylation locus of Neisseria meningitidis expressing class II pili. Infect. Immun. 69, 3597?3604 (2001).
Arora, S. K., Wolfgang, M. C., Lory, S. & Ramphal, R. Sequence polymorphism in the glycosylation island and flagellins of Pseudomonas aeruginosa. J. Bacteriol. 186, 2115?2122 (2004).
Takeuchi, K. et al. Flagellin glycosylation island in Pseudomonas syringae pv. glycinea and its role in host specificity. J. Bacteriol. 185, 6658?6665 (2003). Evidence that flagellin glycosylation has a role in host specificity.
Dorrell, N. et al. Whole genome comparison of Campylobacter jejuni human isolates using a low-cost microarray reveals extensive genetic diversity. Genome Res. 11, 1706?1715 (2001).
Leonard, E. E. et al. Use of an open-reading-frame-specific Campylobacter jejuni DNA microarray as a new genotyping tool for studying epidemiologically related isolates. J. Infect. Dis. 187, 691?694 (2003).
Szymanski, C. M. et al. Detection of conserved N-linked glycans and phase-variable lipooligosaccharides and capsules from campylobacter cells by mass spectrometry and high-resolution magic angle spinning NMR spectroscopy. J. Biol. Chem. 278, 24509?24520 (2003).
Taboada, E. N. et al. Large-scale comparative genomics meta-analysis of Campylobacter jejuni isolates reveals low level of genome plasticity. J. Clin. Microbiol. 42, 4566?4576 (2004).
Pearson, B. M. et al. Comparative genome analysis of Campylobacter jejuni using whole genome DNA microarrays. FEBS Lett. 554, 224?230 (2003).
Fouts, D. E. et al. Major structural differences and novel potential virulence mechanisms from the genomes of multiple Campylobacter species. PLoS Biol. 3, 1?14 (2005).
Baar, C. et al. Complete genome sequence and analysis of Wolinella succinogenes. Proc. Natl Acad. Sci. USA 100, 11690?11695 (2003).
Schaffer, C., Graninger, M. & Messner, P. Prokaryotic glycosylation. Proteomics 1, 248?261 (2001).
Kuo, C., Takahashi, N., Swanson, A. F., Ozeki, Y. & Hakomori, S. An N-linked high-mannose type oligosaccharide, expressed at the major outer membrane protein of Chlamydia trachomatis, mediates attachment and infectivity of the microorganism to HeLa cells. J. Clin. Invest. 98, 2813?2818 (1996).
Lindenthal, C. & Elsinghorst, E. A. Identification of a glycoprotein produced by enterotoxigenic Escherichia coli. Infect. Immun. 67, 4084?4091 (1999). First detailed report of protein glycosylation in Escherichia coli.
Miron, J. & Forsberg, C. W. Characterisation of cellulose-binding proteins that are involved in the adhesion mechanism of Fibrobacter intestinalis DR7. Appl. Microbiol. Biotechnol. 51, 491?497 (1999).
Herrmann, J. L., O'Gaora, P., Gallagher, A., Thole, J. E. & Young, D. B. Bacterial glycoproteins: a link between glycosylation and proteolytic cleavage of a 19-kDa antigen from Mycobacterium tuberculosis. EMBO J. 15, 3547?3554 (1996).
Marceau, M. & Nassif, X. Role of glycosylation at Ser63 in production of soluble pilin in pathogenic Neisseria. J. Bacteriol. 181, 656?661 (1999).
Romain, F. et al. Deglycosylation of the 45/47-kilodalton antigen complex of Mycobacterium tuberculosis decreases its capacity to elicit in vivo or in vitro cellular immune responses. Infect. Immun. 67, 5567?5572 (1999).
Ghosh, S. K. et al. Pathogenic consequences of Neisseria gonorrhoeae pilin glycan variation. Microbes. Infect. 6, 693?701 (2004).
Hamadeh, R. M., Estabrook, M. M., Zhou, P., Jarvis, G. A. & Griffiss, J. M. Anti-Gal binds to pili of Neisseria meningitidis: the immunoglobulin A isotype blocks complement-mediated killing. Infect. Immun. 63, 4900?4906 (1995).
Serino, L. & Virji, M. Genetic and functional analysis of the phosphorylcholine moiety of commensal Neisseria lipopolysaccharide. Mol. Microbiol. 43, 437?448 (2002).
Schultze, M. & Kondorosi, A. The role of lipochitooligosaccharides in root nodule organogenesis and plant cell growth. Curr. Opin. Genet. Dev. 6, 631?638 (1996).
Lindenthal, C. & Elsinghorst, E. A. Enterotoxigenic Escherichia coli TibA glycoprotein adheres to human intestine epithelial cells. Infect. Immun. 69, 52?57 (2001).
Moormann, C., Benz, I. & Schmidt, M. A. Functional substitution of the TibC protein of enterotoxigenic Escherichia coli strains for the autotransporter adhesin heptosyltransferase of the AIDA system. Infect. Immun. 70, 2264?2270 (2002).
Leclerc, G., Wang, S. P. & Ely, B. A new class of Caulobacter crescentus flagellar genes. J. Bacteriol. 180, 5010?5019 (1998).
Gryllos, I., Shaw, J. G., Gavin, R., Merino, S. & Tomas, J. M. Role of flm locus in mesophilic Aeromonas species adherence. Infect. Immun. 69, 65?74 (2001).
Arora, S. K., Bangera, M., Lory, S. & Ramphal, R. A genomic island in Pseudomonas aeruginosa carries the determinants of flagellin glycosylation. Proc. Natl Acad. Sci. USA 98, 9342?9347 (2001).
Schmitz, A., Josenhans, C. & Suerbaum, S. Cloning and characterization of the Helicobacter pylori flbA gene, which codes for a membrane protein involved in coordinated expression of flagellar genes. J. Bacteriol. 179, 987?997 (1997).
Bedouet, L. et al. Evidence for an heterogeneous glycosylation of the Clostridium tyrobutyricum ATCC 25755 flagellin. Microbios 94, 183?192 (1998).
Schirm, M. et al. Structural and genetic characterization of glycosylation of type a flagellin in Pseudomonas aeruginosa. J. Bacteriol. 186, 2523?2531 (2004).
Szymanski, C. M., Burr, D. H. & Guerry, P. Campylobacter protein glycosylation affects host cell interactions. Infect. Immun. 70, 2242?2244 (2002).
Hendrixson, D. R. & DiRita, V. J. Identification of Campylobacter jejuni genes involved in commensal colonization of the chick gastrointestinal tract. Mol. Microbiol. 52, 471?484 (2004).
Szymanski, C. M., Goon, S., Allan, B. & Guerry, P. in Campylobacter: Molecular and Cellular Biology (eds Ketley, J. M. & Konkel, M. E.) 259?273 (Horizon Bioscience, Norwich, 2005).
Jones, M. A. et al. Adaptation of Campylobacter jejuni NCTC11168 to high-level colonization of the avian gastrointestinal tract. Infect. Immun. 72, 3769?3776 (2004).
Larsen, J. C., Szymanski, C. & Guerry, P. N-linked protein glycosylation is required for full competence in Campylobacter jejuni 81-176. J. Bacteriol. 186, 6508?6514 (2004).
Whitfield, C. & Roberts, I. S. Structure, assembly and regulation of expression of capsules in Escherichia coli. Mol. Microbiol. 31, 1307?1319 (1999).
Lin, J., Michel, L. O. & Zhang, Q. CmeABC functions as a multidrug efflux system in Campylobacter jejuni. Antimicrob. Agents Chemother. 46, 2124?2131 (2002).
Helenius, A. & Aebi, M. Intracellular functions of N-linked glycans. Science 291, 2364?2369 (2001). Review of the functions of eukaryotic N -linked glycans in the endoplasmic reticulum.
Wacker, M., Nita-Lazar, M. & Aebi, M. PglB, an oligosaccharyltransferase in the eubacterium Campylobacter jejuni? (Abstracts of the 11th International Workshop on Campylobacter, Helicobacter and related organisms. September 1?5, 2001. Freiburg, Germany). Int. J. Med. Microbiol. 291 (Suppl.), 81 (2001).
Wieland, F. Structure and biosynthesis of prokaryotic glycoproteins. Biochimie 70, 1493?1504 (1988).
Zhu, B. C., Drake, R. R., Schweingruber, H. & Laine, R. A. Inhibition of glycosylation by amphomycin and sugar nucleotide analogs PP36 and PP55 indicates that Haloferax volcanii β-glucosylates both glycoproteins and glycolipids through lipid-linked sugar intermediates: evidence for three novel glycoproteins and a novel sulfated dihexosyl-archaeol glycolipid. Arch. Biochem. Biophys. 319, 355?364 (1995).
Kuntz, C., Sonnenbichler, J., Sonnenbichler, I., Sumper, M. & Zeitler, R. Isolation and characterization of dolichol-linked oligosaccharides from Haloferax volcanii. Glycobiology 7, 897?904 (1997).
Zeitler, R., Hochmuth, E., Deutzmann, R. & Sumper, M. Exchange of Ser-4 for Val, Leu or Asn in the sequon Asn?Ala?Ser does not prevent N-glycosylation of the cell surface glycoprotein from Halobacterium halobium. Glycobiology 8, 1157?1164 (1998).
Imperiali, B. & Hendrickson, T. L. Asparagine-linked glycosylation: specificity and function of oligosaccharyl transferase. Bioorg. Med. Chem. 3, 1565?1578 (1995).
Schirm, M. et al. Flagellin from Listeria monocytogenes is glycosylated with β-O-linked N-acetylglucosamine. J. Bacteriol. 186, 6721?6727 (2004).
Zachara, N. E. & Hart, G. W. O-GlcNAc a sensor of cellular state: the role of nucleocytoplasmic glycosylation in modulating cellular function in response to nutrition and stress. Biochim. Biophys. Acta 1673, 13?28 (2004).
Aebi, M. & Hennet, T. Congenital disorders of glycosylation: genetic model systems lead the way. Trends Cell Biol. 11, 136?141 (2001). Review of the use of Saccharomyces cerevisiae as a model to study inherited human diseases involving congenital disorders of N -linked glycosylation.
Alton, G. et al. Direct utilization of mannose for mammalian glycoprotein biosynthesis. Glycobiology 8, 285?295 (1998).
Davis, J. A. & Freeze, H. H. Studies of mannose metabolism and effects of long-term mannose ingestion in the mouse. Biochim. Biophys. Acta 1528, 116?126 (2001).
Apweiler, R., Hermjakob, H. & Sharon, N. On the frequency of protein glycosylation, as deduced from analysis of the SWISS-PROT database. Biochim. Biophys. Acta 1473, 4?8 (1999).
Varki, A. Biological roles of oligosaccharides: all of the theories are correct. Glycobiology 3, 97?130 (1993).
Betenbaugh, M. J., Tomiya, N., Narang, S., Hsu, J. T. & Lee, Y. C. Biosynthesis of human-type N-glycans in heterologous systems. Curr. Opin. Struct. Biol. 14, 601?606 (2004).
Raju, T. S., Briggs, J. B., Chamow, S. M., Winkler, M. E. & Jones, A. J. Glycoengineering of therapeutic glycoproteins: in vitro galactosylation and sialylation of glycoproteins with terminal N-acetylglucosamine and galactose residues. Biochemistry 40, 8868?8876 (2001).
Elliott, S. et al. Enhancement of therapeutic protein in vivo activities through glycoengineering. Nature Biotechnol. 21, 414?421 (2003).
Bobrowicz, P. et al. Engineering of an artificial glycosylation pathway blocked in core oligosaccharide assembly in the yeast Pichia pastoris: production of complex humanized glycoproteins with terminal galactose. Glycobiology 14, 757?766 (2004).
Imperiali, B. & O'Connor, S. E. Effect of N-linked glycosylation on glycopeptide and glycoprotein structure. Curr. Opin. Chem. Biol. 3, 643?649 (1999). Review of the effect of N -linked glycosylation on protein folding or the stability of the native glycoprotein conjugates.
Gemmill, T. R. & Trimble, R. B. Overview of N- and O-linked oligosaccharide structures found in various yeast species. Biochim. Biophys. Acta 1426, 227?237 (1999).
Luo, Y., Luther, K. B. & Haltiwanger, R. S. Protein O-fucosyltransferase 1 (O-FucT-1) is a soluble enzyme localized in the ER. Glycobiology 14, 1059 (2004).
Hart, G. W. Dynamic O-linked glycosylation of nuclear and cytoskeletal proteins. Annu. Rev. Biochem. 66, 315?335 (1997).
Hanover, J. A. Glycan-dependent signaling: O-linked N-acetylglucosamine. FASEB J. 15, 1865?1876 (2001).
Creuzenet, C. Characterization of Cj1293, a new UDP-GlcNAc C6 dehydratase from Campylobacter jejuni. FEBS Lett. 559, 136?140 (2004).
Creuzenet, C., Schur, M. J., Li, J., Wakarchuk, W. W. & Lam, J. S. FlaA1, a new bifunctional UDP-GlcNAc C6 Dehydratase/C4 reductase from Helicobacter pylori. J. Biol. Chem. 275, 34873?34880 (2000).
Linton, D. et al. Multiple N-acetyl neuraminic acid synthetase (neuB) genes in Campylobacter jejuni: identification and characterization of the gene involved in sialylation of lipo-oligosaccharide. Mol. Microbiol. 35, 1120?1134 (2000).
Bleumink-Pluym, N. M., Verschoor, F., Gaastra, W., van der Zeijst, B. A. & Fry, B. N. A novel approach for the construction of a Campylobacter mutant library. Microbiology 145, 2145?2151 (1999).
Guerry, P., Logan, S. M., Thornton, S. & Trust, T. J. Genomic organization and expression of Campylobacter flagellin genes. J. Bacteriol. 172, 1853?1860 (1990).
Wildt, S. & Gerngross, T. U. The humanization of N-glycosylation pathways in yeast. Nature Rev. Microbiol. 3, 119?128 (2005).
Acknowledgements
We thank S. Logan for the critical reading of this review, J.-R. Brisson, N. M. Young and N. Khieu for constructing the space-filling models and M. Aebi for reviewing the manuscript and providing information prior to publication. We also thank R. Mandrell and W. Miller for providing genomic data for the Campylobacter species described in this review and for assembling online supplementary information S1 (Table). We gratefully acknowledge the NRC Genomics and Health Initiative for providing funding to C.M.S and the Leverhulme Trust and BBSRC for funding to B.W.W.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Related links
Related links
DATABASES
Entrez
Campylobacter upsaliensis RM3195
SwissProt
FURTHER INFORMATION
Desulfovibrio desulfuricans genome
Glossary
- FLAGELLIN
-
The structural protein of which the bacterial flagellum is constructed.
- LIPOPOLYSACCHARIDE
-
(LPS). An important amphiphilic molecule integrated in and extending outward from the outer membrane of the Gram-negative bacterial cell wall; structurally composed of hydrophobic Lipid A (responsible for endotoxin activity), core polysaccharide and hydrophilic O-antigen polysaccharide side chains.
- LIPOOLIGOSACCHARIDE
-
(LOS). Similar to LPS but lacking the O-antigen polysaccharide side chain repeats.
- CAPILLARY ELECTROPHORESIS COUPLED TO ELECTROSPRAY MASS SPECTROMETRY
-
(CE-ESMS). A coupled system where complex mixtures are first separated by capillary electrophoresis before being introduced into the mass spectrometer using electrospray ionization.
- S-LAYER PROTEIN
-
Surface (S) layers are composed of a crystalline array of high-molecular-weight protein or glycoprotein subunits and form a matrix surrounding some bacterial cells.
- CAPSULAR POLYSACCHARIDE
-
(CPS). Also sometimes referred to as K-antigen, capsules are composed of polysaccharide repeats that surround some bacterial cells and are anchored in the membrane by a terminal lipid moiety.
- O-ANTIGEN
-
A polysaccharide antigen extending from the outer membrane of some Gram-negative bacterial cell walls that is a part of the LPS; this repeat is also the serodeterminant for the classical heat-stable O-typing scheme.
- FRAMESHIFT MUTATION
-
A mutation arising from the loss or gain of a base or DNA segment leading to a change in the codon reading frame and thus a change in the amino acids incorporated into the protein.
- PHASE VARIATION
-
Variable expression of a structure that is governed by random frameshift mutations within genes responsible for the biosynthesis of the structure or through changes in the regulation of structure synthesis.
- HIGH-RESOLUTION MAGIC-ANGLE SPINNING NMR
-
(HR-MAS NMR). An adaptation of solid-state NMR where samples are spun rapidly around an axis inclined at an angle of 54.7°, the 'magic angle', with the direction of the magnetic field.
- LIPOCHITOOLIGOSACCHARIDE
-
Plant bacteria signalling molecules, also known as Nod factors, that consist of a backbone of 2?6 β-(1→4)-linked GlcNAc residues with an amide-bound fatty acyl residue (saturated or unsaturated) on the non-reducing terminal GlcN residue. This basic structure has variations that are dependent on each strain or species and determine the host specificity.
Rights and permissions
About this article
Cite this article
Szymanski, C., Wren, B. Protein glycosylation in bacterial mucosal pathogens. Nat Rev Microbiol 3, 225–237 (2005). https://doi.org/10.1038/nrmicro1100
Issue Date:
DOI: https://doi.org/10.1038/nrmicro1100
This article is cited by
-
PglB function and glycosylation efficiency is temperature dependent when the pgl locus is integrated in the Escherichia coli chromosome
Microbial Cell Factories (2022)
-
Genetic and process engineering strategies for enhanced recombinant N-glycoprotein production in bacteria
Microbial Cell Factories (2021)
-
Oligosaccharyltransferase PglB of Campylobacter jejuni is a glycoprotein
World Journal of Microbiology and Biotechnology (2020)
-
Transcriptomic analysis of Campylobacter jejuni grown in a medium containing serine as the main energy source
Archives of Microbiology (2019)
-
Characterization of the pgf operon involved in the posttranslational modification of Streptococcus mutans surface proteins
Scientific Reports (2018)