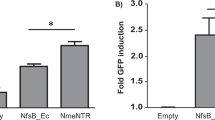
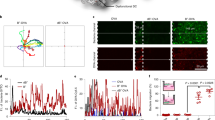
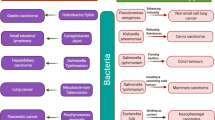
Abstract
During the past decade, the search for an effective system for the selective delivery of high therapeutic doses of anti-cancer agents to tumours has explored a variety of ingenious and increasingly complex biological systems. These systems are most often based on gene therapy and use viral vectors as the delivery vehicle. Invariably, such systems have been found wanting with respect to a lack of tumour specificity, poor levels of transgene expression and inefficient distribution of the vector throughout the tumour mass. By contrast, the ability of intravenously injected clostridial spores to infiltrate, then selectively germinate in, the hypoxic regions of solid tumours seems to be a totally natural phenomenon, which requires no fundamental alterations and is exquisitely specific.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Bagshawe, K. D. Antibody directed enzymes revive anti-cancer prodrug concept. Br. J. Cancer 56, 531–532 (1987).
Dobbelstein, M. Viruses in therapy — royal road or dead end? Virus Res. 92, 219–221 (2003).
St George, J. A. Gene therapy progress and prospects: adenoviral vectors. Gene Ther. 10, 1135–1141 (2003).
Krasnykh, V. et al. Advanced generation adenoviral vectors possess augmented gene transfer efficiency based upon coxsackie adenovirus receptor-independent cellular entry capacity. Cancer Res. 60, 6784–6787 (2000).
Alemany, R., Balague, C. & Curiel, D. T. Replicative adenoviruses in cancer therapy. Nature Biotechnol. 18, 723–727 (2000).
Connolly, J. B. Conditionally replicating viruses in cancer therapy. Gene Ther. 10, 712–715 (2003).
Reddy, J. A. et al. Folate-targeted, cationic liposome-mediated gene transfer into disseminated peritoneal tumors. Gene Ther. 9, 1542–1550 (2002).
Ogris, M. et al. Tumor-targeted gene therapy: strategies for the preparation of ligand-polyethylene glycol-polyethylenimine/DNA complexes. J. Control. Release 91, 173–181 (2003).
De Palma, M., Venneri, M. A. & Naldini, L. In vivo targeting of tumor endothelial cells by systemic delivery of lentiviral vectors. Hum. Gene Ther. 14, 1193–1206 (2003).
Greco, O. et al. Novel chimeric gene promoters responsive to hypoxia and ionizing radiation. Gene Ther. 20, 1403–1411 (2002).
Smrekar, B. et al. Tissue-dependent factors affect gene delivery to tumors in vivo. Gene Ther. 10, 1079–1088 (2003).
Semenez, G. L. Hypoxia-inducible factor 1: master regulator of 02 homeostasis. Curr. Opin. Genet. Dev. 8, 588–594 (1998).
Binley, K. et al. Hypoxia-mediated tumour targeting. Gene Ther. 10, 540–549 (2003).
Harris, A. L. Hypoxia — a key regulatory factor in tumour growth. Nature Rev. Cancer 1, 38–47 (2002).
Minton, N. P., Brown, J. M., Lambin, P. & Anné, J. in Clostridia — Biotechnology and Medical Applications. (eds Bahl, H. & Dürre, P.) 251–270 (Wiley, Weinheim, 2001).
Parker, R. C., Plumber, H. C., Siebenmann, C. O. & Chapman, M. G. Effect of histolyticus infection and toxin on transplantable mouse tumours. Proc. Soc. Exp. Biol. Med. 66, 461–465 (1947).
Malmgren, R. A. & Flanigan, C. C. Localization of the vegetative form of Clostridium tetani in mouse tumours following intravenous spore administration. Cancer Res. 15, 473–478 (1955).
Theys, J. et al. Stable Escherichia coli–Clostridium acetobutylicum shuttle vector for secretion of murine tumor necrosis factor α. Appl. Environ. Microbiol. 65, 4295–4300 (1999).
Theys, J. et al. Clostridium as a tumor-specific delivery system of therapeutic proteins. Cancer Detect. Prevent. 25, 548–557 (2001).
Francis, R. J. et al. A phase I trial of antibody directed enzyme prodrug therapy (ADEPT) in patients with advanced colorectal carcinoma or other CEA producing tumours. Br. J. Cancer 87, 600–607 (2002).
Spooner, R. A. et al. A novel vascular endothelial growth factor-directed therapy that selectively activates cytotoxic prodrugs. Br. J. Cancer 88, 1622–1630 (2003).
Niculescu-Duvaz, I., Spooner, R., Marais, R. & Springer, C. J. Gene-directed enzyme prodrug therapy. Bioconjug. Chem. 9, 4–22 (1998).
Green, N. K. et al. Immune enhancement of nitroreductase-induced cytotoxicity: studies using a bicistronic adenovirus vector. Int. J. Cancer 104, 104–112 (2003).
Okabe, S., Arai, T., Yamashita, H. & Sugihara, K. Adenovirus-mediated prodrug-enzyme therapy for CEA-producing colorectal cancer cells. J. Cancer Res. Clin. Oncol. 129, 367–373 (2003).
Xu, G. & McLeod, H. L. Strategies for enzyme/prodrug cancer therapy. Clin. Cancer Res. 7, 3314–3324 (2001).
Minton, N. P. et al. Chemotherapeutic tumour targeting using clostridial spores. FEMS Microbiol. Rev. 17, 357–364 (1995).
Polak, A., Eschenhof, A., Fernex., M. & Scholer, H. J. Metabolic studies with 5-fluorocytosine-6–14C in mouse, rat, rabbit, dog and man. Chemotherapy 22, 137–153 (1976).
Anlezark, G. M. et al. The bioactivation of 5-(aziridin-1-y1)-2,4-dinitrobenzamide (CB1954). I. Purification and properties of a nitroreductase enzyme from Escherichia coli: a potential enzyme for antibody–directed enzyme prodrug therapy (ADEPT). Biochem. Pharmacol. 44, 2289–2295 (1992).
Mauchline, M. L., Davis, T. O. & Minton, N. P. in Manual of Industrial Microbiology and Biotechnology, 2nd Edition (eds Demain, A. L. & Davies, J. E.) 475–490 (Washington DC, ASM Press, 1999).
Lemmon, M. J. et al. Anaerobic bacteria as a gene delivery system that is controlled by the tumor microenvironment. Gene Ther. 4, 791–796 (1997).
Lambin, P. et al. Colonisation of the Clostridium in the body is restricted to hypoxic and necrotic areas of tumours. Anaerobe 4, 183–188 (1998).
Fox, M. E. et al. Anaerobic bacteria as a delivery system for cancer gene therapy: in vitro activation of 5-fluorocytosine by genetically engineered Clostridia. Gene Ther. 3, 173–178 (1996).
Theys, J. et al. Specific targeting of cytosine deaminase to solid tumors by engineered Clostridium acetobutylicum. Cancer Gene Ther. 8, 294–297 (2001).
Liu, S. C., Minton, N. P., Giaccia, A. J. & Brown, M. J. Anticancer efficacy of systemically delivered anaerobic bacteria as gene therapy vectors targeting tumor hypoxia/necrosis. Gene Ther. 9, 291–296 (2002).
Dang, L. H., Bettegowda, C., Huso, D. L., Kinzler, K. W. & Vogelstein, B. Combination bacteriolytic therapy for the treatment of experimental tumors. Proc. Natl Acad. Sci. USA 98, 15155–15160 (2001).
Nuyts, S. et al. The use of radiation-induced bacterial promoters in anaerobic conditions: a means to control gene expression in clostridium-mediated therapy for cancer. Radiat. Res. 155, 716–723 (2001).
Nuyts, S. et al. Radio-responsive recA promoter significantly increases TNFα production in recombinant clostridia after 2 Gy irradiation. Gene Ther. 8, 1197–1201 (2001).
Nuyts, S. et al. Insertion or deletion of the Cheo box modifies radiation inducibility of Clostridium promoters. Appl. Environ. Microbiol. 67, 4464–4470 (2001).
Jorgensen, H. & Overgaard, J. Combretastatins novel vascular targeting drugs for improving anti-cancer therapy. Combretastatins and conventional therapy. Adv. Exp. Med. Biol. 476, 311–323 (2000).
Theys, J. et al. Improvement of Clostridium tumour targeting vectors evaluated in rat rhabdomyosarcomas. FEMS Immunol. Med. Microbiol. 30, 37–41 (2001).
Liu, S. C. et al. Tumour-specific enzyme/prodrug gene therapy using genetically engineered C. sporogenes as a delivery system combined with vascular-targeting agents. Fourth International Conference on the Molecular Biology and Pathogenesis of the Clostridia, Woods Hole, Massachusetts (2003).
Fujimori, M., Amano, J. & Taniguchi, S. The genus Bifidobacterium for cancer gene therapy. Curr. Opin. Drug Discov. Devel. 5, 200–203 (2002).
Grove, J. I. et al. Generation of Escherichia coli nitroreductase mutants conferring improved cell sensitization to the prodrug CB1954. Cancer Res. 63, 5532–5537 (2003).
Anlezark, G. M. et al. The Bacillus amyloliquefaciens orthologue of B. subtilis ywrO encodes a nitroreductase enzyme which activates the prodrug CB 1954. Microbiology 148, 297–306 (2002).
Spencer, D. I. R. et al. A strategy for mapping and neutralizing conformational immunogenic sites on protein therapeutics. Proteomics 3, 271–279 (2002).
Pawelek, J. M., Low, K. B. & Bermudes, D. Tumor-targeted Salmonella as a novel anticancer vector. Cancer Res. 57, 4537–4544 (1997).
Low, K. B. et al. Lipid A mutant Salmonella with suppressed virulence and TNFα induction retain tumor-targeting in vivo. Nature Biotechnol. 17, 37–41 (1999).
Toso, J. F. et al. Phase I study of the intravenous administration of attenuated Salmonella typhimurium to patients with metastatic melanoma. J. Clin. Oncol. 20, 142–152 (2002).
Zheng, L. M. et al. Tumor amplified protein expression therapy: Salmonella as a tumor-selective protein delivery vector. Oncol. Res. 12, 127–135 (2000).
Cunningham, C. & Nemunaitis, J. A phase I trial of genetically modified Salmonella typhimurium expressing cytosine deaminase (TAPET-CD, VNP20029) administered by intratumoral injection in combination with 5-fluorocytosine for patients with advanced or metastatic cancer. Protocol no: CL–017. Hum. Gene Ther. 12, 1594–1596 (2001).
Yazawa, K., Fujimori, M., Amano, J., Kano, Y. & Taniguchi, S. Bifidobacterium longum as a delivery system for cancer gene therapy: selective localization and growth in hypoxic tumors. Cancer Gene Ther. 7, 269–274 (2000).
Li, X. et al. Bifidobacterium adolescentis as a delivery system of endostatin for cancer gene therapy: selective inhibitor of angiogenesis and hypoxic tumor growth. Cancer Gene Ther. 10, 105–111 (2003).
Acknowledgements
The author wishes to thank Nicola Minion for typing this manuscript, Ben Minton for the initial artwork and the financial support of the European Union.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Related links
Related links
DATABASES
LocusLink
Swiss Prot
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Minton, N. Clostridia in cancer therapy. Nat Rev Microbiol 1, 237–242 (2003). https://doi.org/10.1038/nrmicro777
Issue Date:
DOI: https://doi.org/10.1038/nrmicro777
This article is cited by
-
Clostridium Bacteria: Harnessing Tumour Necrosis for Targeted Gene Delivery
Molecular Diagnosis & Therapy (2024)
-
Dysbiosis of skin microbiome and gut microbiome in melanoma progression
BMC Microbiology (2022)
-
Recent trends and advances in microbe-based drug delivery systems
DARU Journal of Pharmaceutical Sciences (2019)
-
White paper on microbial anti-cancer therapy and prevention
Journal for ImmunoTherapy of Cancer (2018)
-
The expanding role of prodrugs in contemporary drug design and development
Nature Reviews Drug Discovery (2018)