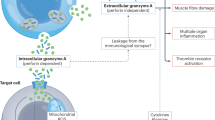
Key Points
-
Cytotoxic T lymphocytes (CTLs) kill their targets by both caspase-dependent and caspase-independent pathways.
-
Redundant cell-death pathways provide protection from viruses or tumours that can evade one pathway or another, such as caspase-resistant tumours that overexpress BCL-2.
-
CTLs and natural killer (NK) cells use the same mechanisms to induce cell death, releasing cytotoxic granules into the synapse formed between the CTL and its target; however, they have different profiles of expression of cytolytic molecules.
-
Granzyme B, which activates the caspases, can also act as an initiator or effector caspase by directly cleaving BID (BH3-interacting domain death agonist) to activate a mitochondrial apoptotic pathway or by cleaving the inhibitor of caspase-activated DNase (ICAD) to activate DNA damage.
-
Granzyme A, the most abundant granzyme, which is expressed by both innate and adaptive immune cytotoxic cells, activates a new caspase-independent cell-death pathway that targets a newly described cytoplasmic complex (the SET complex) to damage DNA by single-stranded nicks.
-
Granzyme C activates yet another cell-death pathway, which is just beginning to be elucidated, that induces prominent mitochondrial swelling and dysfunction.
-
The mechanism used by the membrane-perturbing protein perforin to deliver the granzymes to the cytosol of target cells is still not well understood.
-
Granulysin, a membrane-perturbing protein in human (but not mouse) CTL and NK-cell granules, causes microbial and mammalian cell death by disrupting bacterial and mitochondrial membranes.
Abstract
Granule exocytosis is the main pathway for the immune elimination of virus-infected cells and tumour cells by cytotoxic T lymphocytes and natural killer cells. After target-cell recognition, release of the cytotoxic granule contents into the immunological synapse formed between the killer cell and its target induces apoptosis. The granules contain two membrane-perturbing proteins, perforin and granulysin, and a family of serine proteases known as granzymes, complexed with the proteoglycan serglycin. In this review, I discuss recent insights into the mechanisms of granule-mediated cytotoxicity, focusing on how granzymes A, B and C and granulysin activate cell death through caspase-independent pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Russell, J. H. & Ley, T. J. Lymphocyte-mediated cytotoxicity. Annu. Rev. Immunol. 20, 323–370 (2002).
Kagi, D. et al. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin-deficient mice. Nature 369, 31–37 (1994).
Balkow, S. et al. Concerted action of the FasL/Fas and perforin/granzyme A and B pathways is mandatory for the development of early viral hepatitis but not for recovery from viral infection. J. Virol. 75, 8781–8791 (2001).
Trambas, C. M. & Griffiths, G. M. Delivering the kiss of death. Nature Immunol. 4, 399–403 (2003).
Griffiths, G. M. & Isaaz, S. Granzymes A and B are targeted to the lytic granules of lymphocytes by the mannose-6-phosphate receptor. J. Cell Biol. 120, 885–896 (1993).
Bromley, S. K. et al. The immunological synapse. Annu. Rev. Immunol. 19, 375–396 (2001).
Grakoui, A. et al. The immunological synapse: a molecular machine controlling T cell activation. Science 285, 221–227 (1999).
Dustin, M. L. & Chan, A. C. Signaling takes shape in the immune system. Cell 103, 283–294 (2000).
Davis, D. M. et al. The human natural killer cell immune synapse. Proc. Natl Acad. Sci. USA 96, 15062–15067 (1999).
Vyas, Y. M. et al. Spatial organization of signal transduction molecules in the NK cell immune synapses during MHC class I-regulated noncytolytic and cytolytic interactions. J. Immunol. 167, 4358–4367 (2001).
Davis, D. M. Assembly of the immunological synapse for T cells and NK cells. Trends Immunol. 23, 356–363 (2002).
Estebanez-Perpina, E. et al. Crystal structure of the caspase activator human granzyme B, a proteinase highly specific for an Asp-P1 residue. Biol. Chem. 381, 1203–1214 (2000).
Waugh, S. M., Harris, J. L., Fletterick, R. & Craik, C. S. The structure of the pro-apoptotic protease granzyme B reveals the molecular determinants of its specificity. Nature Struct. Biol. 7, 762–765 (2000).
Sun, J. et al. Importance of the P4' residue in human granzyme B inhibitors and substrates revealed by scanning mutagenesis of the PI-9 reactive center loop. J. Biol. Chem. 276, 15177–15184 (2001).
Rotonda, J. et al. The three-dimensional structure of human granzyme B compared to caspase-3, key mediators of cell death with cleavage specificity for aspartic acid in P1. Chem. Biol. 8, 357–368 (2001).
Beresford, P. J. et al. Granzyme A activates an endoplasmic reticulum-associated caspase-independent nuclease to induce single-stranded DNA nicks. J. Biol. Chem. 276, 43285–43293 (2001).
Kelso, A. et al. The genes for perforin, granzymes A–C and IFN-γ are differentially expressed in single CD8+ T cells during primary activation. Int. Immunol. 14, 605–613 (2002).
Irmler, M. et al. Granzyme A is an interleukin 1β-converting enzyme. J. Exp. Med. 181, 1917–1922 (1995).
Sower, L. E. et al. Extracellular activities of human granzyme A. Monocyte activation by granzyme A versus α-thrombin. J. Immunol. 156, 2585–2590 (1996).
Suidan, H. S. et al. The serine protease granzyme A does not induce platelet aggregation but inhibits responses triggered by thrombin. Biochem. J. 315, 939–945 (1996).
Suidan, H. S. et al. Granzyme A released upon stimulation of cytotoxic T lymphocytes activates the thrombin receptor on neuronal cells and astrocytes. Proc. Natl Acad. Sci. USA 91, 8112–8116 (1994).
Brunner, G., Simon, M. M. & Kramer, M. D. Activation of pro-urokinase by the human T cell-associated serine proteinase HuTSP-1. FEBS Lett. 260, 141–144 (1990).
Metkar, S. S. et al. Cytotoxic cell granule-mediated apoptosis: perforin delivers granzyme B–serglycin complexes into target cells without plasma membrane pore formation. Immunity 16, 417–428 (2002).
Raja, S. M. et al. Cytotoxic cell granule-mediated apoptosis: Characterization of the macromolecular complex of granzyme B with serglycin. J. Biol. Chem. 277, 49523–49530 (2002). A strong biophysical study of the granzyme-B–serglycin interaction.
Balaji, K. N., Schaschke, N., Machleidt, W., Catalfamo, M. & Henkart, P. A. Surface cathepsin B protects cytotoxic lymphocytes from self-destruction after degranulation. J. Exp. Med. 196, 493–503 (2002). This study describes a mechanism for protecting cytotoxic T lymphocytes (CTLs) from their own granule contents released into the synapse during granule exocytosis.
Dupuis, M., Schaerer, E., Krause, K. H. & Tschopp, J. The calcium-binding protein calreticulin is a major constituent of lytic granules in cytolytic T lymphocytes. J. Exp. Med. 177, 1–7 (1993).
Andrin, C. et al. Interaction between a Ca2+-binding protein calreticulin and perforin, a component of the cytotoxic T-cell granules. Biochem. 37, 10386–10394 (1998).
Fraser, S. A., Karimi, R., Michalak, M. & Hudig, D. Perforin lytic activity is controlled by calreticulin. J. Immunol. 164, 4150–4155 (2000).
Fraser, S. A., Michalak, M., Welch, W. H. & Hudig, D. Calreticulin, a component of the endoplasmic reticulum and of cytotoxic lymphocyte granules, regulates perforin-mediated lysis in the hemolytic model system. Biochem. Cell Biol. 76, 881–887 (1998).
Bossi, G. & Griffiths, G. M. Degranulation plays an essential part in regulating cell surface expression of Fas ligand in T cells and natural killer cells. Nature Med. 5, 90–96 (1999).
Wagner, L. et al. β-chemokines are released from HIV-1-specific cytolytic T-cell granules complexed to proteoglycans. Nature 391, 908–911 (1998).
Darmon, A. J., Nicholson, D. W. & Bleackley, R. C. Activation of the apoptotic protease CPP32 by cytotoxic T-cell-derived granzyme B. Nature 377, 446–448 (1995).
Sarin, A. et al. Target cell lysis by CTL granule exocytosis is independent of ICE/Ced-3 family proteases. Immunity 6, 209–215 (1997).
Henkart, P. A., Williams, M. S., Zacharchuk, C. M. & Sarin, A. Do CTL kill target cells by inducing apoptosis? Semin. Immunol. 9, 135–144 (1997).
Chiu, V. K., Walsh, C. M., Liu, C. C., Reed, J. C. & Clark, W. R. Bcl-2 blocks degranulation but not Fas-based cell-mediated cytotoxicity. J. Immunol. 154, 2023–2032 (1995).
Schroter, M., Lowin, B., Borner, C. & Tschopp, J. Regulation of Fas(Apo-1/CD95)- and perforin-mediated lytic pathways of primary cytotoxic T lymphocytes by the protooncogene bcl-2. Eur. J. Immunol. 25, 3509–3513 (1995).
Sutton, V. R., Vaux, D. L. & Trapani, J. A. Bcl-2 prevents apoptosis induced by perforin and granzyme B, but not that mediated by whole cytotoxic lymphocytes. J. Immunol. 158, 5783–5790 (1997).
Trapani, J. A. et al. Efficient nuclear targeting of granzyme B and the nuclear consequences of apoptosis induced by granzyme B and perforin are caspase-dependent, but cell death is caspase-independent. J. Biol. Chem. 273, 27934–27938 (1998).
Beresford, P. J., Xia, Z., Greenberg, A. H. & Lieberman, J. Granzyme A loading induces rapid cytolysis and a novel form of DNA damage independently of caspase activation. Immunity 10, 585–594 (1999). The first study to show that granzyme A activates a new caspase-independent cell-death pathway with features of apoptosis.
Thomas, D. A., Du, C., Xu, M., Wang, X. & Ley, T. J. DFF45/ICAD can be directly processed by granzyme B during the induction of apoptosis. Immunity 12, 621–632 (2000).
Kuwana, T. et al. Bid, Bax and lipids cooperate to form supramolecular openings in the outer mitochondrial membrane. Cell 111, 331–342 (2002).
Barry, M. et al. Granzyme B short-circuits the need for caspase 8 activity during granule-mediated cytotoxic T-lymphocyte killing by directly cleaving Bid. Mol. Cell. Biol. 20, 3781–3794 (2000). The first of several independent studies (references 41–43) to show that granzyme B directly activates mitochondrial pathways of cell death by targeting the BCL-2-family member BID.
Heibein, J. A. et al. Granzyme B-mediated cytochrome c release is regulated by the Bcl-2 family members Bid and Bax. J. Exp. Med. 192, 1391–1402 (2000).
Sutton, V. R. et al. Initiation of apoptosis by granzyme B requires direct cleavage of Bid, but not direct granzyme B-mediated caspase activation. J. Exp. Med. 192, 1403–1414 (2000).
Alimonti, J. B., Shi, L., Baijal, P. K. & Greenberg, A. H. Granzyme B induces BID-mediated cytochrome c release and mitochondrial permeability transition. J. Biol. Chem. 276, 6974–6982 (2001).
Metkar, S. S. et al. Granzyme B activates procaspase-3 which signals a mitochondrial amplification loop for maximal apoptosis. J. Cell Biol. 160, 875–885 (2003).
Wang, G. Q. et al. Resistance to granzyme B-mediated cytochrome c release in Bak-deficient cells. J. Exp. Med. 194, 1325–1337 (2001).
Pinkoski, M. J. et al. Granzyme B-mediated apoptosis proceeds predominantly through a Bcl-2-inhibitable mitochondrial pathway. J. Biol. Chem. 276, 12060–12067 (2001).
MacDonald, G., Shi, L., Vande Velde, C., Lieberman, J. & Greenberg, A. H. Mitochondria-dependent and -independent regulation of granzyme B-induced apoptosis. J. Exp. Med. 189, 131–144 (1999).
Thomas, D. A., Scorrano, L., Putcha, G. V., Korsmeyer, S. J. & Ley, T. J. Granzyme B can cause mitochondrial depolarization and cell death in the absence of BID, BAX and BAK. Proc. Natl Acad. Sci. USA 98, 14985–14990 (2001).
Sharif-Askari, E. et al. Direct cleavage of the human DNA fragmentation factor-45 by granzyme B induces caspase-activated DNase release and DNA fragmentation. EMBO J. 20, 3101–3113 (2001).
Li, L. Y., Luo, X. & Wang, X. Endonuclease G is an apoptotic DNase when released from mitochondria. Nature 412, 95–99 (2001).
Froelich, C. J. et al. Granzyme B/perforin-mediated apoptosis of Jurkat cells results in cleavage of poly(ADP-ribose) polymerase to the 89-kDa apoptotic fragment and less abundant 64-kDa fragment. Biochem. Biophys. Res. Commun. 227, 658–665 (1996).
Andrade, F. et al. Granzyme B directly and efficiently cleaves several downstream caspase substrates: implications for CTL-induced apoptosis. Immunity 8, 451–460 (1998).
Zhang, D., Beresford, P. J., Greenberg, A. H. & Lieberman, J. Granzymes A and B directly cleave lamins and disrupt the nuclear lamina during granule-mediated cytolysis. Proc. Natl Acad. Sci. USA 98, 5746–5751 (2001).
Shi, L., Kraut, R. P., Aebersold, R. & Greenberg, A. H. A natural killer cell granule protein that induces DNA fragmentation and apoptosis. J. Exp. Med. 175, 553–566 (1992).
Shi, L., Kam, C. M., Powers, J. C., Aebersold, R. & Greenberg, A. H. Purification of three cytotoxic lymphocyte granule serine proteases that induce apoptosis through distinct substrate and target cell interactions. J. Exp. Med. 176, 1521–1529 (1992).
Nakajima, H., Park, H. L. & Henkart, P. A. Synergistic roles of granzymes A and B in mediating target cell death by rat basophilic leukemia mast cell tumors also expressing cytolysin/perforin. J. Exp. Med. 181, 1037–1046 (1995).
Heusel, J. W., Wesselschmidt, R. L., Shresta, S., Russell, J. H. & Ley, T. J. Cytotoxic lymphocytes require granzyme B for the rapid induction of DNA fragmentation and apoptosis in allogeneic target cells. Cell 76, 977–987 (1994).
Zajac, A. J., Dye, J. M. & Quinn, D. G. Control of lymphocytic choriomeningitis virus infection in granzyme B deficient mice. Virology 305, 1–9 (2003).
Mullbacher, A. et al. Granzyme A is critical for recovery of mice from infection with the natural cytopathic viral pathogen, ectromelia. Proc. Natl Acad. Sci. USA 93, 5783–5787 (1996).
Pereira, R. A., Simon, M. M. & Simmons, A. Granzyme A, a noncytolytic component of CD8+ cell granules, restricts the spread of herpes simplex virus in the peripheral nervous systems of experimentally infected mice. J. Virol. 74, 1029–1032 (2000).
Pham, C. T., MacIvor, D. M., Hug, B. A., Heusel, J. W. & Ley, T. J. Long-range disruption of gene expression by a selectable marker cassette. Proc. Natl Acad. Sci. USA 93, 13090–13095 (1996).
Shresta, S., Graubert, T. A., Thomas, D. A., Raptis, S. Z. & Ley, T. J. Granzyme A initiates an alternative pathway for granule-mediated apoptosis. Immunity 10, 595–605 (1999).
Johnson, H., Sorrano, L., Korsmeyer, S. J. & Ley, T. J. Cell death induced by granzyme C. Blood 101, 3093–3101 (2003). This paper shows that granzyme C activates a unique caspase-independent cell-death pathway with prominent mitochondrial features and single-stranded DNA damage.
Beresford, P. J., Kam, C. M., Powers, J. C. & Lieberman, J. Recombinant human granzyme A binds to two putative HLA-associated proteins and cleaves one of them. Proc. Natl Acad. Sci. USA 94, 9285–9290 (1997).
Fan, Z., Beresford, P. J., Oh, D. Y., Zhang, D. & Lieberman, J. Tumor suppressor NM23-H1 is a granzyme A-activated DNase during CTL-mediated apoptosis, and the nucleosome assembly protein SET is its inhibitor. Cell 112, 659–672 (2003). This paper describes the mechanism for caspase-independent single-stranded DNA damage activated by granzyme A.
Fan, Z., Beresford, P. J., Zhang, D. & Lieberman, J. HMG2 interacts with the nucleosome assembly protein SET and is a target of the cytotoxic T-lymphocyte protease granzyme A. Mol. Cell Biol. 22, 2810–2820 (2002).
Fan, Z. et al. Cleaving the oxidative repair protein Ape1 enhances cell death mediated by granzyme A. Nature Immunol. 4, 145–153 (2003).
Ma, D. et al. NM23-H1 and NM23-H2 repress transcriptional activities of nuclease-hypersensitive elements in the platelet-derived growth factor-A promoter. J. Biol. Chem. 277, 1560–1567 (2002).
Zhang, D. et al. Induction of rapid histone degradation by the cytotoxic T lymphocyte protease granzyme A. J. Biol. Chem. 276, 3683–3690 (2001).
Jans, D. A. et al. Nuclear targeting of the serine protease granzyme A (fragmentin-1). J. Cell Sci. 111, 2645–2654 (1998).
Jans, D. A., Jans, P., Briggs, L. J., Sutton, V. & Trapani, J. A. Nuclear transport of granzyme B (fragmentin-2). Dependence on perforin in vivo and cytosolic factors in vitro. J. Biol. Chem. 271, 30781–30789 (1996).
Trapani, J. A. et al. Perforin-dependent nuclear entry of granzyme B precedes apoptosis, and is not a consequence of nuclear membrane dysfunction. Cell Death Differ. 5, 488–496 (1998).
Wilharm, E. et al. Generation of catalytically active granzyme K from Escherichia coli inclusion bodies and identification of efficient granzyme K inhibitors in human plasma. J. Biol. Chem. 274, 27331–27337 (1999).
Babe, L. M., Yoast, S., Dreyer, M. & Schmidt, B. F. Heterologous expression of human granzyme K in Bacillus subtilis and characterization of its hydrolytic activity in vitro. Biotechnol. Appl. Biochem. 27, 117–124 (1998).
Edwards, K. M., Kam, C. M., Powers, J. C. & Trapani, J. A. The human cytotoxic T cell granule serine protease granzyme H has chymotrypsin-like (chymase) activity and is taken up into cytoplasmic vesicles reminiscent of granzyme B-containing endosomes. J. Biol. Chem. 274, 30468–30473 (1999).
Woodard, S. L. et al. Purification and characterization of lymphocyte chymase I, a granzyme implicated in perforin-mediated lysis. J. Immunol. 160, 4988–4993 (1998).
Smyth, M. J., Sayers, T. J., Wiltrout, T., Powers, J. C. & Trapani, J. A. Met-ase: cloning and distinct chromosomal location of a serine protease preferentially expressed in human natural killer cells. J. Immunol. 151, 6195–6205 (1993).
Sayers, T. J. et al. The restricted expression of granzyme M in human lymphocytes. J. Immunol. 166, 765–771 (2001).
Trapani, J. A. & Smyth, M. J. Functional significance of the perforin/granzyme cell death pathway. Nature Rev. Immunol. 2, 735–747 (2002).
Lowin, B., Beermann, F., Schmidt, A. & Tschopp, J. A null mutation in the perforin gene impairs cytolytic T lymphocyte- and natural killer cell-mediated cytotoxicity. Proc. Natl Acad. Sci. USA 91, 11571–11575 (1994).
Froelich, C. J. et al. New paradigm for lymphocyte granule-mediated cytotoxicity. J. Biol. Chem. 271, 29073–29079 (1996). This paper shows that the old model of perforin pores might not be correct, as granzyme B can enter cells, but not induce apoptosis, without perforin.
Shi, L. et al. Granzyme B (GraB) autonomously crosses the cell membrane and perforin initiates apoptosis and GraB nuclear localization. J. Exp. Med. 185, 855–866 (1997).
Pinkoski, M. J. et al. Entry and trafficking of granzyme B in target cells during granzyme B–perforin-mediated apoptosis. Blood 92, 1044–1054 (1998).
Browne, K. A. et al. Cytosolic delivery of granzyme B by bacterial toxins: evidence that endosomal disruption, in addition to transmembrane pore formation, is an important function of perforin. Mol. Cell. Biol. 19, 8604–8615 (1999).
Motyka, B. et al. Mannose 6-phosphate/insulin-like growth factor II receptor is a death receptor for granzyme B during cytotoxic T cell-induced apoptosis. Cell 103, 491–500 (2000). This paper shows that the receptor involved in intracellular trafficking of granzymes to the granules is also a cell-surface receptor for internalization of granzyme B.
Trapani, J. A. et al. A clathrin/dynamin- and mannose-6-phosphate receptor-independent pathway for granzyme B-induced cell death. J. Cell Biol. 160, 223–233 (2003). This paper shows that granzyme B can enter cells independently of clathrin-mediated endocytosis or the mannose-6-phosphate receptor.
Uellner, R. et al. Perforin is activated by a proteolytic cleavage during biosynthesis which reveals a phospholipid-binding C2 domain. EMBO J. 16, 7287–7296 (1997).
Masson, D., Peters, P. J., Geuze, H. J., Borst, J. & Tschopp, J. Interaction of chondroitin sulfate with perforin and granzymes of cytolytic T-cells is dependent on pH. Biochemistry 29, 11229–11235 (1990).
Kawasaki, Y. et al. Cell death-associated translocation of plasma membrane components induced by CTL. J. Immunol. 164, 4641–4648 (2000).
Stenger, S. et al. An antimicrobial activity of cytolytic T cells mediated by granulysin. Science 282, 121–125 (1998). This study shows that granulysin can kill mycobacteria.
Ernst, W. A. et al. Granulysin, a T cell product, kills bacteria by altering membrane permeability. J. Immunol. 165, 7102–7108 (2000).
Ochoa, M. T. et al. T-cell release of granulysin contributes to host defense in leprosy. Nature Med. 7, 174–179 (2001).
Dieli, F. et al. Granulysin-dependent killing of intracellular and extracellular Mycobacterium tuberculosis by Vγ9/Vδ2 T lymphocytes. J. Infect. Dis. 184, 1082–1085 (2001).
Canaday, D. H. et al. CD4+ and CD8+ T cells kill intracellular Mycobacterium tuberculosis by a perforin and Fas/Fas ligand-independent mechanism. J. Immunol. 167, 2734–2742 (2001).
Ma, L. L. et al. CD8 T cell-mediated killing of Cryptococcus neoformans requires granulysin and is dependent on CD4 T cells and IL-15. J. Immunol. 169, 5787–5795 (2002).
Anderson, D. H. et al. Granulysin crystal structure and a structure-derived lytic mechanism. J. Mol. Biol. 325, 355–365 (2003).
Gamen, S. et al. Granulysin-induced apoptosis. I. Involvement of at least two distinct pathways. J. Immunol. 161, 1758–1764 (1998).
Kaspar, A. A. et al. A distinct pathway of cell-mediated apoptosis initiated by granulysin. J. Immunol. 167, 350–356 (2001).
Pardo, J. et al. A role of the mitochondrial apoptosis-inducing factor in granulysin-induced apoptosis. J. Immunol. 167, 1222–1229 (2001).
Kuhn, J. R. & Poenie, M. Dynamic polarization of the microtubule cytoskeleton during CTL-mediated killing. Immunity 16, 111–121 (2002).
Stinchcombe, J. C., Bossi, G., Booth, S. & Griffiths, G. M. The immunological synapse of CTL contains a secretory domain and membrane bridges. Immunity 15, 751–761 (2001). This study shows by elegant confocal microscopy that the synapse of CTLs is divided into two domains — a secretory domain for granule exocytosis and a signalling domain.
Bossi, G. et al. The secretory synapse: the secrets of a serial killer. Immunol. Rev. 189, 152–160 (2002).
Dustin, M. L. & Colman, D. R. Neural and immunological synaptic relations. Science 298, 785–789 (2002).
Spaeny-Dekking, E. H. et al. Extracellular granzymes A and B in humans: detection of native species during CTL responses in vitro and in vivo. J. Immunol. 160, 3610–3616 (1998).
Acknowledgements
I thank D. Martinvalet, Z. Fan, L. Shi, T. Ley, P. Henkart and C. Froelich for sharing unpublished data, and the members of my laboratory for helpful discussions. This work was supported by a grant from the National Institutes of Health.
Author information
Authors and Affiliations
Related links
Related links
DATABASES
LocusLink
Glossary
- GRAFT-VERSUS-HOST DISEASE
-
(GVHD). An immune response mounted against the recipient of an allograft by immunocompetent donor T cells derived from the graft. Typically, it is seen in the context of allogeneic bone-marrow transplantation.
- CASPASES
-
A family of cytosolic proteases that contain a cysteine residue in the active site and that cleave their substrates after an aspartic-acid residue. They can be divided into pro-inflammatory caspases (caspases 1, 4, 5 and 11), which cleave and activate pro-inflammatory cytokines, and pro-apoptotic caspases, which cleave and activate pro-apoptotic substrates. Pro-apoptotic caspases comprise initiator caspases (caspases 2, 8 and 9), which, in turn, cleave and activate effector caspases (caspases 3, 6 and 7).
- TRYPTASE
-
An enzyme that (similar to trypsin) cuts after basic amino acids such as lysine and arginine.
- CHYMASE
-
A protease that (similar to chymotrypsin) cuts after hydrophobic amino acids.
- METASE
-
A protease that cuts after methionine residues.
- DEFENSINS
-
Small basic peptides produced by immune cells that are microbicidal and work by damaging bacterial membranes.
- KLENOW POLYMERASE
-
The large fragment of Escherichia coli DNA polymerase I produced after cleavage with subtilisin. The Klenow fragment has 5′→3′ polymerase activity and 3′→5′ exonuclease activity, but no 5′→3′ exonuclease activity. It is used to end-label free 3′ recessed ends of DNA.
- TERMINAL DEOXYNUCLEOTIDYL TRANSFERASE
-
(TdT). An enzyme expressed during lymphocyte development that adds nucleotides to the free 3′ end of DNA breaks. It is used to assay apoptosis by catalysing the addition of radiolabelled or biotinylated nucleotides to sites of DNA damage.
- ANNEXIN-V STAINING
-
Annexin V binds to phosphatidyl serine, which is normally located on the inner leaflet of the plasma membrane, but which flips to the outer layer during apoptosis. Annexin-V staining is often used as an indicator of apoptosis.
Rights and permissions
About this article
Cite this article
Lieberman, J. The ABCs of granule-mediated cytotoxicity: new weapons in the arsenal. Nat Rev Immunol 3, 361–370 (2003). https://doi.org/10.1038/nri1083
Issue Date:
DOI: https://doi.org/10.1038/nri1083
This article is cited by
-
Highly tailorable gellan gum nanoparticles as a platform for the development of T cell activator systems
Biomaterials Research (2022)
-
Moderate static magnetic fields enhance antitumor CD8+ T cell function by promoting mitochondrial respiration
Scientific Reports (2020)
-
Safety, tolerability, pharmacokinetics, and pharmacodynamics of the afucosylated, humanized anti-EPHA2 antibody DS-8895a: a first-in-human phase I dose escalation and dose expansion study in patients with advanced solid tumors
Journal for ImmunoTherapy of Cancer (2019)