Abstract
A long-acting depot formulation of olanzapine that sustains plasma olanzapine concentrations for over a month after a single injection is currently under development. This multicenter, open-label study explored D2 receptor occupancy of a fixed dose of olanzapine pamoate (OP) depot given every 4 weeks. Patients (nine male, five female) with schizophrenia or schizoaffective disorder previously stabilized on oral olanzapine were switched to OP depot 300 mg by intramuscular injection every 4 weeks for 6 months. No visitwise within-group significant changes were found in Brief Psychiatric Rating Scale Total or Clinical Global Impressions-Severity of Illness scores, although seven patients received oral olanzapine supplementation during the first four injection cycles. To minimize impact on D2 occupancy, positron emission tomography (PET) scans were not completed during injection cycles that required supplemental oral olanzapine. Two patients reported transient injection site adverse events, which did not result in discontinuation. The most frequently reported treatment-emergent adverse events were insomnia, aggravated psychosis, and anxiety. Mean striatal D2 receptor occupancy, as measured by [11C]-raclopride PET, was 69% on oral olanzapine (5–20 mg/day) and 50% (trough) on OP depot at steady state. Following an initial decline, occupancy returned to 84% of baseline oral olanzapine occupancy after six injections. Over the study period, D2 receptor occupancy and plasma olanzapine concentrations were significantly correlated (r=0.76, P⩽0.001). OP depot resulted in mean D2 receptor occupancy of approximately 60% or higher at the end of the 6-month study period, a level consistent with antipsychotic efficacy and found during treatment with oral olanzapine. However, supplemental oral olanzapine or another dosing strategy may be necessary to maintain adequate therapeutic response during the first few injection cycles.
Similar content being viewed by others
INTRODUCTION
Patients with schizophrenia often discontinue their antipsychotic medications despite the high efficacy of these drugs (Lieberman et al, 2005). Non-adherence to antipsychotic medications is a primary reason for psychotic relapse and rehospitalization (Perkins, 2002). Long-acting depot antipsychotic agents may improve long-term outcomes, especially among patients who frequently forget to take their medications or refuse treatment because of lack of insight. However, currently only one second-generation antipsychotic agent, risperidone, has a depot formulation available, and it must be administered once every 2 weeks (Gefvert et al, 2005; Kane et al, 2003; Remington et al, 2006).
For most antipsychotic drugs, a striatal dopamine D2 receptor occupancy of ⩾60% has been associated with therapeutic response in patients with schizophrenia (Kapur et al, 2000). This has led to the use of positron emission tomography (PET) to complement pharmacokinetic and dose–response clinical studies, to determine clinically effective doses and dosing intervals of oral and depot antipsychotic drugs (Mamo et al, 2004; Remington et al, 2006). Oral olanzapine, a second-generation antipsychotic agent, has shown dose-dependent striatal D2 receptor occupancy of 60–80% at usual clinical doses (10–20 mg/day) (Kapur et al, 1998). A long-acting depot formulation of olanzapine is currently under development, and pharmacokinetic data from initial clinical studies using this new formulation support a dosing interval of 2–4 weeks (data on file, Eli Lilly and Company). In an effort to complement these pharmacokinetic data with central in vivo occupancy studies, this open-label PET study was conducted using 300 mg olanzapine pamoate (OP) depot administered by intramuscular injection to patients with schizophrenia or schizoaffective disorder. Our primary objective was to determine whether D2 receptor occupancy was sustained for 4 weeks following injection of OP depot, to ascertain the feasibility of administering this formulation once every 4 weeks. Therefore, OP depot was given at 4-week dosing intervals for 6 months. Secondary objectives were to assess the efficacy and safety of this regimen of OP depot.
PATIENTS AND METHODS
Participants
Fourteen participants (nine male, five female) in this three-center (Toronto, ON, Canada; Pittsburgh, PA, USA; New York, NY, USA), open-label study (study code: F1D-EW-HGJW) were inpatients or outpatients, aged 18–50 years, who were diagnosed with DSM-IV schizophrenia or schizoaffective disorder, and who were stable on oral olanzapine treatment for the 4 weeks before study entry. Patients with a history of serious, unstable medical illnesses, who were pregnant or nursing at the time of the study, or had received depot antipsychotic within 6 months of study entry, were excluded. The study was approved by the respective local research ethics boards, and participants entered the study after signing an informed consent.
Study Design
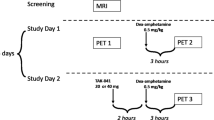
After a 1-week ‘lead-in’ period, in which participants continued to receive their prescribed dose of oral olanzapine (5–20 mg/day), baseline procedures (including plasma olanzapine concentrations and an [11C]-raclopride PET scan proximal to trough plasma concentrations) were completed. Patients then received one intramuscular injection of 300 mg OP depot every 4 weeks for the duration of the study period (6 months), with the total of six injection cycles. Supplementation with oral olanzapine (5–20 mg/day) was permitted to control any worsening of clinical status, as assessed by the investigator, but not during the 4-week injection cycle in which the patient received a PET scan. Benzodiazepines (<40 mg diazepam equivalents/day), benztropine mesylate (<6 mg/day), and anticholinergic were permitted during the study.
Determination of Plasma Olanzapine Concentrations
Sparse venous blood samples (5 ml) were taken from each patient at baseline (Week 0) and at various times throughout the study period including pre-injection troughs (at Week 4 of each cycle). Plasma olanzapine concentrations were estimated from heparinized plasma using high-performance liquid chromatography with electrochemical detection (HPLC-EC; BAS Analytics, West Lafayette, IN).
Positron Emission Tomography
To determine D2 receptor occupancy of olanzapine in vivo, an [11C]-raclopride PET scan of the striatum was performed at baseline (Week 0) plus not more than three of the following time points: 4, 8, 12, 16, 20, and 24 weeks before administration of the next depot injection. PET imaging was completed using standard protocols for [11C]-raclopride PET at the three respective sites. The PET scans after the first injection interval performed 4 weeks after the injection of OP depot reflect trough D2 occupancy levels for the interval before receiving the next injection of OP depot. The majority of the patients in this study were recruited and scanned at the Toronto site, the PET Centre of the Centre for Addiction & Mental Health (8/14 patients), thus the methods described below refer to the Toronto protocol. The numbers of patients at the Pittsburgh and New York sites were 4 and 2, respectively.
Three hundred seventy MBq of [11C]-raclopride (specific activity >11 100 Bq/mmol) were injected intravenously (bolus) and image acquisition was begun simultaneously. PET scanning was conducted with a 15-slice (8-ring) brain PET scanner (GEMS PC2048-Plus; General Electric Medical Systems, Milwaukee, WI) using the non-wobble acquisition mode. After a 10-min transmission scan for attenuation correction of the emission scans, emission scans were obtained every 1 min for the first 10 min, and then every 5 min for a total of 90 min. Scans were obtained while the patient was in the supine position with the head fixed using a thermoplastic face mask (Tru-Scan Imaging, Annapolis, MD), which allowed for repositioning between procedures. To permit accurate delineation of the brain regions for data analysis, each patient had a magnetic resonance image (MRI) scan carried out on a General Electric (GE)-Signal 1.5 Tesla scanner (GE LCC 9.4; General Electric Medical Systems, Milwaukee, WI). The image was acquired using two sequences: a conventional spin-echo pulse sequence (slice thickness, 3 mm), and a high-resolution gradient-echo pulse sequence (SPGR; slice thickness, 1.5 mm). PET and MRI images were then co-registered using Review 8.0 software to use the MRI for delineating regions of interest (ROIs) (Studholme et al, 1997).
ROIs used were the caudate and putamen, with the cerebellum used as a reference region. The ROI analysis was completed using Alice 3.1 (Perceptive Systems, Boulder, CO), which allows the rater to draw ROIs on summed PET images (representing averaged images of the dynamic time series). The ROIs were drawn by a single rater on two axial slices for the cerebellum (around the outermost border of cerebellar cortex), and two axial slices for the striatum. The ROIs were drawn such that their volume was larger than twice the full-width at half-maximum to minimize errors owing to partial volume effects (Volkow et al, 1993). The ROIs were then transferred to the dynamic PET images using the same software, and a time activity curve was generated and used in the analysis.
D2-receptor binding potential (BP) was calculated using previously described methods (Mamo et al, 2004). An average of the striatum/cerebellum ratio minus one obtained between 30 and 75 min of scanning was taken as a measure of the equilibrium BP (Farde et al, 1986). This timing was chosen because it has been shown that the ratio remains stable during this period, shows a very high correlation (r>0.95) with analytically derived estimates of D2 BP (Kapur et al, 1996), is highly reliable with a scan–rescan standard deviation of 6%, and has been standardized with a high intra- and inter-rater reliability (intra-class correlation coefficients: r>0.95) (Kapur et al, 1998). Receptor occupancy for a given dose was then calculated as the percentage reduction of receptor BP with the drug treatment compared with unmedicated baseline (100 × (1−(BPdrug scan/BPunmedicated baseline))). To ensure internal consistency, site-specific age-corrected measures of BP were obtained from a previously collected data set of antipsychotic-free healthy subjects. The data from these patients were analyzed by the same rater to ensure within-study consistency. The absence of the patients' own unmedicated baseline values introduces a potential error: for D2-receptor occupancy this error, as calculated on the basis of variance in the data from antipsychotic-naïve patients, is expected to vary from 0 to 9% for patients with 50% occupancy and 0–4% for patients who have 80% occupancy.
Psychiatric and Tolerability Assessments
Psychiatric stability was evaluated every 2 weeks using mean change from baseline to end point and visitwise scores on the Brief Psychiatric Rating Scale (BPRS; Overall, 1972) Total and the Clinical Global Impressions-Severity of Illness (CGI-S; Guy, 1976) scale. Tolerability was evaluated by monitoring for injection site reactions, treatment-emergent adverse events, and baseline-to-end point changes in vital signs (eg, blood pressure and ECGs) and weight. Severity of the extrapyramidal symptoms, parkinsonism and akathisia, were measured using the Simpson–Angus Scale and Barnes Akathisia Scale, respectively.
Statistical Analyses
Bivariate correlation analysis was used to examine the relationship between the primary variables of interest. Nonlinear regression analysis was used to evaluate the relationship between plasma olanzapine concentration and receptor occupancy (SPSS Inc., Chicago, IL) using the following equation: occupancy=Emax (plasma concentration/(plasma concentration+EC50)), where Emax is the maximum receptor occupancy and EC50 is the estimated plasma olanzapine concentration (ng/ml) associated with 50% maximal receptor occupancy. The plasma olanzapine concentration associated with 50% receptor occupancy (EC50) was determined.
For the BPRS Total and CGI-S mean changes, within-group t-tests were performed at each visit to test whether any of the changes were significantly different from zero. Baseline-to-end point changes on these scales were analyzed using the last-observation-carried-forward, whereas visitwise changes were evaluated with the observed-cases approach. Significant levels were set at P<0.05, with SAS Version 8 used. Visitwise within group t-tests were used to assess tolerability.
RESULTS
Baseline Characteristics and Disposition
Of the 20 patients who entered the study, six were excluded because the protocol entry criteria were not met and one, because of patient decision. Thus, 14 patients (five female) were enrolled into the study and received at least one injection of 300 mg OP depot. Their mean age (±SD) was 34.7±9.8 years, and the mean duration of illness (±SD) was 13.5±10.6 years. Half of the study sample were cigarette smokers. All patients were on oral olanzapine for a minimum of 4 weeks before study entry and the dose of their medication had not changed for at least 2 weeks before entry in study. Three patients discontinued, one each owing to the following: an adverse event (Week 16, seizure secondary to water intoxication in patient with historical diagnosis of epilepsy), patient decision (Week 8, personal decision), and physician decision (Week 4, continuous elevated creatine kinase (CK) laboratory values, the patient had a history of CK elevation with previous antipsychotic medication). Data for these patients were included up to the point of discontinuation.
Plasma Olanzapine Concentrations
At baseline, after the administration of oral olanzapine doses of 7.5, 10, 12.5, 15, and 20 mg, mean plasma olanzapine concentrations were 7.4, 22.3, 31.2, 18.6, and 67.9 ng/ml, respectively. Mean plasma olanzapine concentration across these doses was 37.4±31.2 ng/ml (range: 6.14–116.09 ng/ml; n=13, baseline data not available for one patient) with an average daily oral olanzapine dose at baseline of approximately 15 mg/day vs a mean OP depot dose of 10.7 mg/day. There was large individual variability in mean steady-state exposure. Mean plasma olanzapine concentrations initially declined, then appeared to reach steady-state by approximately Week 8, as suggested by the similarity of mean concentrations after this time through the duration of the study (Table 1). The mean exposure at steady-state after the administration of 300 mg/4 weeks OP depot was 20.3±11.2 ng/ml.
D2 Receptor Occupancy
All patients received a PET scan at baseline, although each patient could only receive four PET scans in total during the study. To minimize the effect of oral supplementation on the PET data, PET scans were not completed in OP depot dosing cycles that required oral supplementation. Two patients received scans at Week 1, five patients at Week 2, four at Week 3, and six at Week 4. Four patients received scans 4 weeks after the second (Week 8), third (Week 12), fourth (Week 16), fifth (Week 20), and sixth (Week 24) injections of depot.
The mean D2 occupancy at baseline was 69.1±15.2% (n=14), reflecting the occupancy of the oral olanzapine dose of the study group (15.2±4.8 mg/day). Over the entire study period, an expected curvilinear asymptotic significant relationship was found between plasma olanzapine concentrations and D2 receptor occupancy (r=0.76, P⩽0.001) (Figure 1). These data were fitted to the equation described in the Patients and Methods section to yield a concentration associated with 50% of maximal occupancy (EC50) of 11.0±1.3 ng/ml (mean±SE). The EC50 was estimated after pooling the data across investigator sites because no notable differences in parameter estimates were obtained when the data from each of the sites were fitted separately.
Percent D2 receptor occupancy as a function of plasma olanzapine concentrations at 4 Weeks after Injection of 300 mg OP Depot (N=14). The regression line has been fit to the rectangular hyperbole equation: occupancy=Emax × (plasma concentration/(plasma concentration+EC50)), where Emax is the maximum receptor occupancy with an estimated EC50 (±SD)=11.0±1.3 ng/ml.
During the first injection cycle, D2 receptor occupancy declined to approximately 70% of baseline values (occupancy change, mean±SD, %: 21.0±15.0) (Table 1; Figure 2), consistent with mean plasma olanzapine concentrations after the first OP depot dose being lower than the steady-state range of oral olanzapine concentrations. Beyond the second injection cycle, a gradual increase was observed in mean D2 occupancy, paralleling the accumulation of systemic olanzapine exposure, which reached a mean of 60% in the final two injection cycles. Thus, by the fifth injection cycle, the D2 occupancy returned to 84% of the baseline occupancy level on oral olanzapine.
Psychiatric Stability
Mean (±SD) change from baseline to end point (Week 24) was −2.3±9.5 on the BPRS Total and −0.07±1.21 on the CGI-S, none of which were statistically significant.
At baseline, mean (±SD) scores were 9.1±8.1 for the BPRS Total and 2.7±0.8 for the CGI-S. Upon visual inspection, the visitwise (biweekly) mean change scores on the BPRS Total showed some symptomatic worsening during the first 6 weeks of the study (1.4 points at 6 weeks; Figure 3); however, none of these changes were significantly different from zero. Visitwise mean change scores on the CGI-S were also not significantly different from zero (data not shown). It is worth emphasizing that the absence of change, as measured by the BPRS Total and the CGI-S, might reflect a contribution of supplemental oral olanzapine to the depot medication on an as needed basis during the first four injection cycles (described below).
Seven patients (18 total instances), in the opinion of the investigator, required supplemental oral olanzapine to address early signs of clinical deterioration. The need for supplemental oral olanzapine was generally restricted to the first three injection cycles (with the exception of two instances of oral supplementation in the fourth injection cycle). Of note, in two of these patients, oral olanzapine dosage was decreased approximately 25% within 3 weeks of study entry.
Tolerability
Insomnia, aggravated psychosis, and anxiety were the most frequently reported treatment-emergent adverse events (n=5 patients each). Dizziness and increased weight were spontaneously reported by three patients each. One patient reported an injection site granuloma, and another patient reported injection site induration, pain, and swelling. These injection site reactions were transient and did not result in discontinuation. Despite the fact that the patients had been stabilized on oral olanzapine for a minimum of 4 weeks before study entry, there was still a small net baseline-to-end point increase in weight (mean (±SD) 0.40±3.8 kg (NS)), with two patients reporting ⩾7% increase from baseline (one had received oral olanzapine for 416 days before study entry, and the other 37 days). Two patients experienced a significant decrease (3–31 mm Hg and −6–34 mm Hg) in orthostatic systolic blood pressure, and one patient experienced a potentially clinically significant prolonged ECG QTc interval (Bazett's; from 401 ms at baseline to 454 ms at Week 24). Mean (±SD) baseline-to-end point change on the Simpson–Angus Scale was −0.21±1.63, and on the Barnes Akathisia Scale, −0.14±0.53. One patient spontaneously reported akathisia at Week 12 (during the third injection cycle).
DISCUSSION
In this 6-month open-label study, we evaluated striatal dopamine D2 receptor occupancy, plasma olanzapine concentrations, and tolerability of 300 mg OP depot administered every 4 weeks for 6 months in patients with schizophrenia or schizoaffective disorder previously stabilized on oral olanzapine. Depot injection of 300 mg every 4 weeks resulted in mean steady-state plasma olanzapine concentrations of 20.3±11.2 ng/ml among these non-acutely ill patients, similar to the 23 ng/ml threshold that predicted response among acutely ill patients with schizophrenia treated with 2.5–17.5 mg/day oral olanzapine (Perry et al, 2001). The mean daily oral olanzapine dose at baseline was 15 mg, yielding a slightly higher mean plasma concentration across this range of 37.4±31.2 ng/ml. Thus, the slight discrepancy in steady-state plasma olanzapine concentrations is understandable. Consistently, D2 occupancy stabilized very close to baseline levels (mean occupancy was approximately 60% at 6 months compared with 70% at baseline). This reflects the asymptotic nonlinear relationship that is known to exist between plasma concentrations and D2 receptor occupancy with atypical and typical antipsychotic drugs, including olanzapine (Tauscher et al, 2002). These changes were mirrored by seven patients receiving oral supplementation within the first four injection cycles, suggesting central D2 occupancy generally required for antipsychotic efficacy (Nordström et al, 1993) was not attained in these patients until steady-state plasma concentrations were reached. Overall, the results of this study appear to support 4-week dosing intervals for 300 mg of OP depot, although some oral supplementation or alternative dosing strategy may be necessary to maintain adequate therapeutic response during the first few injections cycles. This slight drop in mean steady-state exposure, compared with the mean oral baseline concentration, is understandable given that the daily dose delivered by depot injection of 300 mg/4 weeks was approximately 10 mg/day.
During the first four injection cycles, seven patients received supplementation with oral olanzapine, although all patients were able to taper dosing within three injection cycles. No patients received oral supplementation at the time of PET scanning; thus, the occupancy data would not have been influenced. This supplemental dosing reflects physician-noted clinical deterioration, paralleling the trend toward general worsening on the BPRS Total within the first two injection cycles. This transient clinical deterioration could have been related to the initial decline in central D2 receptor occupancy, which was resolved by the increase in occupancy owing to supplementation. Oral olanzapine supplementation diminished when occupancy with OP depot treatment reached ⩾60% steady-state plasma concentrations during the fourth injection cycle. These data suggest that, although the 4-week dosing strategy was adequate beyond the first four injection cycles, a dose of 300 mg OP depot every 4 weeks alone may be less than optimal for maintaining therapeutic response in patients stabilized on oral olanzapine. Thus, oral supplementation in the first three injection cycles may be necessary to maintain therapeutic response at the dose and dosing interval used in this study. Alternative strategies to address the decreased central occupancy during the first four injection cycles may include the administration of higher doses (eg, 400 mg/4 weeks) or increasing the frequency of depot dosing for the first three months (eg, giving the OP depot once every 2 weeks).
While this study is limited by the small number of patients compounded by sparse sampling across time (owing to the limit of four PET scans per patient), the use of a fixed dose within an open-label design, and the lack of active or placebo comparator, we do not believe that these limitations detract from the conclusions drawn. This study was completed in three different sites, introducing the possibility of inconsistent PET scanning acquisition methodology across the respective study sites. To address this, we estimated the olanzapine plasma concentration that would be expected to result in 50% occupancy (EC50) across the respective sites and found no appreciable differences between sites, suggesting that our results were not influenced by location. It should also be pointed out that because there were many more pharmacokinetics samples than D2 receptor scans, the plasma concentration and D2 occupancy data obtained at any given time point do not necessarily represent the same population. Finally, the conclusions drawn are only applicable to individuals who are stabilized on oral olanzapine for a minimum of 4 weeks before commencing OP depot treatment.
As this study involved a change in medication, it would have been ethically unreasonable not to allow for the use of anticholinergics and benzodiazepines on an as needed basis. It has previously been reported that lorazepam increases the BP of [11C]-raclopride in anesthetized baboons beyond test–retest values, a finding thought to be related to the release of endogenous dopamine (Dewey et al, 1992). Hence, in the current study, the occupancy results for patients receiving benzodiazepines may have been underestimated to a small degree. Similarly, it has previously been reported that scopolamine decreases the BP in healthy human volunteers (Dewey et al, 1993), giving rise to the possibility of over-estimation of occupancy for patients receiving anticholinergics.
Conclusion
Although future controlled studies are needed to evaluate the efficacy of dosing strategies, the results suggest that, in patients with schizophrenia or schizoaffective disorder previously stabilized on oral olanzapine for a minimum of 4 weeks, 300 mg OP depot administered intramuscularly every 4 weeks may be sufficient for therapeutic levels of central D2 receptor occupancy. However, our results also suggest that some patients may need supplemental oral olanzapine, at least within the first three injection cycles, to maintain optimal therapeutic response. Nonetheless, by the end of 6 months of treatment with OP depot, D2 receptor occupancy reached levels that are consistant with antipsychotic efficacy. Furthermore, both the D2 receptor occupancy attained and the tolerability profile of OP depot were consistent with those found during treatment with oral olanzapine, and, importantly, injection site adverse events were reported in only two patients and were transient and did not result in study discontinuation.
References
Dewey SL, Smith GS, Logan J, Brodie JD, Simkowitz P, MacGregor RR et al (1993). Effects of central cholinergic blockade on striatal dopamine release measured with positron emission tomography in normal human subjects. Proc Natl Acad Sci USA 90: 11816–11820.
Dewey SL, Smith GS, Logan J, Brodie JD, Yu DW, Ferrieri RA et al (1992). GABAergic inhibition of endogenous dopamine release measured in vivo with 11C-raclopride and positron emission tomography. J Neurosci 12: 3773–3780.
Farde L, Hall H, Ehrin E, Sedvall G (1986). Quantitative analysis of D2 dopamine receptor binding in the living human brain by PET. Science 231: 258–261.
Gefvert O, Eriksson B, Persson P, Helldin L, Bjorner A, Mannaert E et al (2005). Pharmacokinetics and D2 receptor occupancy of long-acting injectable risperidone (Risperdal Consta) in patients with schizophrenia. Int J Neuropsychopharmacol 8: 27–36.
Guy W (1976). ECDEU Assessment Manual for Psychopharmacology revised edition. US Department of Health, Education and Welfare: Bethesda, MD.
Kane JM, Eerdekens M, Lindenmayer JP, Keith SJ, Lesem M, Karcher K (2003). Long-acting injectable Risperidone: efficacy and satey of the first long-acting atypical antipsychotic. Am J Psychiatry 160: 1125–1132.
Kapur S, Zipursky R, Jones C, Remington G, Houle S (2000). Relationship between dopamine D(2) occupancy, clinical response, and side effects: a double-blind PET study of first-episode schizophrenia. Am J Psychiatry 157: 514–520.
Kapur S, Zipursky RB, Jones C, Remington GJ, Wilson AA, DaSilva J et al (1996). The D2 receptor occupancy profile of loxapine determined using PET. Neuropsychopharmacol 15: 562–566.
Kapur S, Zipursky RB, Remington G, Jones C, DaSilva J, Wilson AA et al (1998). 5-HT2 and D2 receptor occupancy of olanzapine in schizophrenia: a PET investigation. Am J Psychiatry 155: 921–928.
Lieberman JA, Stroup TS, McEvoy JP, Swartz MS, Rosenheck RA, Perkins DO et al (2005). Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 353: 1209–1223.
Mamo D, Sedman E, Tillner J, Sellers EM, Romach MK, Kapur S (2004). EMD 281014, a specific and potent 5HT2 antagonist in humans: a dose-finding PET study. Psychopharmacol 175: 382–388.
Nordström AL, Farde L, Wiesel FA, Forslund K, Pauli S, Halldin C et al (1993). Central D2-dopamine receptor occupancy in relation to antipsychotic drug effects: a double-blind PET study of schizophrenic patients. Biol Psychiatry 33: 227–235.
Overall JE (1972). The Brief Psychiatric Rating Scale in Psychopharmacology Research, Psychometric Laboratory Reports, No. 29. University of Texas: Galveston.
Perkins DO (2002). Predictors of noncompliance in patients with schizophrenia. J Clin Psychiatry 63: 1121–1128.
Perry PJ, Lund BC, Sanger T, Beasley C (2001). Olanzapine plasma concentrations and clinical response: acute phase results of the North American olanzapine trial. J Clin Psychopharmacol 21: 14–20.
Remington G, Mamo D, Labelle A, Reiss J, Shammi C, Mannaert E et al (2006). A PET study evaluating dopamine D2 receptor occupancy for long-acting injectable rsiperidone. Am J Psychiatry 163: 396–401.
Studholme C, Hill DL, Hawkes DJ (1997). Automated three-dimensional registration of magnetic resonance and positron emission tomography brain images by multiresolution optimization of voxel similarity measures. Med Phys 24: 25–35.
Tauscher J, Jones C, Remington G, Zipursky RB, Kapur S (2002). Significant dissociation of brain and plasma kinetics with antipsychotics. Mol Psychiatry 7: 317–321.
Volkow ND, Fowler JS, Wang GJ, Dewey SL, Schlyer D, MacGregor R et al (1993). Reproducibility of repeated measures of carbon-11-raclopride binding in the human brain. J Nucl Med 34: 609–613 (correction, 34(5):838).
Acknowledgements
We thank Chet Mathis, PhD, and Debra Montrose, PhD, of Western Psychiatric Institute and Clinic, Pittsburgh, PA, for their help with this study and Scott W Andersen, MS, of Eli Lilly and Company, for statistical consultation.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was sponsored by Eli Lilly and Company.
CONFLICT OF INTEREST
Drs Taylor, Kothare, and McDonnell are employees of Eli Lilly and Company and share-holders of Lilly stock; Dr Kapur has received grant support from Lilly, Astra Zeneca, Janssen, Neuromolecular Inc., Pfizer, and Wyeth Ayerst and has served as a speaker, consultant, or scientific advisor for Lilly, Astra Zeneca, Bristol Meyers Squibb, Glaxo Smith Kline, Janssen, Pfizer, Solvay Kingswood, and Wyeth Ayerst.
Rights and permissions
About this article
Cite this article
Mamo, D., Kapur, S., Keshavan, M. et al. D2 Receptor Occupancy of Olanzapine Pamoate Depot Using Positron Emission Tomography: An Open-label Study in Patients with Schizophrenia. Neuropsychopharmacol 33, 298–304 (2008). https://doi.org/10.1038/sj.npp.1301409
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.npp.1301409
Keywords
This article is cited by
-
Post-Injection Delirium/Sedation Syndrome in Patients Treated with Olanzapine Pamoate: Mechanism, Incidence, and Management
CNS Drugs (2015)
-
A pooled analysis of injection site-related adverse events in patients with schizophrenia treated with olanzapine long-acting injection
BMC Psychiatry (2014)
-
Post-injection delirium/sedation syndrome in patients with schizophrenia treated with olanzapine long-acting injection, I: analysis of cases
BMC Psychiatry (2010)