Abstract
Wistar–Kyoto (WKY) rats provide a model of stress-induced depressive behavior, because they show enhanced vulnerability to the effects of stressors. The present study examined differences in the behavioral response to different types of antidepressant drugs between WKY and Sprague–Dawley (SD) rats in the forced swimming test (FST). WKY rats displayed significantly greater immobility than SD rats during their exposure to the FST. The noradrenergic antidepressant, desipramine, produced a dose-dependent reduction of immobility and increase of climbing behavior in the SD rats. In WKY rats, desipramine reduced immobility at a lower dose and produced increases of both swimming and climbing behavior. The serotonergic compounds, fluoxetine and 8-OH-DPAT, produced dose-dependent reductions of immobility and increases of swimming behavior in the FST in SD rats, but the response to the serotonergic drugs were blunted in WKY rats. These results indicate that genetic or constitutive differences may determine the distinct behavioral profiles for antidepressant compounds with selective pharmacological effects in different rat strains, and these effects may be related to genetic heterogeneity of antidepressant responses in depressed patients.
Similar content being viewed by others
Main
There has been interest in performance differences between different rodent strains on behavioral tests for antidepressants, because such differences may identify underlying constitutive factors that contribute to the vulnerability to clinical depression in humans. A number of authors have reported robust interstrain differences both in baseline performances and the response to antidepressant drugs in animal behavioral tests that are sensitive to antidepressant drugs (Porsolt et al. 1978; Van der Heyden et al. 1987; Overstreet et al. 1992; Paré, 1992; Armario et al. 1995). One inbred rat strain, the Wistar–Kyoto (WKY) rat bred initially from the Wistar rat as the control strain for the spontaneously hypertensive rat or SHR (Okamoto and Aoki 1963), has been proposed as an animal model of depressive behavior (Paré 1989; Paré and Redei 1993; Marti and Armario 1996), because they consistently demonstrate exaggerated behavioral and physiological responses to stress across a variety of situations in comparison to other strains. The WKY strain is one of the most susceptible to developing learned helplessness (Wieland et al. 1986; Paré, 1994) and demonstrates higher levels of behavioral immobility at baseline in the forced swimming test (FST; Paré 1994; Armario et al. 1995), two rodent tests sensitive to antidepressant drugs. Also, WKY rats show greater signs of emotionality and a tendency to freeze in stressful situations (Berger and Starzic 1988), exhibit little exploratory behavior in the open field (Paré 1993; Paré 1996), and demonstrate greater hypo neophagia; that is, the fear of feeding in a novel environment when hungry, than other rat strains (Paré 1994). As expected from this profile, WKY rats readily acquire passive avoidance behaviors, but they demonstrate deficits when emitting active avoidance or anxiety-related behaviors (Paré 1994). Physiologically, the WKY strain is more susceptible to developing gastric ulceration to stress than other rat strains (Paré 1990, 1994), and they secrete higher levels of adrenocorticotropic hormone in response to restraint plus cold stress (Paré and Redei 1993).
The FST is a behavioral test in rodents that predicts the clinical efficacy of many types of antidepressant treatments (Porsolt et al 1977, 1978). This test induces the development of immobility when a rodent has been placed in a tank of deep water for an extended period of time and makes only those movements necessary to keep its head above water (Porsolt et al. 1977). The development of immobility is usually facilitated by prior exposure to a swimming pretest or other stressors (Borsini et al. 1989). Whether administered chronically or after brief subchronic administration, antidepressant drugs decrease the duration of immobility in the FST. The FST is sensitive to the effects of a number of major classes of antidepressant treatments and is also relatively selective, because few other psychoactive drugs produce similar behavioral effects (Borsini and Meli 1988). Recently, a behavior sampling technique was developed to score, in addition to immobility, active behaviors emitted by rats in the FST, such as swimming and climbing (Detke et al. 1995; Lucki 1997). Studies using this technique have shown that, in addition to changes in immobility, at least two distinct active behavioral patterns were produced by antidepressants that were probably mediated by different neurotransmitters (Lucki 1997). For example, selective NE reuptake inhibitors, such as desipramine and maprotiline, increased climbing without altering swimming; selective serotonin reuptake inhibitors (SSRIs), such as fluoxetine, paroxetine, and sertraline, increased swimming without affecting climbing, and drugs with mixed effects at norepinephrine and serotonin transporters were able to increase both patterns of active behavior (Detke et al. 1995; Reneric and Lucki 1998).
The present study compared behavioral responses of two rat strains in the FST, WKY rats and Sprague–Dawley (SD) rats, an outbred reference strain frequently used in laboratory tests with antidepressant drugs. Active behavioral response components were studied for both rat strains in the FST (Detke et al. 1995). The study examined potential differences between strains in response to pharmacologically different types of antidepressant drugs; either the selective norepinephrine reuptake inhibitor desipramine or the selective serotonin reuptake inhibitor fluoxetine. In addition, the selective 5-HT1A receptor agonist 8-hydroxy-2-(di-n-propylamino) tetralin (8-OH-DPAT; Wieland and Lucki 1990; Lucki et al. 1994; Detke et al. 1995) was studied. The purpose of this study was to measure the different active behaviors emitted by the two rat strains at baseline in the FST, compare the response to noradrenergic and serotonergic drugs between rat strains, and determine whether distinctive active response components to antidepressants in the FST demonstrated previously in SD rats were maintained in the WKY strain. The results showed that these rat strains not only demonstrate differences in baseline FST behavior, but also in the response to pharmacologically different types of antidepressant drugs in the FST.
MATERIALS AND METHODS
Animals
Male Sprague–Dawley and Wistar–Kyoto rats (Charles River, Wilmington, MA), weighing 150 to 175 g upon arrival, were housed in groups of three in polycarbonate cages. They were maintained on a 12-h alternating light–dark schedule (lights on 07:00–19:00 hours) in a temperature-controlled (22°C) colony room with free access to food and water. The animal facility was approved by the AALAC. Rats were handled for 3 to 5 days before behavioral testing. All housing and behavioral procedures conform to the Principles of Laboratory Animal Care issued by the National Institutes of Health, with local laws and regulations, and were approved by The University of Pennsylvania Institutional Animal Care and Use Committee.
Forced Swimming Test (FST)
The procedure used was previously described by Detke et al. (1995) and was very similar to that described previously by others (Porsolt et al. 1978; Borsini and Meli 1988), except that the water was deeper. Swim sessions were conducted by placing rats in individual glass cylinders (46 cm × 20 cm) containing 23 to 25°C water 30 cm deep, so rats could not support themselves by touching the bottom with their feet. Two swimming sessions were conducted, between 12:00 and 18:00 hours: an initial 15-min pretest followed 24 h later by a 5-min test. Drug treatments were administered during the period between the two sessions. Following both swimming sessions, the rats were removed from the cylinders, dried with paper towels, placed in heated cages for 15 min, and then returned to their home cages. Test sessions were videotaped from an overhead view (Panasonic color video camera and recorder) for scoring later.
Behavioral Scoring
A time-sampling technique was used to score several behaviors during a single viewing. This method has previously been described and shown to be reliable and valid for detecting the effects of different antidepressant drugs (Detke et al. 1995). At the end of each 5-s period during the test session, the scorer rated the rat's behavior as one of the following three categories: (1) immobility—floating in the water without struggling and making only those movements necessary to keep the head above water; (2) swimming—making active swimming motions, more than necessary to merely keep the head above water; that is, moving around in the cylinder; and (3) climbing—making active movements with the forepaws in and out of the water, usually directed against the walls. All behavioral scoring was done by a single rater, blind to the treatment condition.
Drugs
Drug treatments in the FST were administered with a subchronic pattern. Three injections were administered 23.5, 5, and 1 h before beginning the given test. All drugs were administered subcutaneously (SC) in a volume equivalent to 2 ml/kg. Doses were calculated as mg/kg base, dissolved in deionized water to enhance solubility, and freshly prepared each morning. The doses tested for each drug were: desipramine (5, 10, and 20 mg/kg), fluoxetine (5, 10, 20, and 40 mg/kg), and 8-OH-DPAT (0.25, 0.5, and 1.0 mg/kg). Control rats received deionized water.
± 8-hydroxy-2-(di-n-propylamino) tetralin HBr (8-OH-DPAT) was purchased from Research Biochemicals (Natick, MA). Desipramine HCl was purchased from Sigma Chemical Co. (St. Louis, MO). Fluoxetine HCl was obtained as a gift from Eli Lilly Co. (Indianapolis, IN).
Statistical Analysis
An experiment usually consisted of 32 subjects of the same strain assigned randomly to groups consisting of eight animals each. Each experiment included one control group. A two-way analysis of variance (ANOVA) was performed to analyze differences between strains of rats and drug treatments on the behavioral scores in the FST (immobility, swimming, and climbing). Fisher's PLSD test was used to compare the values of the control group to the other experimental groups. The results obtained separately with 40 mg/kg fluoxetine were analyzed using Student's t-test (two-tailed).
RESULTS
During the 15-min pretest, the WKY rats demonstrated high levels of immobility and low frequencies of the other active behaviors, climbing and swimming, virtually from the onset of the pretest session (data not shown). In contrast, SD rats demonstrated a maximum immobility response only after 10 to 15 min of the pretest and showed higher frequencies of both active behaviors during the pretest session. During the test sessions after treatment with saline, WKY rats demonstrated a significantly greater frequency of immobility and lower frequency of climbing than SD rats (see values for saline in Figures 1, 2, 3.)
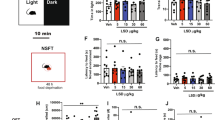
Effects of the selective norepinephrine reuptake inhibitor desipramine in the rat FST in SD and WKY rats. Mean counts (+ 1 SEM) of immobility, swimming, and climbing behaviors are shown when sampled every 5 s during the 5-min FST test period. n = 8 rats per group. After administration of the pretest, rats were injected with either saline (SAL, open bars) or varying doses desipramine 23.5, 5, and 1 h before the FST test period. Asterisks represent values following desipramine that differ significantly from the corresponding saline control group, * p < .05; ** p < .01. Crosses represent significant differences in baseline values between strains, p < .01.
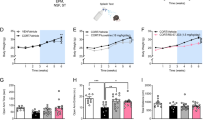
Effects of the selective serotonin reuptake inhibitor fluoxetine in the rat FST in SD and WKY rats. Mean counts (+ 1 SEM) of immobility, swimming, and climbing behaviors are shown when sampled every 5 s during the 5-min FST test period. n = 8 rats per group. After administration of the pretest, rats were injected with either saline (SAL, open bars) or varying doses fluoxetine 23.5, 5, and 1 h before the FST test period. Asterisks represent values following fluoxetine that differ significantly from the corresponding saline control group, ** p < .01. Crosses represent significant differences in baseline values between strains, p < .01.
Effects of the selective 5-HT1A receptor agonist 8-OH-DPAT in the rat FST in SD and WKY rats. Mean counts (+1 SEM) of immobility, swimming, and climbing behaviors are shown when sampled every 5 s during the 5-min FST test period. n = 8 rats per group. After administration of the pretest, rats were injected with either saline (SAL, open bars) or varying doses 8-OH-DPAT 23.5, 5, and 1 h before the FST test period. Asterisks represent values following 8-OH-DPAT that differ significantly from the corresponding saline control group, * p < .05; ** p < .01. Crosses represent significant differences in baseline values between strains, p < .01.
The effects of treatment with desipramine on the behaviors of SD and WKY rats in the FST are illustrated in Figure 1. Desipramine decreased the frequency of immobility in a dose-dependent manner in both strains. However, the effects were produced from different behavioral baselines, and desipramine reduced immobility significantly in WKY rats at a dose that was ineffective in SD rats. The profile of active behaviors showed that desipramine significantly increased climbing without affecting swimming in SD rats, as reported previously (Detke et al. 1995). In contrast, desipramine increased both swimming and climbing behaviors in WKY rats. The results of the two-way ANOVA test for immobility, swimming, and climbing behaviors for rats treated with desipramine are shown in Table 1 The statistical analysis revealed significant main effects of Treatment and Strain for immobility, a trend toward a Treatment × Strain interaction for swimming and significant main effects for Treatment, Strain, and their interaction for climbing.
The effects of administering fluoxetine to SD and WKY rats in the FST are shown in Figure 2. Fluoxetine significantly decreased immobility and increased swimming without affecting climbing behavior in SD rats, as reported previously (Detke et al. 1995). In WKY rats, however, fluoxetine did not alter immobility significantly, and only the highest dose of fluoxetine (20 mg/kg) increased swimming behavior significantly. The results of the two-way ANOVA test (Table 1) revealed significant main effects of Treatment and Strain for immobility and swimming and significant main effects for Treatment, Strain, and their interaction for climbing. Because of the lack of effect of fluoxetine in WKY rats at doses that were behaviorally active in SD rats, a separate group of WKY rats (n = 9) was tested at a higher dose of fluoxetine (40 mg/kg). These results (immobility: 46 ± 3.4 (mean ± 1 SEM), 0.05 < p < .10 vs. saline; swimming: 9.8 ± 2.56, p > .05; climbing: 4.2 ± 1.3, 0.05 < p < .10) indicate that the behavioral insensitivity continued, even at a relatively high test dose of fluoxetine.
Finally, behavior changes in the FST produced by the 5-HT1A receptor agonist 8-OH-DPAT in SD and WKY rats are shown in Figure 3. 8-OH-DPAT decreased immobility and increased swimming behavior without affecting climbing in SD rats. In contrast, 8-OH-DPAT did not produce any significant behavioral effect in WKY rats at any dose tested. The results from the two-way ANOVA (Table 1) showed a significant main effect for Strain and Treatment × Strain interaction for immobility and a trend for a significant main effect for Treatment, a significant effect for Strain, and significant Treatment × Strain interaction for swimming. Post hoc comparisons (Figure 3) indicated that 8-OH-DPAT changed these behaviors in SD but not in WKY rats. Climbing behavior demonstrated only a significant main effect for Strain, because 8-OH-DPAT did not alter the frequency of this behavior.
DISCUSSION
The purpose of this study was to measure behavioral differences between WKY and SD rats in the FST and to compare their behavioral responses to different types of antidepressant drugs. WKY rats are a behavioral model for depressive behavior, because they are prone to adopt passive coping strategies in response to stress and, therefore, may model one aspect of clinical depression in humans (Paré and Redei 1993; Paré 1994; see Introduction). In this study, rats from both strains reacted differently when submitted to forced swimming, in agreement with previous reports (Paré 1989; Marti and Armario 1996). WKY rats developed behavioral immobility more rapidly and displayed fewer active behaviors during the pretest period than SD rats, and these strain differences were reflected in consistently different baseline values during the test period for immobility and climbing behavior, but not consistently for swimming.
The two strains not only differed in baseline FST behavior but also in their response to pharmacologically different types of antidepressant drugs in the FST. The noradrenergic antidepressant desipramine produced a dose-dependent reduction of immobility and an increase of climbing in SD rats, in agreement with previous studies (Detke et al. 1995; Lucki 1997). Desipramine also produced a reduction of immobility in WKY rats, but these effects were shown relative to the higher baseline value of immobility shown by the WKY rats. The behavioral response of WKY rats after treatment with desipramine was similar to SD rats that did not receive drug treatment, although it would be unwise to characterize this behavioral change as a “normalization.” Although changes in sensitivity to desipramine were difficult to quantify with differing baseline values, a lower dose of desipramine was behaviorally effective in WKY rats than it was in SD rats. Corresponding with a reduction of immobility, desipramine increased the frequency of both climbing and swimming behaviors. The increased swimming behavior was a prominent component of the response of WKY rats to desipramine, possibly because it was the predominate active behavior under these conditions; whereas, it played no role in the response of SD rats to desipramine. Because increased swimming behavior is the response pattern usually produced by SSRIs in SD rats, it is possible that the altered response pattern of WKY rats to desipramine could involve serotonergic substrates in this strain. Enthusiasm for serotonergic involvement was diminished by the insensitivity of WKY rats to the behavioral effects of serotonergic compounds, such as fluoxetine and 8-OH-DPAT, in the FST, although some interaction between noradrenergic and serotonergic systems might still mediate this effect.
These results are similar to the antidepressant-like effects of chronic desipramine treatment in the FST in WKY rats reported by Paré (1992), who rated floating and struggling behaviors in the FST. However, these results disagree with those of Lahmame, Armario, and associates. These investigators reported that WKY rats were subsensitive in their response to the acute administration of desipramine or imipramine in the FST, although they did respond to chronic treatment with imipramine (15 mg/kg, once daily for 14 days) (Lahmame and Armario 1996; Lahmame et al. 1997). A number of procedural differences might contribute to the discrepant results between Lahmame and those of Paré and ourselves, although both laboratories use FST scoring procedures that measure active behaviors. For example, the present study and Paré (1992) used a deeper water depth (30 cm) than Lahmame (18 cm) in the test cylinder. A deeper water depth would increase the frequency of active behaviors and prevent rats from supporting themselves by touching the bottom of the cylinder (Detke and Lucki 1995); whereas, a shallow water depth would facilitate the development of adaptive postural positions by touching the bottom of the cylinder, especially in WKY rats, which favor passive behavioral coping strategies. The shallow water depth would inhibit WKY rats from engaging in active behaviors that were a prominent component of their antidepressant response in the present study. In addition, Lahmame used an acute dosing procedure without the benefit of a pretest, which would be expected to be less sensitive for detecting antidepressant behavioral effects (Borsini et al. 1989). Different suppliers of WKY rats (Charles River, U.S. or France) could also account for differences between studies. It would be interesting to examine changes in the FST in WKY rats produced by chronic administration of different types of antidepressants (c.f. Detke et al. 1997), because chronic administration of imipramine altered FST performance in WKY rats (Lahmame et al. 1997).
Although WKY rats were responsive to desipramine, WKY rats showed a significantly reduced response to the serotonergic antidepressant fluoxetine, causing only small and inconsistent effects of swimming behavior up to 40 mg/kg. In contrast, fluoxetine produced a dose-dependent reduction of immobility and increase of swimming behavior in SD rats, as observed previously (Detke et al. 1995; Lucki 1997). These results suggest that different rat strains may demonstrate great variability in the behavioral response to antidepressants according to their pharmacological selectivity.
The effects of fluoxetine are likely to involve the release of serotonin, because prior depletion of serotonin prevents the behavioral effects of fluoxetine but not desipramine (Page et al. 1999, in press). Although it is likely that a number of different 5-HT receptors may participate in the therapeutic effects of SSRIs, it is known that 5-HT1A receptor agonists produce behavioral responses similar to those of established antidepressants (Wieland and Lucki 1990; Lucki et al. 1994). However, the antidepressant-like action of 8-OH-DPAT was also reduced in WKY rats. This suggests that insensitivity to the behavioral effects of serotonergic antidepressants is likely to involve more than changes in the release of serotonin and involve postsynaptic receptor activation and signaling. However, the refractory response of WKY rats to the serotonergic compounds may be related preferentially to stress, because WKY and SD rats respond equally to other responses produced by 8-OH-DPAT, such as hypothermia and the serotonin syndrome (unpublished data).
The underlying neural substrates associated with the depressive behavioral characteristics and altered antidepressant drug sensitivity shown by WKY rats is unknown. Among the most salient neurochemical changes reported was that WKY rats show a significantly higher density of noradrenergic transporter sites and augmented changes in β- and α2-adrenergic receptors in response to stress, as compared to SD rats (Tejani-Butt et al. 1994). A number of stressors seem to alter brain noradrenergic activity (Valentino et al. 1993), and it is possible that altered noradrenergic activity could underlie the behavioral responses of WKY rats or their response to desipramine. Exposure to the FST can also alter the density of monoamine receptors and their regulation by antidepressant drug treatments (Duncan et al. 1985; Paul et al. 1990). However, there were no major differences in the densities of 5-HT1A, 5-HT2 or β-adrenergic receptors reported in WKY rats when compared with other rat strains, either at baseline, in response to stress, or after drug treatment (Paré and Tejani-Butt 1996; Lahmame et al. 1997). Exposure to the FST is also known to produce changes in the release of dopamine, norepinephrine, and serotonin in a variety of brain regions, and these effects interact with antidepressant drug treatments (Rosetti et al. 1993; Kirby and Lucki 1997). It is possible that the regulation of monoamine release by stressors and antidepressant drugs differs between rat strains, and these effects account for the ability of desipramine to counteract the effects of stress in WKY rats.
Finally, it may also be important to consider that WKY rats seem constitutively more sensitive to stress than other rat strains (Paré and Redei 1993). The enhanced response of WKY rats to stress may also generate exaggerated levels of plasma ACTH and corticosterone secretion evoked by the FST when compared to SD rats (Rittenhouse, Lopez-Rubalcava, and Lucki unpublished data). Because serotonin is involved in stimulating the hypothalamic–pituitary axis and corticosterone exerts multiple regulatory actions on different components of the serotonergic system, it is possible that an interaction between stress hormones and the serotonergic system could account for the decreased sensitivity of WKY rats to the antidepressant actions of serotonergic compounds.
In conclusion, genetic differences that model characteristics of depressive behavior can distinctly alter the behavioral effects of antidepressant drugs with selective pharmacological actions. In the present study, WKY rats showed enhanced depressive behavior (increased immobility and decreased climbing behavior) in the FST, which was sensitive to the tricyclic antidepressant desipramine but insensitive to serotonergic antidepressants. WKY rats possibly model a subtype of depressive behavior, characterized by enhanced psychomotor retardation and response to stress, which demonstrates enhanced sensitivity to noradrenergic antidepressants and resistance toward serotonergic antidepressants. There is evidence from precipitated relapse studies supporting distinguishable neurochemical contributions to the clinical efficacy of antidepressant drugs according to their neuropharmacological substrates (Heninger et al. 1996). There is also evidence for considerable genetic heterogeneity of human norepinephrine and serotonin transporters, which has been associated with variability of innate behavioral and neurochemical characteristics (Lesch 1997; Jonsson et al. 1998). Differential sensitivity to the behavioral effects of desipramine and fluoxetine by different rat strains, such as WKY rats, may provide important information concerning the underlying substrates that might contribute to a selective clinical response in some depressed patients and resistance to treatment in others.
References
Armario A, Gavalda A, Marti J . (1995): Comparison of the behavioral and endocrine response to forced swimming stress in five inbred strains of rats. Psychoneuroendocrinology 20: 879–890
Berger DF, Starzic JJ . (1988): Contrasting lever-press avoidance behavior of spontaneously hypertensive and normotensive rats (rattus norvegicus). J Comp Psychol 102: 279–286
Borsini F, Lecci A, Sessarego A, Frassine R, Meli A . (1989): Discovery of antidepressant activity by forced swimming test may depend on pre-exposure of rats to a stressful situation. Psychopharmacology 97: 183–188
Borsini F, Meli A . (1988): Is the forced swimming test a suitable model for revealing antidepressant activity? Psychopharmacology 94: 147–160
Detke MJ, Johnson J, Lucki I . (1997): Acute and chronic antidepressant drug treatment in the rat forced swimming test model of depression. Exp Clin Psychopharmacol 5: 107–112
Detke MJ, Lucki I . (1995): Detection of serotonergic and noradrenergic antidepressants in the rat forced swimming test: The effects of water depth. Behav Brain Res 73: 43–46
Detke MJ, Rickels M, Lucki I . (1995): Active behaviors in the rat forced swimming test differentially produced by serotonergic and noradrenergic antidepressants. Psychopharmacology 121: 66–72
Duncan GE, Paul IA, Harden TK, Mueller RA, Stumpf WE, Breese GR . (1985): Rapid down-regulation of beta adrenergic receptors by combining antidepressant drugs with forced swim: A model of antidepressant-induced neural adaptation. J Pharmacol Exp Ther 234: 402–408
Heninger GR, Delgado PL, Charney DS . (1996): The revised monoamine theory of depression: A modulatory role for monoamines, based on new findings from monoamine depletion experiments in humans. Pharmacopsychiatry 29: 2–11
Jonsson EG, Nothen MM, Gustavsson JP, Neidt H, Bunzel R, Propping P, Sedvall GC . (1998): Polymorphisms in the dopamine, serotonin, and norepinephrine transporter genes and their relationships to monoamine metabolite concentrations in CSF of healthy volunteers. Psychiat Res 79: 1–9
Kirby LG, Lucki I . (1997): Interaction between the forced swimming test and fluoxetine treatment on extracellular 5-hydroxytryptamine and 5-hydroxyindoleacetic acid in the rat. J Pharmacol Exp Ther 282: 967–976
Lahmame A, Armario A . (1996): Differential responsiveness of inbred strains of rats to antidepressants in the forced swimming test: Are Wistar–Kyoto rats an animal model of subsensitivity to antidepressants? Psychopharmacology 123: 191–198
Lahmame A, del Arco C, Pazos A, Yritia M, Armario A . (1997): Are Wistar–Kyoto rats a genetic animal model of depression resistant to antidepressants? Eur J Pharmacol 337: 115–123
Lesch K-P . (1997): Molecular biology, pharmacology, and genetics of the serotonin transporter: Psychobiological and clinical implications. In Baumgarten HG, Gothert M (eds), Serotoninergic Neurons and 5-HT Receptors in the CNS. Berlin, Springer. pp 670–705
Lucki I . (1997): The forced swimming test as a model for core and component behavioral effects of antidepressant drugs. Behav Pharmacol 8: 523–532
Lucki I, Singh A, Kreiss DS . (1994): Antidepressant-like behavioral effects of serotonin receptor agonists. Neurosci Biobehav Rev 18: 85–95
Marti J, Armario A . (1996): Forced swimming behavior is not related to corticosterone levels achieved in the test. A study with four inbred rat strains. Physiol Behav 59: 369–372
Okamoto K, Aoki K . (1963): Development of a strain of spontaneously hypertensive rats. Japan Circ J 27: 282–293
Overstreet DH, Rezvani AH, Janowsky DS . (1992): Genetic animal models of depression and ethanol preference provide support for cholinergic and serotonergic involvement in depression and alcoholism. Biol Psychiat 31: 919–936
Page ME, Detke MJ, Dalvi A, Kirby LG, Lucki I . (1999): Serotonergic mediation of the effects of fluoxetine, but not desipramine, in the rat forced swimming test. Psychopharmacology in press.
Paré WP . (1989): Behavioral despair test predicts ulceration in WKY rats. Physiol Behav 46: 483–487
Paré WP . (1990): Technique and strain comparisons in stress ulcer. Ann NY Acad Sci 597: 223–230
Paré WP . (1992): Learning behavior, escape behavior, and depression in an ulcer-susceptible rat strain. Integrat Physiol Behav Sci 27: 130–141
Paré WP . (1993): Passive-avoidance behavior in Wistar–Kyoto (WKY), Wistar, and Fischer-344 rats. Physiol Behav 54: 845–852
Paré WP . (1994): Open field, learned helplessness, defensive burying, and forced-swim tests in WKY rats. Physiol Behav 55: 433–439
Paré WP . (1996): Enhanced retrieval of unpleasant memories influenced by shock controllability, shock sequence, and rat strain. Biol Psychiat 39: 808–813
Paré WP, Redei E . (1993): Depressive behavior and stress ulcer in Wistar–Kyoto rats. J Physiol (Paris) 87: 229–238
Paré WP, Tejani-Butt SM . (1996): Effect of stress on the behavior and 5-HT system in Sprague–Dawley and Wistar–Kyoto rat strains. Integr Physiol Behav Sci 31: 112–121
Paul IA, Duncan GE, Kuhn C, Mueller RA, Hong JS, Breese GR . (1990): Neural adaptation in imipramine-treated rats processed in forced swim test: Assessment of time course, handling, rat strain, and amine uptake. J Pharmacol Exp Ther 252: 997–1005
Porsolt RD, Le Pichon M, Jalfre M . (1977): Depression: A new model sensitive to antidepressant treatments. Nature 266: 730–732
Porsolt RD, Anton G, Deniel M, Jalfre M . (1978): Behavioral despair in rats: A new model sensitive to antidepressant treatments. Eur J Pharmacol 47: 379–391
Rénéric J-P, Lucki I . (1998): Antidepressant behavioral effects by dual inhibition of monoamine reuptake in the rat forced swimming test. Psychopharmacology 136: 190–197
Rosetti ZL, Lai M, Hmaidan Y, Gessa GL . (1993): Depletion of mesolimbic dopamine during behavioral despair: Partial reversal by chronic imipramine. Eur J Pharmacol 242: 313–315
Tejani-Butt SM, Paré WP, Yang J . (1994): Effect of repeated novel stressors on depressive behavior and brain norepinephrine receptor system in Sprague–Dawley and Wistar–Kyoto (WKY) rats. Brain Res 649: 27–35
Valentino RJ, Foote SL, Page ME . (1993): The locus coeruleus as a site for integrating corticotropin-releasing factor and noradrenergic mediation of stress responses. Ann NY Acad Sci 697: 173–188
Van der Heyden JAM, Molewijk E, Olivier B . (1987): Strain differences in response to drugs in the tail suspension test for antidepressant activity. Psychopharmacology 92: 127–130
Wieland S, Boren JL, Consroe PF, Martin A . (1986): Stock differences in the susceptibility of rats to learned helplessness. Life Sci 39: 937–944
Wieland S, Lucki I . (1990): Antidepressant-like activity of 5-HT1A agonists measured with the forced swim test. Psychopharmacology 101: 497–504
Acknowledgements
We greatly appreciate the comments of Drs. Wade Berrettini, Michelle Page, and Ashutosh Dalvi on a previous version of this manuscript. CL-R was a Pan-American fellow jointly supported by the National Institute of Health (USA) and CONACyT (Mexico). Other parts of this research were supported by USPHS Grant MH 36262.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López-Rubalcava, C., Lucki, I. Strain Differences in the Behavioral Effects of Antidepressant Drugs in the Rat Forced Swimming Test. Neuropsychopharmacol 22, 191–199 (2000). https://doi.org/10.1016/S0893-133X(99)00100-1
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S0893-133X(99)00100-1
Keywords
This article is cited by
-
Positive maternal affect during mother–litter interaction is reduced in new mother rats exhibiting a depression-like phenotype
Scientific Reports (2023)
-
Habenula as a Possible Target for Treatment-Resistant Depression Phenotype in Wistar Kyoto Rats
Molecular Neurobiology (2023)
-
Persistent muscle hyperalgesia after adolescent stress is exacerbated by a mild-nociceptive input in adulthood and is associated with microglia activation
Scientific Reports (2022)
-
The Role of Glucocorticoid Hormones in the Stress-Protective Effects of Hypoxic Postconditioning in Models of Depression and Post-Traumatic Stress Disorder in Rats
Neuroscience and Behavioral Physiology (2021)
-
Cell encapsulation enhances antidepressant effect of the mesenchymal stem cells and counteracts depressive-like behavior of treatment-resistant depressed rats
Molecular Psychiatry (2020)