Abstract
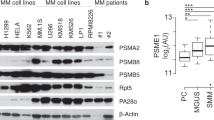
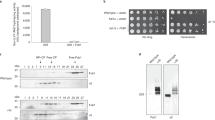
Proteasome inhibitors, such as the dipeptide boronic acid bortezomib, are emerging as important tools in the treatment of the fatal hematologic malignancy multiple myeloma. Despite the recent US Food and Drug Administration approval of bortezomib (PS341, Velcade) for the treatment of refractory multiple myeloma, many of the basic pharmacologic parameters of bortezomib and its mode of action on myeloma cells remain to be determined. We describe the synthesis and use of a cell-permeant active site–directed probe, which allows profiling of proteasomal activities in living cells. When we compared proteasome activity patterns in cultured cells and crude cell extracts with this probe, we observed substantial differences, stressing the importance for bioassays compatible with live cells to ensure accuracy of such measurements. Using this probe, we investigated the in vivo subunit specificities of bortezomib and another inhibitor, MG132.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hideshima, T. et al. The proteasome inhibitor PS-341 inhibits growth, induces apoptosis, and overcomes drug resistance in human multiple myeloma cells. Cancer Res. 61, 3071–3076 (2001).
Rock, K.L. & Goldberg, A.L. Degradation of cell proteins and the generation of MHC class I–presented peptides. Annu. Rev. Immunol. 17, 739–797 (1999).
Glickman, M.H. & Ciechanover, A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol. Rev. 82, 373–428 (2002).
Kisselev, A.F. & Goldberg, A.L. Proteasome inhibitors: from research tools to drug candidates. Chem. Biol. 8, 739–758 (2001).
Adams, J. et al. Proteasome inhibitors: a novel class of potent and effective antitumor agents. Cancer Res. 59, 2615–2622 (1999).
Dahlmann, B., Ruppert, T., Kloetzel, P.M. & Kuehn, L. Subtypes of 20S proteasomes from skeletal muscle. Biochimie 83, 295–299 (2001).
Driscoll, J., Brown, M.G., Finley, D. & Monaco, J.J. MHC-linked LMP gene products specifically alter peptidase activities of the proteasome. Nature 365, 262–264 (1993).
Gaczynska, M., Rock, K.L. & Goldberg, A.L. Gamma-interferon and expression of MHC genes regulate peptide hydrolysis by proteasomes. Nature 365, 264–267 (1993).
Bogyo, M. et al. Covalent modification of the active site threonine of proteasomal beta subunits and the Escherichia coli homolog HslV by a new class of inhibitors. Proc. Natl Acad. Sci. USA 94, 6629–6634 (1997).
Greenbaum, D., Medzihradszky, K.F., Burlingame, A. & Bogyo, M. Epoxide electrophiles as activity-dependent cysteine protease profiling and discovery tools. Chem. Biol. 7, 569–581 (2000).
Liu, Y., Patricelli, M.P. & Cravatt, B.F. Activity-based protein profiling: the serine hydrolases. Proc. Natl Acad. Sci. USA 96, 14694–14699 (1999).
Adam, G.C., Cravatt, B.F. & Sorensen, E.J. Profiling the specific reactivity of the proteome with non-directed activity-based probes. Chem. Biol. 8, 81–95 (2001).
Adam, G.C., Sorensen, E.J. & Cravatt, B.F. Proteomic profiling of mechanistically distinct enzyme classes using a common chemotype. Nat. Biotechnol. 20, 805–809 (2002).
Kessler, B.M. et al. Extended peptide-based inhibitors efficiently target the proteasome and reveal overlapping specificities of the catalytic beta-subunits. Chem. Biol. 8, 913–929 (2001).
Ovaa, H. et al. Chemistry in living cells: detection of active proteasomes by a two-step labeling strategy. Angew. Chem. Int. Edn. Engl. 42, 3626–3629 (2003).
Dantuma, N.P., Lindsten, K., Glas, R., Jellne, M. & Masucci, M.G. Short-lived green fluorescent proteins for quantifying ubiquitin/proteasome-dependent proteolysis in living cells. Nat. Biotechnol. 18, 538–543 (2000).
Hideshima, T. et al. The proteasome inhibitor PS-341 inhibits growth, induces apoptosis, and overcomes drug resistance in human multiple myeloma cells. Cancer Res. 61, 3071–3076 (2001).
Bogyo, M., Shin, S., McMaster, J.S. & Ploegh, H.L. Substrate binding and sequence preference of the proteasome revealed by active-site-directed affinity probes. Chem. Biol. 5, 307–320 (1998).
Garcia-Echeverria, C. Recent advances in the identification and development of 20S proteasome inhibitors. Mini Rev. Med. Chem. 2, 247–259 (2002).
Lightcap, E.S. et al. Proteasome inhibition measurements: clinical application. Clin. Chem. 46, 673–683 (2000).
Stein, R.L., Melandri, F. & Dick, L. Kinetic characterization of the chymotryptic activity of the 20S proteasome. Biochemistry 35, 3899–3908 (1996).
Fiebiger, E., Story, C., Ploegh, H.L. & Tortorella, D. Visualization of the ER-to-cytosol dislocation reaction of a type I membrane protein. EMBO J. 21, 1041–1053 (2002).
Lindsten, K., Menendez-Benito, V., Masucci, M.G. & Dantuma, N.P. A transgenic mouse model of the ubiquitin/proteasome system. Nat. Biotechnol. 21, 897–902 (2003).
Luker, G.D., Pica, C.M., Song, J., Luker, K.E. & Piwnica-Worms, D. Imaging 26S proteasome activity and inhibition in living mice. Nat. Med. 9, 969–973 (2003).
Elliott, P.J., Soucy, T.A., Pien, C.S., Adams, J. & Lightcap, E.S. Assays for proteasome inhibition. Methods Mol. Med. 85, 163–172 (2003).
Acknowledgements
The authors thank M.A. Leeuwenburgh for providing AdaAhx3L3VS. This work was financially supported by the National Institutes of Health (H.L.P. and P.J.G.), a grant from AstraZeneca (H.L.P. and P.J.G.), the Netherlands Organization for Scientific Research (H.O.), a National Cancer Institute SPORE grant Career Developmental Award (H.O.), the Dutch Cancer Society (H.O.), a Multiple Myeloma Research Foundation Senior Research Award (B.M.K.), the Dr. Saal van Zwanenberg Stichting (M.V.), the Austrian Academy of Sciences (E.F.), the Koninklijke Hollandsche Maatschappij der Wetenschappen (C.R.B.), and by the Stichting Fonds Doctor Catharine van Tussenbroek (C.R.B.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
H.P. and P.J.G. are supported in part through a grant from the Oncology Division of AstraZeneca. No reagents or potential products from AstraZeneca were used in this study. At the present time, AstraZeneca does not stand to benefit in any way from the publication of data contained herein.
Supplementary information
Supplementary Fig. 1
Incubation of HeLa cells with inhibitor 3. (PDF 50 kb)
Supplementary Fig. 2
H-H COSY NMR spectrum of inhibitor 3 in CDCl3 (PDF 77 kb)
Supplementary Fig. 3
1H-NMR spectrum of inhibitor 3 in CDCl3 (PDF 24 kb)
Supplementary Fig. 4
13C (APT) NMR spectrum of inhibitor 3 in CDCl3 (PDF 16 kb)
Rights and permissions
About this article
Cite this article
Berkers, C., Verdoes, M., Lichtman, E. et al. Activity probe for in vivo profiling of the specificity of proteasome inhibitor bortezomib. Nat Methods 2, 357–362 (2005). https://doi.org/10.1038/nmeth759
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth759
This article is cited by
-
Interplay between proteasome inhibitors and NF-κB pathway in leukemia and lymphoma: a comprehensive review on challenges ahead of proteasome inhibitors
Cell Communication and Signaling (2024)
-
A proteasomal β5 subunit of Haemonchus contortus with a role in the growth, development and life span
Parasites & Vectors (2023)
-
Dual network analysis of transcriptome data for discovery of new therapeutic targets in non-small cell lung cancer
Oncogene (2023)
-
Functional and quantitative evaluation of the 20S proteasome in serum and intracellular in145 moroccan patients with hematologic malignancies
Clinical Proteomics (2022)
-
Pharmacological targeting of endoplasmic reticulum stress in disease
Nature Reviews Drug Discovery (2022)