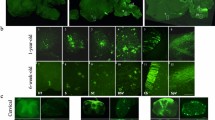
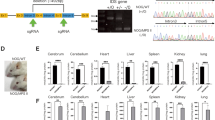
Abstract
Metachromatic leukodystrophy (MLD) is a lipidosis caused by deficiency of arylsulfatase A (ARSA). Although the genetics of MLD are known, its pathophysiology is not understood. The disease leads to progressive demyelination and early death and no effective treatment is available. We used lentiviral vectors to deliver a functional ARSA gene (human ARSA) into the brain of adult mice with germ-line inactivation of the mouse gene encoding ARSA, As2. We report sustained expression of active enzyme throughout a large portion of the brain, with long-term protection from development of neuropathology and hippocampal-related learning impairments. We show that selective degeneration of hippocampal neurons is a central step in disease pathogenesis, and provide evidence that in vivo transfer of ARSA by lentiviral vectors reverts the disease phenotype in all investigated areas. Therefore, in vivo gene therapy offers a unique option for MLD and other storage diseases affecting the central nervous system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kolodney, E.H. & Fluharty A.L. Metachromatic Leukodistrophy and multiple sulfatase deficiency: Sulfatide lipidosis. in The Metabolic and Molecular Bases of Inherited Disease 6th edn. (eds. Scriver, C.R., Beaudet, A.L, Sly W.S. & Valle, D.) 2693–7391 (McGraw-Hill, New York, 1995).
Neufeld, E.F. Lysosomal disease. Annu. Rev. Biochem 60, 257–280 (1991).
Kapaun, P. et al. A slow progression of juvenile metachromatic leukodystrophy 6 years after bone marrow transplantation. J. Child Neurol. 14, 222–228 (1999).
Krivit, W., Peters, C. & Shapiro, E.G. Bone marrow transplantation as effective treatment of central nervous system disease in globoid cell leukodystrophy, metachromatic leukodystrophy, adrenoleukodystrophy, mannosidosis, fucosidosis, aspartylglucosaminuria, Hurler, Maroteaux-Lamy, and Sly syndromes, and Gaucher disease type III. Curr. Opin. Neurol. 12, 167–176 (1999).
Ohashi, T., Eto, Y., Learish, R. & Barranger, J.A. Correction of enzyme deficiency in metachromatic leukodystrophy fibroblasts by retroviral-mediated transfer of the human arylsulphatase A gene. J. Inherit. Metab. Dis. 16, 881–885 (1993).
Learish, R. et al. Retroviral gene transfer and sustained expression of human arylsulfatase A. Gene. Ther. 3, 343–349 (1996).
Sangalli, A. et al. Transduced fibroblasts and metachromatic leukodystrophy lymphocytes transfer arylsulfatase A to myelinating glia and deficient cells in vitro. Hum. Gene. Ther. 9, 2111–2119 (1998).
Penzien, J.M. et al. Compound heterozygosity for metachromatic leukodystrophy and arylsulfatase A pseudodeficiency alleles is not associated with progressive neurological disease. Am. J. Hum. Genet. 52, 557–564 (1993).
Leinekugel, P., Michel, S., Conzelmann, E. & Sandhoff, K. Quantitative correlation between the residual activity of β-hexosaminidase A and arylsulfatase A and the severity of the resulting lysosomal storage disease. Hum. Genet. 88, 513–523 (1992).
Hess, B. et al. Phenotype of arylsulfatase A-deficient mice: relationship to human metachromatic leukodystrophy. Proc. Natl. Acad. Sci. USA 93, 14821–14826 (1996).
Gieselmann, V. et al. Metachromatic leukodystrophy: molecular genetics and an animal model. J. Inherit. Metab. Dis. 21, 564–574 (1998).
Naldini, L. et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 272, 263–267 (1996).
Naldini, L., Blomer, U., Gage, F.H., Trono, D. & Verma, I.M. Efficient transfer, integration, and sustained long-term expression of the transgene in adult rat brains injected with a lentiviral vector. Proc. Natl. Acad. Sci. USA 93, 11382–11388 (1996).
Stein, C. et al. Cloning and expression of human arylsulfatase A. J. Biol. Chem. 264, 1252–1259 (1989).
Kreysing, J. et al. Structure of the mouse arylsulfatase A gene and cDNA. Genomics 19, 249–256 (1994).
Zufferey, R. et al. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J. Virol. 72, 9873–9880 (1998).
Zufferey, R., Donello, J.E., Trono, D. & Hope, T.J. Woodchuck hepatitis virus posttranscriptional regulatory element enhances expression of transgenes delivered by retroviral vectors. J. Virol. 73, 2886–2892 (1999).
Deglon, N. et al. Self-inactivating lentiviral vectors with enhanced transgene expression as potential gene transfer system in Parkinson's disease. Hum. Gene Ther. 11, 179–190 (2000).
Zufferey, R., Nagy, D., Mandel, R.J., Naldini, L. & Trono, D. Multiply attenuated lentiviral vector achieves efficient gene delivery in vivo. Nature Biotechnol. 15, 871–875 (1997).
Dull, T. et al. A third-generation lentivirus vector with a conditional packaging system. J. Virol. 72, 8463–8471 (1998).
Monti, E. et al. Uptake and metabolism of a fluorescent sulfatide analogue in cultured skin fibroblasts. Biochim. Biophys. Acta 1124, 80–87 (1992).
Olton, D.S., Walker, J.A. & Gage, F.H. Hippocampal connections and spatial discrimination. Brain Res. 139, 215–308 (1978).
Brambilla, R. et al. A role for the Ras signaling pathway in synaptic transmission and long-term memory. Nature 390, 281–286 (1997).
Minichiello, L. et al. Essential role for TrkB receptors in hippocampus-mediated learning. Neuron 24, 401–414 (1999).
Cestari, V., Mele, A., Oliverio, A. & Castellano, C. Amygdala lesions block the effect of cocaine on memory in mice. Brain Research 713, 286–289 (1996).
Rogan, M.T., Stäubli, U.V. & LeDoux, J.E. Fear conditioning induces associative long-term potentiation in the amygdala. Nature 390, 604–607 (1997).
Kempermann, G., Kuhn, H.G. & Gage, F.H. More hippocampal neurons in adult mice living in an enriched evironment. Nature 386, 493–495 (1997).
Gould, E. et al. Hippocampal neurogenesis in adult Old World primates. Proc. Natl. Acad. Sci. USA 96, 5263–5267 (1999).
Nemni, R. et al. Effect of chronic tratment with recombinant inerleukin-2 on the central nervous system of adult and old mice. Brain. Res. 591, 248–252 (1992).
Venters, H.D. et al. A new mechanism of neurodegeneration: a proinflammatory cytokine inhibits receptor signaling by a survival peptide. Proc. Natl. Acad. Sci. USA 96, 9879–9884 (1999).
Kolesnick, R.N. & Kronke, M. Regulation of ceramide production and apoptosis. Ann. Rev. Physiol. 60, 643–665 (1998).
D'Hooge, R., Coenen, R., Gieselmann, V., Lullmann-Rauch, R. & De Deyn, P.P. Decline in brainstem auditory-evoked potentials coincides with loss of spiral ganglion cells in arylsulfatase A-deficient mice. Brain Res. 847, 352–356 (1999).
D'Hooge, R. et al. Neuromotor alterations and cerebellar deficits in aged arylsulfatase A-deficient transgenic mice. Neurosci. Lett. 273, 93–96 (1999).
Sands, M.S. et al. Enzyme replacement therapy for murine mucopolysaccharidosis type VII. J. Clin. Invest. 93, 2324–2231 (1994).
O'Connor, L.H. et al. Enzyme replacement therapy for murine mucopolysaccharidosis type VII leads to improvements in behavior and auditory function. J. Clin. Invest. 101, 1394–1400 (1998).
Taylor, R.M. & Woolfe, J.H. Decreased lysosomal storage in the adult MPS VII mouse brain in the vicinity of grafts of retroviral vector-corrected fibroblasts secreting high levels of β-glucuronidase. Nature Med. 3, 771–774 (1997).
Ohashi, T. et al. Adenovirus-mediated gene transfer and expression of human β-glucuronidase gene in the liver, spleen, and central nervous system in mucopolysaccharidosis type VII mice. Proc. Natl. Acad. Sci. USA 94, 1287–1292 (1997).
Bosch, A., Perret, E., Desmaris, N. & Heard, J.M. Long-term and significant correction of brain lesions in adult mucopolysaccharidosis Type VII mice using recombinant AAV vectors. Mol. Ther. 1, 63–71 (2000).
Baum, J., Dodgson, K.S. & Spencer, B. The assay of arylsulfatase A and B in human urine. Clin. Chim. Acta 4, 453–455 (1959).
Ferraresi, S. et al. Toward a transgenic mouse model of remyelination. Mult. Scler. 3, 80–83 (1997).
Papaioannou, V.E & Fox, J.G. Efficacy of tribromoethanol anesthesia in mice. Lab. Anim. Sci. 43,189–192 (1993).
Viani, P., Marchesini, S., Cestaro, B. & Gatt, S. Correlation of the dispersion state of pyrene cerebroside sulfate and its uptake and degradation by cultured cells. Biochim. Biophys. Acta 1002, 20–27 (1989).
McGaugh, J.L. & Landfield, P.W. Delayed development of amnesia following electroconvulsive shock. Physiol. Behav. 5, 1109–1113 (1970).
Vos, J.P. et al. Cultured oligodendrocytes metabolize a fluorescent analogue of sulphatide; inhibition by monensin. Biochim. Biophys. Acta 1126, 269–276 (1992).
Marchesini, S. et al. Synthesis, spectral properties and enzymatic hydrolysis of fluorescent derivatives of cerebroside sulfate containing long-wavelength-emission probes. Chem. Phys. Lipids 53, 165–175 (1990).
Acknowledgements
We thank V. Gieselmann for providing the As2−/− founder mice and for discussions on the model; T. Dull, M. Kelly and A. Follenzi for help with vector construction and production; G. Cusella for help with histology; U. Matzner for the hARSA antibody and suggestions for the immunocapture assay; and L. Wrabetz, A. Malgaroli, R. Brambilla and A. Mallamaci for helpful comments and critical reading of the manuscript. This work was supported by grants form Telethon, Italy, and from the Associazione Malattie Rare Mauro Baschirotto.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Consiglio, A., Quattrini, A., Martino, S. et al. In vivo gene therapy of metachromatic leukodystrophy by lentiviral vectors: correction of neuropathology and protection against learning impairments in affected mice. Nat Med 7, 310–316 (2001). https://doi.org/10.1038/85454
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/85454
This article is cited by
-
Retroviral vectors and transposons for stable gene therapy: advances, current challenges and perspectives
Journal of Translational Medicine (2016)
-
Arylsulfatase A Overexpressing Human iPSC-derived Neural Cells Reduce CNS Sulfatide Storage in a Mouse Model of Metachromatic Leukodystrophy
Molecular Therapy (2015)
-
Long-term correction of biochemical and neurological abnormalities in MLD mice model by neonatal systemic injection of an AAV serotype 9 vector
Gene Therapy (2014)
-
Increased concentration of the CSF Tau protein and its phosphorylated form in the late juvenile metachromatic leukodystrophy form: a case report
Journal of Neural Transmission (2012)
-
Gene Therapy of Chronic Granulomatous Disease: The Engraftment Dilemma
Molecular Therapy (2011)