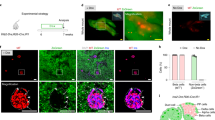
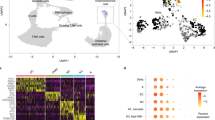
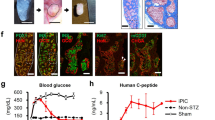
Abstract
The nature and even existence of adult pancreatic endocrine stem or progenitor cells is a subject of controversy in the field of beta-cell replacement for diabetes. One place to search for such cells is in the nonendocrine fraction of cells that remain after islet isolation, which consist of a mixture of epithelia and mesenchyme. Culture in G418 resulted in elimination of the mesenchymal cells, leaving a highly purified population of nonendocrine pancreatic epithelial cells (NEPECs). To evaluate their differentiation potential, NEPECs were heritably marked and transplanted under the kidney capsule of immunodeficient mice. When cotransplanted with fetal pancreatic cells, NEPECs were capable of endocrine differentiation. We found no evidence of beta-cell replication or cell fusion that could have explained the appearance of insulin positive cells from a source other than NEPECs. Nonendocrine-to-endocrine differentiation of NEPECs supports the existence of endocrine stem or progenitor cells within the epithelial compartment of the adult human pancreas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Shapiro, A.M. et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N. Engl. J. Med. 343, 230–238 (2000).
Hirshberg, B. et al. Benefits and risks of solitary islet transplantation for type 1 diabetes using steroid-sparing immunosuppression: the National Institutes of Health experience. Diabetes Care 26, 3288–3295 (2003).
Bonner-Weir, S., Baxter, L.A., Schuppin, G.T. & Smith, F.E. A second pathway for regeneration of adult exocrine and endocrine pancreas. A possible recapitulation of embryonic development. Diabetes 42, 1715–1720 (1993).
Guz, Y., Nasir, I. & Teitelman, G. Regeneration of pancreatic beta cells from intra-islet precursor cells in an experimental model of diabetes. Endocrinology 142, 4956–4968 (2001).
Sarvetnick, N.E. & Gu, D. Regeneration of pancreatic endocrine cells in interferon-gamma transgenic mice. Adv. Exp. Med. Biol. 321, 85–89 (1992).
Dor, Y., Brown, J., Martinez, O.I. & Melton, D.A. Adult pancreatic beta-cells are formed by self-duplication rather than stem-cell differentiation. Nature 429, 41–46 (2004).
Bonner-Weir, S. et al. In vitro cultivation of human islets from expanded ductal tissue. Proc. Natl. Acad. Sci. USA 97, 7999–8004 (2000).
Lardon, J., Huyens, N., Rooman, I. & Bouwens, L. Exocrine cell transdifferentiation in dexamethasone-treated rat pancreas. Virchows Arch. 444, 61–65 (2004).
Zulewski, H. et al. Multipotential nestin-positive stem cells isolated from adult pancreatic islets differentiate ex vivo into pancreatic endocrine, exocrine, and hepatic phenotypes. Diabetes 50, 521–533 (2001).
Lechner, A., Leech, C.A., Abraham, E.J., Nolan, A.L. & Habener, J.F. Nestin-positive progenitor cells derived from adult human pancreatic islets of Langerhans contain side population (SP) cells defined by expression of the ABCG2 (BCRP1) ATP-binding cassette transporter. Biochem. Biophys. Res. Commun. 293, 670–674 (2002).
Abraham, E.J., Leech, C.A., Lin, J.C., Zulewski, H. & Habener, J.F. Insulinotropic hormone glucagon-like peptide-1 differentiation of human pancreatic islet-derived progenitor cells into insulin-producing cells. Endocrinology 143, 3152–3161 (2002).
Rajagopal, J., Anderson, W.J., Kume, S., Martinez, O.I. & Melton, D.A. Insulin staining of ES cell progeny from insulin uptake. Science 299, 363 (2003).
Levine, F. & Mercola, M. No pancreatic endocrine stem cells? N. Engl. J. Med. 351, 1024–1026 (2004).
Scharfmann, R. Control of early development of the pancreas in rodents and humans: implications of signals from the mesenchyme. Diabetologia 43, 1083–1092 (2000).
Beattie, G.M. et al. Acid beta-galactosidase: a developmentally regulated marker of endocrine cell precursors in the human fetal pancreas. J. Clin. Endocrinol. Metab. 78, 1232–1240 (1994).
Jensen, J. Gene regulatory factors in pancreatic development. Dev. Dyn. 229, 176–200 (2004).
Tuch, B.E., Ng, A.B., Jones, A. & Turtle, J.R. Histologic differentiation of human fetal pancreatic explants transplanted into nude mice. Diabetes 33, 1180–1187 (1984).
Sandler, S. et al. Tissue culture of human fetal pancreas. Effects of human serum on development and endocrine function of isletlike cell clusters. Diabetes 36, 1401–1407 (1987).
Gao, R., Ustinov, J., Korsgren, O. & Otonkoski, T. In vitro neogenesis of human islets reflects the plasticity of differentiated human pancreatic cells. Diabetologia 48, 2296–2304 (2005).
Gao, R. et al. Characterization of endocrine progenitor cells and critical factors for their differentiation in human adult pancreatic cell culture. Diabetes 52, 2007–2015 (2003).
Heimberg, H. et al. Adult human pancreatic duct and islet cells exhibit similarities in expression and differences in phosphorylation and complex formation of the homeodomain protein Ipf-1. Diabetes 49, 571–579 (2000).
Heremans, Y. et al. Recapitulation of embryonic neuroendocrine differentiation in adult human pancreatic duct cells expressing neurogenin 3. J. Cell Biol. 159, 303–312 (2002).
Bouwens, L., Lu, W.G. & De Krijger, R. Proliferation and differentiation in the human fetal endocrine pancreas. Diabetologia 40, 398–404 (1997).
Street, C.N. et al. Enriched human pancreatic ductal cultures obtained from selective death of acinar cells express pancreatic and duodenal homeobox gene-1 age-dependently. The Review of Diabetic Studies 1, 66–79 (2004).
Bogdani, M. et al. Formation of insulin-positive cells in implants of human pancreatic duct cell preparations from young donors. Diabetologia 46, 830–838 (2003).
Halban, P.A. Cellular sources of new pancreatic beta cells and therapeutic implications for regenerative medicine. Nat. Cell Biol. 6, 1021–1025 (2004).
Halaban, R. & Alfano, F.D. Selective elimination of fibroblasts from cultures of normal human melanocytes. In Vitro 20, 447–450 (1984).
Klein, T. et al. Nestin is expressed in vascular endothelial cells in the adult human pancreas. J. Histochem. Cytochem. 51, 697–706 (2003).
Lardon, J., Rooman, I. & Bouwens, L. Nestin expression in pancreatic stellate cells and angiogenic endothelial cells. Histochem. Cell Biol. 117, 535–540 (2002).
Humphrey, R.K. et al. Characterization and isolation of promoter-defined nestin-positive cells from the human fetal pancreas. Diabetes 52, 2519–2525 (2003).
Esni, F., Stoffers, D.A., Takeuchi, T. & Leach, S.D. Origin of exocrine pancreatic cells from nestin-positive precursors in developing mouse pancreas. Mech. Dev. 121, 15–25 (2004).
Delacour, A., Nepote, V., Trumpp, A. & Herrera, P.L. Nestin expression in pancreatic exocrine cell lineages. Mech. Dev. 121, 3–14 (2004).
Leibowitz, G. et al. Gene transfer to human pancreatic endocrine cells using viral vectors. Diabetes 48, 745–753 (1999).
Lechner, A. et al. No evidence for significant transdifferentiation of bone marrow into pancreatic beta-cells in vivo. Diabetes 53, 616–623 (2004).
Wang, X. et al. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature 422, 897–901 (2003).
Wurmser, A.E. & Gage, F.H. Stem cells: cell fusion causes confusion. Nature 416, 485–487 (2002).
Wurmser, A.E. et al. Cell fusion-independent differentiation of neural stem cells to the endothelial lineage. Nature 430, 350–356 (2004).
Boyer, B., Tucker, G.C., Valles, A.M., Franke, W.W. & Thiery, J.P. Rearrangements of desmosomal and cytoskeletal proteins during the transition from epithelial to fibroblastoid organization in cultured rat bladder carcinoma cells. J. Cell Biol. 109, 1495–1509 (1989).
Gershengorn, M.C. et al. Epithelial-to-mesenchymal transition generates proliferative human islet precursor cells. Science 306, 2261–2264 (2004).
Halvorsen, T.L., Beattie, G.M., Lopez, A.D., Hayek, A. & Levine, F. Accelerated telomere shortening and senescence in human pancreatic islet cells stimulated to divide in vitro. J. Endocrinol. 166, 103–109 (2000).
Finegood, D.T., Scaglia, L. & Bonner-Weir, S. Dynamics of beta-cell mass in the growing rat pancreas. Estimation with a simple mathematical model. Diabetes 44, 249–256 (1995).
Tyrberg, B., Andersson, A. & Borg, L.A. Species differences in susceptibility of transplanted and cultured pancreatic islets to the β-cell toxin alloxan. Gen. Comp. Endocrinol. 122, 238–251 (2001).
Butler, A.E. et al. Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 52, 102–110 (2003).
Beattie, G.M., Rubin, J.S., Mally, M.I., Otonkoski, T. & Hayek, A. Regulation of proliferation and differentiation of human fetal pancreatic islet cells by extracellular matrix, hepatocyte growth factor, and cell-cell contact. Diabetes 45, 1223–1228 (1996).
Wang, S. et al. Isolation and characterization of a cell line from the epithelial cells of the human fetal pancreas. Cell Transplant. 6, 59–67 (1997).
Beattie, G.M., Butler, C. & Hayek, A. Morphology and function of cultured human fetal pancreatic cells transplanted into athymic mice: a longitudinal study. Cell Transplant. 3, 421–425 (1994).
Acknowledgements
We thank the islet isolation centers in Edmonton, Seattle, Minnesota and the Whittier Institute for providing human islets and nonendocrine cells, and C. Wright, O. Madsen, M. Magnuson and J. Hutton for providing antibodies and for discussions. We thank R. Bajpai for discussion and virus preparation. This work was funded by grants from the US National Institute of Diabetes and Digestive and Kidney Diseases and the Juvenile Diabetes Research Foundation (to F.L., M.M. and J.R.T.L.), the Beta Cell Biology Consortium (to F.L.) and the Larry L. Hillblom Foundation (to B.T.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Insulin staining of NEPCs before and after transplantation. (PDF 297 kb)
Supplementary Fig. 2
Characterization of NEPECs. (PDF 243 kb)
Supplementary Fig. 3
Proliferation of adult and fetal cells. (PDF 356 kb)
Supplementary Fig. 4
Lentiviral infections. (PDF 356 kb)
Supplementary Fig. 5
Additional analyses of NEPEC/ICC transplants. (PDF 291 kb)
Supplementary Fig. 6
Confocal sectioning of NEPECs after transplantation with ICCs. (PDF 431 kb)
Supplementary Fig. 7
Detection of fusion between NEPECs and fetal cells. (PDF 139 kb)
Rights and permissions
About this article
Cite this article
Hao, E., Tyrberg, B., Itkin-Ansari, P. et al. Beta-cell differentiation from nonendocrine epithelial cells of the adult human pancreas. Nat Med 12, 310–316 (2006). https://doi.org/10.1038/nm1367
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1367
This article is cited by
-
Pre-existing beta cells but not progenitors contribute to new beta cells in the adult pancreas
Nature Metabolism (2021)
-
Current progress in stem cell therapy for type 1 diabetes mellitus
Stem Cell Research & Therapy (2020)
-
Prognostic relevance of UCH-L1 and α-internexin in pancreatic neuroendocrine tumors
Scientific Reports (2017)
-
Cell therapies for pancreatic beta-cell replenishment
Italian Journal of Pediatrics (2016)
-
Geniposide promotes beta-cell regeneration and survival through regulating β-catenin/TCF7L2 pathway
Cell Death & Disease (2015)